Decoding Caspase Activation: A Strategic Guide to Distinguish Apoptosis from Pyroptosis in Research and Drug Discovery
This article provides a comprehensive guide for researchers and drug development professionals on distinguishing between apoptotic and pyroptotic caspase activation.
Decoding Caspase Activation: A Strategic Guide to Distinguish Apoptosis from Pyroptosis in Research and Drug Discovery
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on distinguishing between apoptotic and pyroptotic caspase activation. It covers the foundational biology of caspases as central regulators of programmed cell death, detailing their specific roles in non-inflammatory apoptosis versus highly inflammatory pyroptosis. The content explores established and emerging methodologies for detecting and quantifying caspase activity, addresses common experimental challenges in differentiating these pathways, and presents validation frameworks for confirming cell death modality. By synthesizing current knowledge on caspase crosstalk, pathway switching, and therapeutic targeting, this resource aims to enhance experimental accuracy and inform the development of novel therapeutics for cancer, inflammatory disorders, and neurodegenerative diseases.
The Molecular Architects: Defining Caspase Roles in Apoptosis and Pyroptosis Pathways
Caspases as Master Regulators of Programmed Cell Death
Troubleshooting Guides and FAQs
Frequently Asked Questions
Q1: My experiment suggests caspase-1 is activating, but I'm not observing pyroptosis. What could be happening? A1: Caspase-1 can activate different cell death pathways depending on cellular context and available substrates. In the absence of its primary pyroptotic substrate, Gasdermin D (GSDMD), caspase-1 can trigger apoptosis by activating caspases-3 and -7 [1]. To investigate:
- Verify GSDMD expression and cleavage status via Western blot
- Check for apoptotic markers (PARP cleavage, caspase-3/7 activation)
- Consider that caspase-1 can induce apoptosis when GSDMD is absent or inactive
Q2: Why do I detect caspase-3 activation alongside pyroptotic markers? Is this possible? A2: Yes, this is biologically plausible. Caspase-3, traditionally an apoptotic executioner, can induce pyroptosis by cleaving Gasdermin E (GSDME) [2]. The cell death mode depends on GSDME expression levels:
- High GSDME expression: Caspase-3 cleavage of GSDME induces pyroptosis
- Low GSDME expression: Caspase-3 activation leads to classical apoptosis This pathway serves as a molecular switch between apoptotic and pyroptotic cell death [2].
Q3: How does caspase-8 influence cross-talk between different programmed cell death pathways? A3: Caspase-8 serves as a critical molecular switch between apoptosis, necroptosis, and pyroptosis [3] [4]. Its functions include:
- Initiating extrinsic apoptosis by activating caspase-3
- Cleaving GSDMC to induce pyroptosis
- Inhibiting necroptosis by cleaving RIPK1 and RIPK3
- Converting apoptosis to pyroptosis by cleaving GSDMD The specific pathway activated depends on cellular context and inhibition status [3].
Q4: What are the key methodological considerations when detecting caspase activation in my experiments? A4: Traditional antibody-based methods (Western blot) provide fundamental insights but have limitations [5]. For comprehensive analysis:
- Use multiple complementary techniques (antibody-based, activity assays, live imaging)
- Consider temporal dynamics - caspase activation occurs at different timepoints
- Employ spatial monitoring techniques (FRET sensors, live-cell imaging) to track activation in real-time
- Validate with mass spectrometry for identifying caspase substrates and cleavage products [5]
Experimental Protocols for Distinguishing Apoptotic vs. Pyroptotic Caspase Activation
Protocol 1: Differential Caspase Activation Profiling
Objective: Distinguish between apoptotic and pyroptotic caspase activation patterns in cell culture models.
Materials:
- Appropriate cell culture system (primary macrophages, THP-1, RAW 264.7)
- Caspase inhibitors: Z-VAD-FMK (pan-caspase), Ac-YVAD-CHO (caspase-1 selective), Ac-DEVD-CHO (caspase-3 selective)
- LDH release assay kit for membrane integrity
- Western blot equipment and antibodies
Procedure:
- Seed cells at appropriate density and apply apoptotic (etoposide, 50µM) or pyroptotic (Val-boroPro, 10µM; LPS transfection) stimuli [1]
- Pre-treat parallel samples with caspase inhibitors 1 hour prior to stimulation
- Collect samples at 0, 2, 4, 8, and 16-hour timepoints
- Analyze by:
- Western blot for caspase cleavage (caspase-1, -3, -8), substrate cleavage (GSDMD, GSDME, PARP)
- LDH release to quantify membrane integrity
- Microscopy for morphological assessment (membrane blebbing vs. swelling)
Expected Results:
- Apoptotic stimuli: Caspase-3/7 activation, PARP cleavage, membrane blebbing, minimal LDH release
- Pyroptotic stimuli: Caspase-1/4/11 activation, GSDMD/GSDME cleavage, cell swelling, significant LDH release
Protocol 2: Gasdermin Cleavage and Pore Formation Assay
Objective: Determine gasdermin family member involvement in caspase-mediated cell death.
Materials:
- GSDMD and GSDME knockout cell lines (CRISPR/Cas9 generated)
- Antibodies against GSDMD-NT, GSDME-NT, full-length gasdermins
- Propidium iodide (PI) uptake assay
- IL-1β/IL-18 ELISA kits
Procedure:
- Treat wild-type and gasdermin-deficient cells with apoptotic and pyroptotic stimuli
- Monitor PI uptake by flow cytometry every 30 minutes for 6 hours
- Collect supernatants for cytokine measurement (IL-1β, IL-18) by ELISA
- Analyze cell lysates by Western blot for gasdermin cleavage
- Use super-resolution microscopy to visualize plasma membrane pores
Interpretation:
- GSDMD-dependent pyroptosis: Caspase-1/4/5/11 cleaves GSDMD, releasing NT fragment that forms plasma membrane pores, enabling PI uptake and IL-1β/IL-18 release [6] [7]
- GSDME-dependent pyroptosis: Caspase-3 cleaves GSDME, producing pyroptotic morphology despite apoptotic initiation [2]
Table 1: Caspase Functions in Programmed Cell Death Pathways
| Caspase | Primary Pathway | Key Substrates | Morphological Features | Inflammatory Output |
|---|---|---|---|---|
| Caspase-1 | Pyroptosis | GSDMD, pro-IL-1β, pro-IL-18 | Cell swelling, membrane rupture | High (IL-1β, IL-18 release) |
| Caspase-3 | Apoptosis/Pyroptosis | PARP, GSDME, GSDMD | Blebbing (apoptosis) or swelling (pyroptosis) | Low (apoptosis) or High (pyroptosis) |
| Caspase-8 | Apoptosis/Pyroptosis Switch | Caspase-3, GSDMC, RIPK1 | Variable based on context | Context-dependent |
| Caspase-4/5/11 | Pyroptosis (non-canonical) | GSDMD | Cell swelling, membrane rupture | High (DAMP release) |
| Caspase-9 | Apoptosis (intrinsic) | Caspase-3/7 | Membrane blebbing, chromatin condensation | Low |
Table 2: Key Experimental Markers for Distinguishing Cell Death Pathways
| Parameter | Apoptosis | Pyroptosis | Necroptosis |
|---|---|---|---|
| Key Caspases | Caspase-3/7/8/9 | Caspase-1/4/5/11 | Caspase-8 inhibition |
| Effector Proteins | - | GSDMD/GSDME-NT | p-MLKL |
| Membrane Integrity | Maintained then permeabilized | Rapid pore formation | Membrane rupture |
| Nuclear Morphology | Condensation, fragmentation | Condensation | Disintegration |
| Inflammation | Minimal | Robust | Robust |
| LDH Release | Late | Early | Early |
| Key Assays | PARP cleavage, Annexin V | GSDMD cleavage, IL-1β release | p-MLKL detection |
Research Reagent Solutions
Table 3: Essential Research Reagents for Caspase and Cell Death Studies
| Reagent | Function/Application | Specific Examples |
|---|---|---|
| Caspase Inhibitors | Pathway dissection, mechanistic studies | Z-VAD-FMK (pan-caspase), Ac-YVAD-CHO (caspase-1), Ac-DEVD-CHO (caspase-3) [8] |
| Activity Assays | Caspase activation quantification | Fluorogenic substrates (WEHD- AFC for caspase-1, DEVD-AFC for caspase-3) [5] |
| Antibodies | Detection of cleavage, activation | Anti-cleaved caspase-3, anti-GSDMD-NT, anti-p-MLKL |
| Live-Cell Imaging Probes | Real-time activation monitoring | FLICA reagents, FRET-based caspase sensors [5] |
| Genetic Tools | Pathway requirement determination | CRISPR/Cas9 knockout cells (GSDMD-/-, caspase-1-/-) [1] |
| Cytokine Detection | Inflammatory output measurement | IL-1β, IL-18 ELISA kits [7] |
| Membrane Integrity Assays | Pyroptosis/necrosis quantification | LDH release, propidium iodide uptake [7] |
Signaling Pathway Visualizations
Caspase Regulation of Cell Death Pathways
Experimental Decision Workflow
Technical Support Center
Troubleshooting Guides & FAQs
Q1: My Western blot for cleaved Caspase-3 is inconsistent. What could be the cause? A: Inconsistent detection of cleaved Caspase-3 (17/19 kDa) is common. Key considerations:
- Sample Preparation: Apoptosis is a rapid, transient process. Ensure you are harvesting cells at the correct time point. Use positive controls (e.g., Staurosporine-treated cells). Always include protease and phosphatase inhibitors in your lysis buffer.
- Antibody Specificity: Many antibodies cross-react with other cleaved caspases or unrelated proteins. Validate your antibody using Caspase-3 knockout cell lines or specific siRNA knockdown.
- Unexpected Cleavage in Pyroptosis: Gasdermin E (GSDME), when cleaved by Caspase-3, can induce pyroptosis. If your cells express GSDME, Caspase-3 activation may lead to pyroptotic lysis instead of classic apoptotic morphology, complicating interpretation.
Q2: How can I distinguish between Death Receptor (Extrinsic) and Mitochondrial (Intrinsic) apoptosis pathways in my experiment? A: You must assay for the specific initiator caspases and their upstream regulators.
- Assay for Initiator Caspases:
- Extrinsic Pathway: Measure Caspase-8 activation (Western blot for cleaved fragments, or a Caspase-8 activity assay using IETD-pNA/-AFC substrates).
- Intrinsic Pathway: Measure Caspase-9 activation (Western blot, or LEHD-pNA/-AFC substrates).
- Inhibit Key Nodes: Use specific inhibitors.
- Caspase-8 inhibitor Z-IETD-FMK can block extrinsic apoptosis.
- Bcl-2 overexpression or BH3 mimetics can modulate the intrinsic pathway.
- Monitor Pathway-Specific Proteins:
- Extrinsic: Look for FADD recruitment and DISC formation by immunoprecipitation.
- Intrinsic: Measure cytochrome c release from mitochondria into the cytosol via fractionation or immunofluorescence.
Q3: I see Caspase-1 and Caspase-3 activation simultaneously. Is my cell undergoing apoptosis or pyroptosis? A: This is a critical distinction for your thesis. Simultaneous activation suggests inflammatory apoptosis or a mixed cell death phenotype.
- Key Differentiator: Check for Gasdermin D (GSDMD) cleavage.
- Pyroptosis: Caspase-1 cleaves GSDMD to form pores in the plasma membrane, leading to lytic cell death. Detect the GSDMD-NT fragment.
- Apoptosis: Executioner caspases (Caspase-3/7) cleave and inactivate GSDMD, preventing pyroptosis.
- Morphology: Use microscopy. Apoptosis features membrane blebbing and apoptotic bodies without significant plasma membrane rupture until late stages. Pyroptosis features rapid plasma membrane pore formation, cell swelling, and lysis.
- IL-1β/IL-18 Secretion: Pyroptosis is characterized by the release of mature IL-1β and IL-18 via GSDMD pores. Measure these cytokines in the supernatant by ELISA.
Q4: Why is my Caspase-9 activity assay negative despite clear signs of apoptosis? A: This can occur due to several reasons:
- Alternative Activation Pathways: The Mitochondrial pathway can sometimes be amplified by Caspase-8 cleavage of Bid (tBid), leading to mitochondrial outer membrane permeabilization (MOMP) without strong, direct Caspase-9 activation.
- Feedback Loop: Executioner caspases (Caspase-3/7) can cleave and further activate Caspase-9 in a feedback amplification loop. The initial, transient activation of Caspase-9 might be below your detection limit.
- Inhibitor of Apoptosis Proteins (IAPs): XIAP potently binds and inhibits Caspase-9. Use SMAC mimetics in your experiment to antagonize IAPs and unmask Caspase-9 activity.
Data Presentation
Table 1: Key Characteristics of Apoptotic Caspases
| Caspase | Role | Zymogen Pro-form (kDa) | Active Subunits (kDa) | Preferred Tetrapeptide Substrate Motif (P4-P1) | Key Endogenous Substrates |
|---|---|---|---|---|---|
| Caspase-8 | Initiator (Extrinsic) | 55 | 18, 10 | (I/L/V)ETD | Caspase-3, Caspase-7, Bid, RIPK1 |
| Caspase-9 | Initiator (Intrinsic) | 45-50 | 35, 10 (with Apaf-1) | (I/L/V)EHD | Caspase-3, Caspase-7 |
| Caspase-10 | Initiator (Extrinsic) | 55, 59 | 17-20, 10-12 | (I/L/V)EAD | Caspase-3, Caspase-7, Bid |
| Caspase-3 | Executioner | 32-35 | 17, 12 | DEVD | PARP, DFF45/ICAD, GSDME, PKCδ |
| Caspase-6 | Executioner | 34 | 18, 11 | VEID | Lamin A/C, Caspase-8, Caspase-9 |
| Caspase-7 | Executioner | 35 | 20, 11 | DEVD | PARP, DFF45/ICAD |
Table 2: Distinguishing Apoptotic vs. Pyroptotic Caspase Activation
| Feature | Apoptosis | Pyroptosis |
|---|---|---|
| Key Initiator Caspases | Caspase-8, -9, -10 | Caspase-1, -4, -5, -11 |
| Key Executioner Caspases | Caspase-3, -6, -7 | (Caspase-1, -4, -5, -11 cleave GSDMD directly) |
| Key Substrate | PARP, Lamin, DFF45 | Gasdermin D (GSDMD) |
| Gasdermin Protein Fate | GSDMD is cleaved and inactivated by Caspase-3/7 | GSDMD is cleaved and activated (GSDMD-NT forms pores) |
| Morphology | Cell shrinkage, membrane blebbing, apoptotic bodies | Cell swelling, plasma membrane pore formation, lysis |
| Inflammation | Generally non-inflammatory or anti-inflammatory | Highly inflammatory (release of IL-1β, IL-18, DAMPs) |
Experimental Protocols
Protocol 1: Differentiating Apoptosis and Pyroptosis via Immunoblotting
Objective: To simultaneously assess key markers of apoptosis (cleaved Caspase-3) and pyroptosis (cleaved GSDMD) from the same sample.
Materials:
- RIPA Lysis Buffer with protease inhibitors (e.g., PMSF, Aprotinin, Leupeptin).
- Precast SDS-PAGE gels (4-20% gradient recommended).
- Antibodies: Anti-Cleaved Caspase-3 (Asp175), Anti-GSDMD (Full length and N-terminal), Anti-β-Actin (loading control).
Method:
- Stimulation: Treat cells with a pro-apoptotic stimulus (e.g., 1 µM Staurosporine, 6 hrs) or a pro-pyroptotic stimulus (e.g., 500 ng/mL LPS + 5 mM ATP for 4 hrs in macrophages).
- Harvesting: Gently scrape adherent cells in ice-cold PBS. Pellet cells by centrifugation (500 x g, 5 min, 4°C).
- Lysis: Lyse cell pellet in 100-200 µL RIPA buffer on ice for 30 min. Vortex briefly every 10 min.
- Clarification: Centrifuge at 14,000 x g for 15 min at 4°C. Transfer the supernatant (whole cell lysate) to a new pre-chilled tube.
- Protein Quantification: Use a BCA assay to determine protein concentration.
- Western Blot: Load 20-30 µg of protein per lane. Resolve by SDS-PAGE, transfer to PVDF membrane, and probe with primary antibodies overnight at 4°C.
- Interpretation:
- Apoptosis: Presence of ~17/19 kDa Cleaved Caspase-3 bands.
- Pyroptosis: Presence of ~30-35 kDa GSDMD-NT fragment. Loss of full-length GSDMD may also be observed.
Protocol 2: Measuring Caspase Activity Using Fluorogenic Assays
Objective: To quantitatively measure the activity of specific caspases in cell lysates.
Materials:
- Caspase Lysis Buffer (50 mM HEPES, 100 mM NaCl, 0.1% CHAPS, 10 mM DTT, 1 mM EDTA, pH 7.4).
- Fluorogenic substrates: Ac-DEVD-AFC (for Caspase-3/7), Ac-IETD-AFC (for Caspase-8), Ac-LEHD-AFC (for Caspase-9).
- Black 96-well plates.
Method:
- Prepare Lysates: Lyse treated cells in Caspase Lysis Buffer (without detergents that inhibit activity). Use 50-100 µL per 1x10^6 cells.
- Prepare Reaction: In a black 96-well plate, add:
- 50 µL of cell lysate (adjust protein concentration to be equal across samples).
- 50 µL of Caspase Lysis Buffer containing 50 µM of the respective AFC-conjugated substrate.
- Incubate and Measure: Incubate the plate at 37°C for 1-2 hours. Measure fluorescence (Ex: 400 nm, Em: 505 nm) every 15-30 minutes using a plate reader.
- Analysis: Plot fluorescence over time. The slope of the linear range represents caspase activity. Normalize to protein concentration or vehicle-treated control.
Mandatory Visualization
Diagram Title: Apoptotic Signaling Pathways
Diagram Title: Cell Fate Decision: Apoptosis vs Pyroptosis
The Scientist's Toolkit
Table 3: Essential Research Reagents for Apoptotic Caspase Studies
| Reagent | Function & Application |
|---|---|
| Z-VAD-FMK (Pan-Caspase Inhibitor) | A cell-permeable, irreversible inhibitor of all caspases. Used as a broad control to confirm caspase-dependent cell death. |
| Ac-DEVD-AFC/pNA (Caspase-3/7 Substrate) | Fluorogenic (AFC) or colorimetric (pNA) substrate used to measure the enzymatic activity of executioner caspases in lysates. |
| Ac-IETD-AFC/pNA (Caspase-8 Substrate) | Substrate for measuring the activity of the initiator caspase-8 from the extrinsic pathway. |
| Anti-Cleaved Caspase-3 (Asp175) Antibody | A widely used antibody for detecting the activated form of Caspase-3 by Western blot or immunofluorescence. A hallmark of apoptosis. |
| Anti-Gasdermin D (N-Terminal) Antibody | Critical reagent for distinguishing pyroptosis from apoptosis. Detects the active, pore-forming fragment of GSDMD. |
| Recombinant Active Caspase-3 | Used as a positive control in Western blots, activity assays, or for in vitro cleavage assays to identify novel substrates. |
| Staurosporine | A potent, broad-spectrum kinase inducer that reliably triggers the intrinsic apoptotic pathway; a standard positive control for apoptosis. |
| SMAC Mimetics (e.g., Birinapant) | Small molecules that antagonize IAP proteins, thereby promoting Caspase-9 activation and sensitizing cells to intrinsic apoptosis. |
Frequently Asked Questions (FAQs)
Q1: How can I definitively distinguish between Caspase-1 and Caspase-4/5/11 activation in my human macrophage cultures? A1: Use a combination of genetic and pharmacological tools. Caspase-1 is activated by canonical inflammasomes (e.g., NLRP3, AIM2), while Caspase-4/5 are direct cytosolic LPS sensors.
- Genetic: Use CRISPR/Cas9 to generate single knockouts of CASP1, CASP4, or CASP5.
- Pharmacological: Use the Caspase-1 specific inhibitor VX-765 (Belnacasan). It will not inhibit Caspase-4/5-driven pyroptosis.
- Stimuli: Use intracellular LPS delivery (e.g., transfection) to selectively activate Caspase-4/5. Use NLRP3 activators like nigericin + LPS priming for Caspase-1.
Q2: My LDH release assay is positive, but I see no GSDMD cleavage by western blot. What could be wrong? A2: This discrepancy suggests potential assay interference or non-pyroptotic cell death.
- Check Antibody Specificity: Ensure your anti-GSDMD antibody recognizes both full-length and the cleaved N-terminal fragment. The N-terminal fragment may run at ~30-35 kDa and can be difficult to detect if the cleavage is inefficient or the antibody is poor.
- Alternative Death Pathways: The LDH release could be from apoptosis (secondary necrosis) or necroptosis. Perform additional assays:
- Annexin V/PI Staining: Apoptotic cells are Annexin V+/PI- (early) or Annexin V+/PI+ (late). Pyroptotic cells are often Annexin V-/PI+ rapidly.
- Necroptosis Inhibition: Use the RIPK1 inhibitor Necrostatin-1 to rule out necroptosis.
- Time Course: Perform a time-course experiment. GSDMD cleavage is rapid and transient; you may have missed the peak.
Q3: Why do I detect Caspase-3/7 activation in my model of LPS-transfected macrophages, which should be pure pyroptosis? A3: Caspase-3 activation can occur downstream of pyroptosis as a secondary event.
- Bystander Effect: Pyroptotic cells release ATP and other molecules that can activate the NLRP3 inflammasome and Caspase-1 in neighboring cells, leading to a complex death milieu.
- Cross-talk with Apoptosis: In some contexts, Caspase-1 can cleave and activate Caspase-3. Furthermore, GSDMD-derived pores can lead to mitochondrial damage and apoptosome formation.
- Experimental Confirmation: To confirm pyroptosis is the primary death mechanism, use a GSDMD inhibitor (e.g., Necrosulfonamide, Disulfiram) or GSDMD knockout cells. If cell death and IL-1β release are abolished, pyroptosis is the primary driver.
Q4: What is the best positive control for studying Caspase-11 (mouse) or Caspase-4/5 (human) mediated pyroptosis? A4: The most specific positive control is transfection of ultrapure LPS into the cytoplasm.
- Method: Use a liposome-based transfection reagent (e.g., Lipofectamine 2000, FuGENE HD) or electroporation to deliver purified LPS (e.g., E. coli O111:B4) directly into the cytosol of murine or human macrophages.
- Key Readouts:
- Cell swelling and LDH release (pyroptosis).
- Cleavage of GSDMD (not GSDME).
- Secretion of IL-1α and HMGB1 (but not Caspase-1-dependent IL-1β, unless a second signal is present).
Troubleshooting Guide
Problem: Inconsistent pyroptosis induction with nigericin.
- Potential Cause 1: Insufficient Priming.
- Solution: Ensure cells are properly "primed" with a TLR agonist (e.g., LPS for 2-4 hours) to upregulate NLRP3 and pro-IL-1β before adding nigericin.
- Potential Cause 2: Serum Concentration.
- Solution: High serum concentrations can sequester nigericin. Reduce serum to 1-2% during nigericin treatment.
- Potential Cause 3: Potassium Depletion.
- Solution: NLRP3 activation by nigericin requires potassium efflux. Ensure your treatment buffer contains physiological levels of K+ unless the protocol specifically calls for a low-K+ buffer.
Problem: High background IL-1β secretion in untreated controls.
- Potential Cause 1: LPS Contamination.
- Solution: Use endotoxin-free reagents, tips, and tubes. Test culture media and reagents for LPS using an LAL assay.
- Potential Cause 2: Mechanical Stress.
- Solution: Avoid excessive pipetting or vortexing of cells, which can activate inflammasomes. Handle cells gently.
- Potential Cause 3: Mycoplasma Contamination.
- Solution: Routinely test cell cultures for mycoplasma, a potent inducer of inflammatory cytokine secretion.
Table 1: Key Characteristics of Pyroptotic Caspases
| Feature | Caspase-1 (Human/Mouse) | Caspase-4/5 (Human) | Caspase-11 (Mouse) |
|---|---|---|---|
| Activator | Canonical Inflammasomes (NLRP3, AIM2, etc.) | Direct Cytosolic LPS | Direct Cytosolic LPS |
| Upstream Adaptor | ASC | None (Direct) | None (Direct) |
| Key Target | Pro-IL-1β, Pro-IL-18, GSDMD | GSDMD | GSDMD |
| Inhibitor | VX-765, Ac-YVAD-CMK | Not well characterized; Z-WEHD-FMK (pan) | Not well characterized |
| Primary Readout | IL-1β/18 secretion, Pyroptosis | Pyroptosis, IL-1α secretion | Pyroptosis, IL-1α secretion |
Table 2: Markers to Distinguish Apoptosis from Pyroptosis
| Assay | Apoptosis | Pyroptosis |
|---|---|---|
| Morphology | Cell shrinkage, membrane blebbing | Cell swelling, plasma membrane rupture |
| Key Caspase | Caspase-3/7, Caspase-8, Caspase-9 | Caspase-1, Caspase-4/5/11 |
| Gasdermin Cleavage | GSDME (by Caspase-3) | GSDMD (by Caspase-1/4/5/11) |
| Membrane Integrity | Intact until late stage (Annexin V+ / PI-) | Rapidly permeabilized (Annexin V- / PI+) |
| Cytokine Release | Generally anti-inflammatory | Pro-inflammatory (IL-1β, IL-18) |
Experimental Protocols
Protocol 1: Detecting GSDMD Cleavage by Western Blot
- Objective: To confirm pyroptotic caspase activation via cleavage of its primary effector, GSDMD.
- Materials: RIPA Lysis Buffer, protease inhibitors, SDS-PAGE gel, transfer apparatus, anti-GSDMD antibody (cleavage-specific antibodies are available).
- Steps:
- Stimulate cells (e.g., THP-1 macrophages, BMDMs) with your pyroptosis inducer (e.g., 5µM nigericin for 45 min, or transfection with 1µg/mL LPS for 4-6 hours).
- Lyse cells directly in 1X Laemmli buffer or RIPA buffer with inhibitors.
- Boil samples for 5-10 minutes.
- Separate proteins by SDS-PAGE (12-15% gel recommended).
- Transfer to PVDF membrane.
- Block with 5% BSA in TBST for 1 hour.
- Incubate with primary anti-GSDMD antibody (1:1000) overnight at 4°C.
- Wash and incubate with HRP-conjugated secondary antibody (1:5000) for 1 hour.
- Develop with ECL reagent. Look for the appearance of the ~30-35 kDa N-terminal fragment (GSDMD-NT) and the decrease in full-length GSDMD (~53 kDa).
Protocol 2: LDH Release Assay for Pyroptosis Quantification
- Objective: To quantitatively measure plasma membrane rupture, a hallmark of pyroptosis.
- Materials: Commercial LDH Cytotoxicity Assay Kit.
- Steps:
- Seed cells in a 96-well plate.
- Treat cells with your stimulus. Include a "High Control" (cells lysed with lysis buffer from the kit) and a "Low Control" (media only).
- Following the treatment period, centrifuge the plate at 250xg for 4 minutes.
- Carefully transfer 50-100 µL of supernatant from each well to a new 96-well plate.
- Add the LDH reaction mixture from the kit to each well containing supernatant.
- Incubate for 30 minutes at room temperature, protected from light.
- Measure absorbance at 490 nm and 680 nm (reference wavelength).
- Calculate % Cytotoxicity: [(Experimental - Low Control) / (High Control - Low Control)] x 100.
Pathway & Workflow Visualizations
Pyroptosis Signaling Pathways
Pyroptosis Assay Workflow
The Scientist's Toolkit
Table 3: Essential Research Reagents for Pyroptosis Studies
| Reagent | Function / Target | Example Use Case |
|---|---|---|
| Ultrapure LPS | TLR4 agonist for priming; Transfected for non-canonical pathway. | Priming THP-1 cells before nigericin treatment. |
| Nigericin | K+ ionophore; potent NLRP3 activator. | Inducing canonical pyroptosis in primed macrophages. |
| VX-765 (Belnacasan) | Selective Caspase-1 inhibitor. | Confirming Caspase-1-dependent events. |
| Disulfiram | FDA-approved drug that inhibits GSDMD pore formation. | Blocking the final step of pyroptosis. |
| Anti-GSDMD Antibody | Detects full-length and cleaved GSDMD. | Western blot confirmation of pyroptosis. |
| Recombinant IL-1β | Positive control for ELISA. | Generating a standard curve for IL-1β quantification. |
| Lipofectamine 2000 | Transfection reagent for cytosolic LPS delivery. | Activating the non-canonical Caspase-4/5/11 pathway. |
| LDH Assay Kit | Quantifies lactate dehydrogenase released from dead cells. | Measuring pyroptosis-associated membrane rupture. |
Frequently Asked Questions (FAQs)
Q1: I've detected activated Caspase-3 in my cells. Does this definitively confirm apoptosis is occurring?
A: Not necessarily. While Caspase-3 is the primary executioner caspase in apoptosis, recent research has revealed its role in other cell death pathways. Specifically, Caspase-3 can cleave Gasdermin E (GSDME), converting an apoptotic signal into pyroptosis [9] [10]. Furthermore, studies in microglial models of multiple sclerosis show that Caspase-3 activation can coexist with and promote GSDMD-mediated pyroptosis [11]. Therefore, confirmatory tests for pyroptotic markers (e.g., GSDMD/GSDME cleavage, IL-1β release) are essential when Caspase-3 is detected, especially in inflammatory contexts.
Q2: My cells are showing membrane blebbing. Is this specific to apoptosis?
A: Membrane blebbing is a classic feature of apoptosis but is not exclusive to it. It can also occur during pyroptosis [12] [10]. To distinguish between them, you must investigate further:
- Apoptotic blebbing leads to the formation of apoptotic bodies containing cellular debris, which are neatly packaged for silent phagocytosis [12] [13].
- Pyroptotic blebbing is associated with the formation of "pyroptotic bodies" and is driven by Gasdermin D (GSDMD) pore formation in the plasma membrane, which ultimately leads to lytic cell rupture [10] [11]. Assessing membrane integrity with dyes like propidium iodide (which enters pyroptotic cells late after membrane rupture) and checking for GSDMD cleavage is crucial [12].
Q3: How can I experimentally rule out apoptosis when studying a new form of inflammatory cell death?
A: A combination of positive and negative markers is required. The table below outlines a recommended experimental approach [12]:
| Assay Type | Target/Action | What to Measure | Expected Result to Rule Out Apoptosis |
|---|---|---|---|
| Viability & Cytolysis | Lactate Dehydrogenase (LDH) Release | Membrane integrity via extracellular LDH activity [12]. | Significant increase, indicating lytic death. |
| Caspase Activity | Caspase-1 or Caspase-4/5/11 | Activity using specific substrates or cleavage by Western blot [12] [10]. | Activation present. |
| Caspase Activity | Caspase-8 or Caspase-9 (initiators) | Activity or cleavage [5] [14]. | No activation. |
| Key Effector | Gasdermin D (GSDMD) | Cleavage (to GSDMD-NT) via Western blot [10] [11]. | Cleavage present. |
| Morphology | Live-cell Imaging | Real-time observation of cell swelling and membrane rupture [15] [11]. | Lytic morphology observed. |
| Pharmacological Inhibition | Pan-caspase inhibitor (e.g., Z-VAD-FMK) | Cell death and lysis [9] [11]. | Cell death may be partially inhibited, but lytic component persists if pyroptosis is active. |
| Genetic Inhibition | GSDMD Knockdown (siRNA) | Cell death and lysis [11]. | Significant reduction in lytic death. |
Q4: What are the key morphological differences I should look for under microscopy?
A: The table below summarizes the core morphological hallmarks to distinguish these death pathways [12] [10] [13]:
| Feature | Apoptosis | Pyroptosis |
|---|---|---|
| Cell Swelling | No | Yes |
| Plasma Membrane | Intact until late stages (secondary necrosis) | Pore formation, leading to rupture |
| Membrane Blebbing | Yes, forming apoptotic bodies | Yes, forming pyroptotic bodies |
| Chromatin Condensation | Yes, nuclear fragmentation | Yes, but the nucleus often remains intact [10] |
| Inflammatory Response | No (immunologically silent) | Yes (release of IL-1β, IL-18, DAMPs) |
| Phagocytic Clearance | Efficient, by neighboring cells | Overwhelmed, due to cell lysis |
Troubleshooting Guides
Problem 1: Inconsistent Pyroptosis Induction
- Problem: Expected GSDMD cleavage and IL-1β release are not observed after stimulation with a known inflammasome activator.
- Solution: Follow this systematic workflow to identify the failure point.
Problem 2: Differentiating Caspase-3-Mediated Apoptosis from Pyroptosis
- Problem: You observe Caspase-3 activation and lytic cell death, and you need to determine if it's secondary necrosis from apoptosis or Caspase-3/GSDME-mediated pyroptosis.
- Solution: This decision tree outlines the critical experiments to resolve this common ambiguity.
The Scientist's Toolkit: Essential Research Reagents
This table lists key reagents used to detect and differentiate apoptotic and pyroptotic cell death, as referenced in the scientific literature [12] [9] [14].
| Reagent Category | Specific Example | Function & Application |
|---|---|---|
| Caspase Activity Assays | Fluorogenic substrates (e.g., DEVD-afc for Caspase-3) | Quantify enzymatic activity of specific caspases in cell lysates using fluorescence or luminescence plate readers [14]. |
| Antibody-Based Detection | Anti-cleaved Caspase-3; Anti-cleaved GSDMD; Anti-phospho-MLKL (S358) | Detect specific cleavage products (indicating activation) by Western Blot, Flow Cytometry, or Immunofluorescence [12] [14] [11]. |
| Cell Viability & Cytolysis Assays | Lactate Dehydrogenase (LDH) Release Assay | Measure the release of the cytoplasmic enzyme LDH into the supernatant, a key indicator of lytic cell death like pyroptosis and necroptosis [12]. |
| Membrane Integrity Dyes | Propidium Iodide (PI), 7-AAD, DRAQ7 | Dyes that are impermeable to live cells but stain the DNA of cells with compromised plasma membranes, identifying late-stage lytic death [12]. |
| Early Apoptosis Markers | Annexin V conjugates | Binds to phosphatidylserine (PS) exposed on the outer leaflet of the plasma membrane in early apoptosis, often used in conjunction with PI [15]. |
| Pharmacological Inhibitors | Z-VAD-FMK (pan-caspase); VX-765 (Caspase-1); Nec-1s (RIPK1); Disulfiram (GSDMD) | Chemically inhibit key nodes of cell death pathways to establish mechanistic dependency. Note: Always validate specificity and use multiple inhibitors where possible [12] [9] [11]. |
| Genetic Tools | siRNA/shRNA (e.g., targeting GSDMD, Caspase-1); OptoCDEs [15] | Knock down key proteins to confirm functional role. Optogenetic tools allow spatiotemporally precise induction of specific death pathways [15] [11]. |
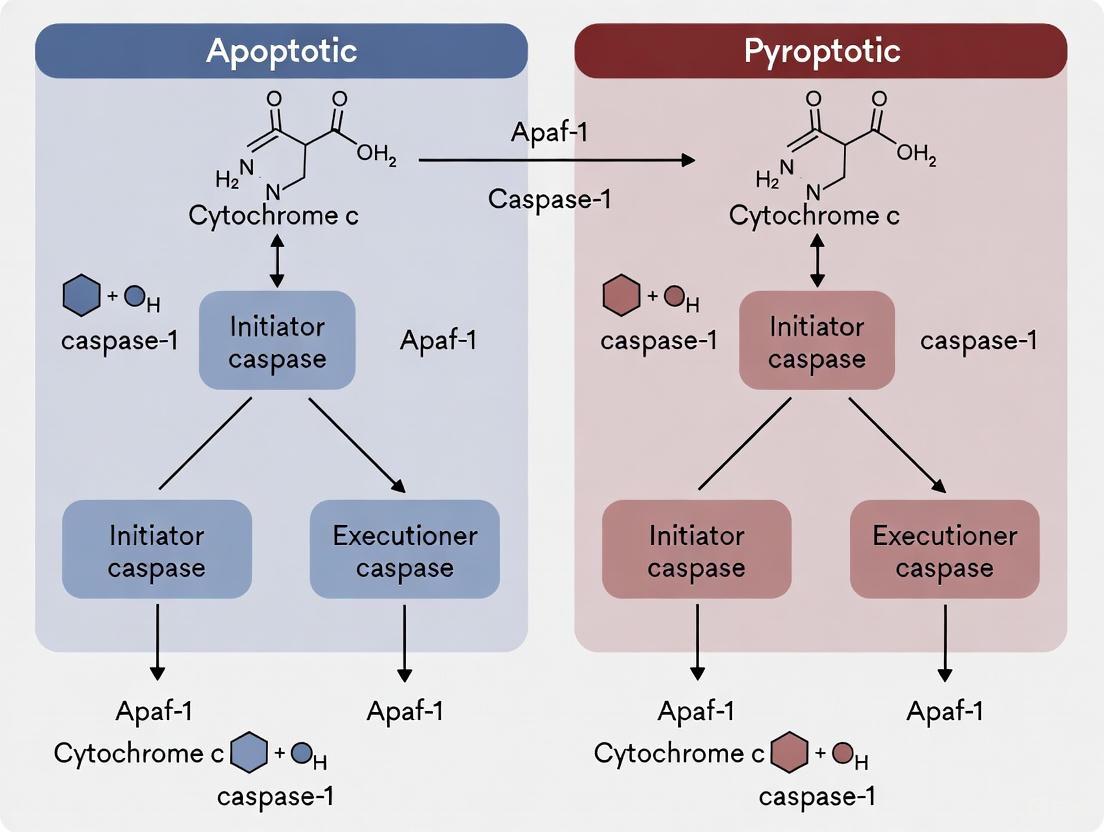
For decades, caspases were neatly categorized as either apoptotic (caspase-2, -3, -6, -7, -8, -9, -10) or inflammatory (caspase-1, -4, -5, -11) based on their primary roles in cell death and immunity [16]. This traditional classification has been fundamentally challenged by recent research revealing extensive crosstalk between these pathways and context-dependent caspase functions. Caspases are critical regulators of cell death, development, innate immunity, host defense, and disease [16]. They operate at the core of both non-lytic (apoptosis) and innate immune lytic (pyroptosis and PANoptosis) pathways [16], and their functional relationships are more interconnected than previously recognized.
The emerging paradigm recognizes that caspase functions are not confined to singular pathways but operate within complex regulatory networks. Apoptotic caspases can drive lytic inflammatory cell death, while inflammatory caspases can participate in apoptotic-like processes [16]. This technical support document provides troubleshooting guidance and FAQs to help researchers navigate the experimental challenges of studying these complex caspase interactions within the broader thesis of distinguishing apoptotic versus pyroptotic caspase activation.
Caspase Crosstalk Mechanisms: Key Signaling Pathways
Molecular Switches Between Apoptosis and Pyroptosis
How does caspase-3 function as a switch between apoptosis and pyroptosis?
Caspase-3, traditionally considered the main executioner caspase in apoptosis, plays a pivotal role in determining cell death modality through its interaction with gasdermin proteins. The key mechanism involves GSDME/DFNA5 cleavage, which can convert apoptotic signals into pyroptotic outcomes [17] [18].
- Mechanism: Activated caspase-3 cleaves GSDME, generating an N-terminal fragment that oligomerizes and forms pores in the plasma membrane, leading to pyroptosis [17] [18].
- Cellular Context: The cell death outcome depends on GSDME expression levels. Cells with high GSDME expression directly undergo pyroptosis, while those with low GSDME expression undergo secondary necrosis or apoptosis followed by pyroptosis [17]. Cells without GSDME expression exclusively undergo apoptosis [17].
- Therapeutic Relevance: This switching mechanism is particularly important in chemotherapy, where chemotherapeutic drugs activate caspase-3, which can then trigger either apoptosis or pyroptosis depending on GSDME status [17].
Table 1: Caspase-3 Substrates and Functional Consequences
| Caspase-3 Substrate | Cleavage Effect | Cell Death Pathway | Biological Context |
|---|---|---|---|
| PARP | Inactivates DNA repair | Apoptosis | Classical apoptotic substrate [1] |
| GSDME | Releases pore-forming N-terminal fragment | Pyroptosis | Switching mechanism from apoptosis to pyroptosis [17] [18] |
| GSDMD | Inactivates protein; blocks pyroptosis | Apoptosis (blocks pyroptosis) | Bidirectional crosstalk mechanism [1] |
Bidirectional Crosstalk and Pathway Interference
Can apoptotic and pyroptotic pathways regulate each other?
Yes, research reveals extensive bidirectional crosstalk between apoptotic and pyroptotic pathways, creating complex regulatory networks:
- Caspase-1 to Apoptosis: In GSDMD knockout cells, caspase-1 activation can lead to apoptosis through activation of caspases-3 and -7, demonstrating that GSDMD is the only caspase-1 substrate that induces pyroptosis [1].
- Caspase-3/7 to Pyroptosis Blockade: During apoptosis, caspases-3 and -7 cleave GSDMD at a site distinct from inflammatory caspases, inactivating the protein and eliminating the cell's ability to undergo pyroptosis [1] [18].
- Caspase-8 Integrative Functions: Caspase-8 serves as a critical node, participating in apoptosis, necroptosis, and inflammasome regulation, and can cleave multiple gasdermin family members (GSDMD, GSDMC) [19] [18].
Diagram 1: GSDME-dependent cell fate decision via caspase-3
The PANoptosis Framework: Integrated Cell Death
Conceptual Framework and Molecular Complexes
PANoptosis represents an integrated inflammatory cell death pathway that combines features of pyroptosis, apoptosis, and necroptosis, driven by a central molecular complex called the PANoptosome [20] [18]. This framework explains why these death pathways cannot be studied in complete isolation.
- PANoptosome Complex: A multiprotein complex that can engage multiple modes of cell death simultaneously, containing molecular components from pyroptosis, apoptosis, and necroptosis pathways [18].
- Shared Components: Several PANoptosomes have been identified, including ZBP1-PANoptosome, AIM2-PANoptosome, RIPK1-PANoptosome, and NLRP12-PANoptosome [18].
- Disease Relevance: PANoptosis has been implicated in various diseases, including cancer, Alzheimer's disease, and inflammatory conditions [20] [18] [21].
Table 2: PANoptosome Complexes and Their Activators
| PANoptosome Type | Key Sensor/Component | Primary Activators | Caspases Involved |
|---|---|---|---|
| ZBP1-PANoptosome | ZBP1 | Influenza A virus (IAV), viral RNA | Caspase-1, -3, -6, -8 [18] |
| AIM2-PANoptosome | AIM2 | Cytosolic DNA, Francisella novicida | Caspase-1, -3, -8 [18] |
| RIPK1-PANoptosome | RIPK1 | TNF-α, cellular stress | Caspase-1, -3, -8 [18] |
| NLRP12-PANoptosome | NLRP12 | Salmonella, bacterial infection | Caspase-1, -3, -8 [18] |
Troubleshooting Guide: Experimental Challenges
Distinguishing Cell Death Modalities
FAQ: My experiments show mixed cell death morphology. How can I distinguish apoptotic vs. pyroptotic caspase activation?
Mixed morphology often indicates concurrent activation of multiple death pathways. Implement the following experimental approaches:
- Multi-Parameter Assessment: Combine multiple detection methods rather than relying on a single assay (see Table 3).
- Genetic Knockouts: Use GSDMD and GSDME knockout cells to isolate specific pathways. In GSDMD-deficient cells, caspase-1 activation leads to apoptosis rather than pyroptosis [1].
- Caspase Activity Profiling: Measure specific caspase activities with substrate-based assays while inhibiting other caspases.
- Morphological Analysis: Use real-time imaging to track membrane blebbing (apoptosis) versus cell swelling and pore formation (pyroptosis).
Table 3: Key Characteristics for Distinguishing Cell Death Types
| Feature | Apoptosis | Pyroptosis | Necroptosis |
|---|---|---|---|
| Key Caspases | Caspase-3, -8, -9 | Caspase-1, -4, -5, -11 | RIPK1/RIPK3/MLKL |
| Morphology | Cell shrinkage, membrane blebbing | Cell swelling, pore formation | Organelle swelling, membrane rupture |
| Membrane Integrity | Maintained until late stages | Pore formation, membrane rupture | Loss of membrane integrity |
| Inflammation | Minimal | Strongly inflammatory | Inflammatory |
| Key Markers | PARP cleavage, phosphatidylserine exposure | GSDMD cleavage, IL-1β release | MLKL phosphorylation |
Specific Experimental Protocols
FAQ: What is a robust experimental approach to study caspase crosstalk in my cellular model?
The following protocol systematically assesses caspase crosstalk:
Protocol: Assessing Caspase-Mediated Death Switching
Baseline Characterization:
Genetic Manipulation:
- Generate or obtain GSDMD and GSDME knockout lines using CRISPR/Cas9 technology [1].
- Use siRNA for transient knockdown of specific caspases or gasdermins.
Stimulation and Inhibition:
Multi-Parametric Readouts:
- Cell Viability: Measure LDH release for membrane integrity [21].
- Caspase Activity: Use fluorogenic substrate assays for caspases-1, -3, -8.
- Gasdermin Cleavage: Detect cleavage by Western blot (GSDMD: ~31kD N-terminal; GSDME: ~35kD N-terminal).
- Morphology: Use real-time imaging with propidium iodide and membrane dyes.
Diagram 2: Experimental workflow for caspase crosstalk analysis
Research Reagent Solutions
Table 4: Essential Reagents for Caspase Crosstalk Research
| Reagent Category | Specific Examples | Function/Application | Considerations |
|---|---|---|---|
| Caspase Inhibitors | Z-VAD-FMK (pan-caspase), VX-765 (caspase-1), Z-DEVD-FMK (caspase-3) | Pathway blockade to determine specific caspase contributions | Test multiple concentrations; check specificity [1] [16] |
| Gasdermin Inhibitors | Disulfiram, Necrosulfonamide | Inhibit GSDMD pore formation | Can have off-target effects; include controls [19] |
| Genetic Tools | CRISPR/Cas9 for GSDMD, GSDME knockout; siRNA for transient knockdown | Define essentiality of specific components | Validate knockout/knockdown efficiency [1] |
| Activity Assays | Fluorogenic substrates (e.g., WEHD-afc for caspase-1, DEVD-afc for caspase-3) | Quantitative caspase activity measurement | Use specific buffer conditions for different caspases |
| Antibodies | Anti-cleaved GSDMD, Anti-cleaved GSDME, Anti-cleaved caspase-3 | Detect active forms in Western blot, IF | Validate specificity with knockout controls |
| Cell Death Inducers | Chemotherapeutic agents (doxorubicin, cisplatin), DPP8/9 inhibitors (Val-boroPro) | Activate specific death pathways | Titrate for optimal response in your system [17] [1] |
| Demethylating Agents | Decitabine (DAC) | Reverse GSDME silencing in cancer cells | Pre-treatment often required (24-48h) [17] |
The traditional dichotomy between apoptotic and inflammatory caspases has been superseded by a more nuanced understanding of caspase networks featuring extensive crosstalk and context-dependent functions. Successfully distinguishing apoptotic versus pyroptotic caspase activation requires:
- Multi-modal assessment rather than single-parameter readouts
- Genetic manipulation of key switching molecules (GSDMD, GSDME)
- Temporal analysis of caspase activation and substrate cleavage
- Consideration of cellular context, including expression levels of gasdermins
This technical guidance provides established methodologies and troubleshooting approaches to navigate the experimental challenges in this rapidly evolving field. As research progresses, the continued development of more specific tools and techniques will further illuminate the complex interplay between caspase functions and their roles in health and disease.
From Theory to Bench: Analytical Techniques for Detecting and Quantifying Caspase Activation
Within cell death research, accurately distinguishing between the intricate pathways of apoptosis and pyroptosis is fundamental. A key experimental strategy involves profiling the specific cleavage and activation of caspases, the proteases that act as central conductors of these processes. This guide provides detailed Western blot methodologies and troubleshooting advice to help researchers confidently differentiate between initiator and executioner caspase activation, thereby clarifying the mode of cell death in their experimental systems.
Core Concepts: Caspases in Cell Death Pathways
Caspases are a family of cysteine-aspartate proteases that play central roles in programmed cell death. They are typically synthesized as inactive zymogens (pro-caspases) and require proteolytic cleavage for activation [22]. Based on their function and position in the signaling cascade, they are categorized as follows:
- Initiator Caspases (Caspase-8, -9, -10): These are the first to be activated in response to pro-death signals. They propagate the death signal by cleaving and activating downstream executioner caspases [23] [24].
- Executioner Caspases (Caspase-3, -6, -7): Once activated by initiator caspases, they carry out the dismantling of the cell by cleaving a broad range of structural and functional cellular substrates [23] [25].
- Inflammatory Caspases (Caspase-1, -4, -5, -11): These are primarily involved in the activation of pyroptosis, a lytic and inflammatory form of cell death. They cleave substrates like gasdermin D (GSDMD) and pro-inflammatory cytokines [23] [1].
The table below summarizes the key morphological and molecular differences between apoptosis and pyroptosis.
Table 1: Key Characteristics of Apoptosis and Pyroptosis
| Feature | Apoptosis | Pyroptosis |
|---|---|---|
| Morphology | Cell shrinkage, membrane blebbing, formation of apoptotic bodies [23] | Cell swelling, plasma membrane pore formation, eventual lysis [23] [1] |
| Inflammation | Immunologically silent or anti-inflammatory [24] | Highly pro-inflammatory [23] [24] |
| Key Initiators | Caspase-8, -9 (initiator); Caspase-3, -7 (executioner) [23] [22] | Caspase-1, -4, -5 (inflammatory caspases) [23] [1] |
| Key Effectors | Cleavage of hundreds of cellular proteins (e.g., PARP, cytokeratin-18) [25] [1] | Cleavage of Gasdermin D (GSDMD) to form plasma membrane pores; processing of IL-1β and IL-18 [23] [1] |
| Pathway Crosstalk | Caspase-3/-7 can cleave and inactivate GSDMD, thereby suppressing pyroptosis [1] | In the absence of GSDMD, Caspase-1 can activate Caspase-3/-7, leading to apoptosis [1] |
Figure 1: Caspase Activation Pathways in Apoptosis and Pyroptosis. The diagram illustrates the distinct initiator mechanisms of extrinsic, intrinsic, and pyroptotic pathways, and highlights the crosstalk between them, particularly the mutual inhibition between executioner caspases and Gasdermin D.
Essential Protocols & Workflows
Standard Western Blot Protocol for Caspase Activation
Detecting caspase cleavage by Western blot is a conventional method to demonstrate the induction of apoptosis and differentiate it from other pathways [22].
Protocol Steps:
Protein Extraction & Quantification:
- Lyse cells in a suitable RIPA buffer supplemented with protease and phosphatase inhibitors.
- Centrifuge lysates to remove insoluble debris.
- Quantify protein concentration using a standardized method (e.g., BCA assay).
Gel Electrophoresis & Transfer:
- Cast and run an SDS-PAGE gel appropriate for resolving proteins in the 10-50 kDa range (cleaved caspase fragments) and higher (pro-caspases and loading controls).
- A 4-20% gradient gel is often ideal. Load 20-50 µg of total protein per well.
- Transfer proteins from the gel to a nitrocellulose or PVDF membrane.
Immunoblotting:
- Block the membrane with 5% non-fat milk or BSA in TBST for 1 hour.
- Incubate with primary antibodies against your target caspases and loading controls overnight at 4°C.
- Wash the membrane and incubate with an appropriate HRP-conjugated secondary antibody.
- Detect using a chemiluminescent substrate and image the blot.
Key Analysis Points:
- Pro-caspase Band: The higher molecular weight band represents the inactive zymogen. A decrease in its intensity often indicates activation.
- Cleaved Fragments: The appearance of lower molecular weight bands (e.g., p17/p19 for caspase-3, p18 for caspase-7, p35/p37 for caspase-9) confirms activation [22] [25].
- Additional Apoptosis Markers: Probing for cleaved substrates like PARP (89 kDa fragment) serves as a robust secondary confirmation of executioner caspase activity [25] [1].
Strategic Multilateral Blotting to Discriminate Cell Death
To conclusively distinguish between apoptosis and pyroptosis, a multi-target Western blot approach is recommended. The workflow below outlines this strategy.
Figure 2: Experimental Workflow for Discriminating Cell Death Pathways. This strategic multilateral blotting approach allows for the simultaneous assessment of key markers from multiple cell death pathways, enabling clear interpretation and identification of potential crosstalk.
The Scientist's Toolkit: Key Reagents
Table 2: Essential Research Reagents for Caspase and Cell Death Analysis
| Reagent / Assay | Specific Function | Key Application Notes |
|---|---|---|
| Anti-Cleaved Caspase-3 (p17/p19) | Detects active executioner caspase-3 fragments [25] | Primary confirmation of executioner caspase activity in apoptosis. |
| Anti-Cleaved Caspase-7 (p18) | Detects active executioner caspase-7 fragments [25] | Co-detection with cleaved caspase-3 confirms robust executioner activation. |
| Anti-Cleaved Caspase-8 | Detects active initiator caspase-8 fragments [22] | Marker for the extrinsic apoptosis pathway. |
| Anti-Cleaved Caspase-9 (p35/p37) | Detects active initiator caspase-9 fragments [22] [25] | Marker for the intrinsic (mitochondrial) apoptosis pathway. |
| Anti-Cleaved PARP (p89) | Detects caspase-3/7-cleaved PARP fragment [25] [1] | High-quality readout for executioner caspase activity; very stable signal. |
| Anti-Cleaved GSDMD (p30) | Detects active N-terminal fragment of Gasdermin D [1] | Definitive marker for pyroptosis. |
| Anti-Caspase-1 (p20) | Detacts cleaved, active inflammatory caspase-1 [1] | Marker for canonical pyroptosis pathway. |
| Caspase Inhibitor (z-VAD-fmk) | Pan-caspase inhibitor; blocks both apoptosis and pyroptosis [25] | Control to confirm caspase-dependent cell death. |
| Caspase-3/7 Inhibitor (DEVD-fmk) | Selective inhibitor of executioner caspases [25] | Tool to dissect the specific role of executioner caspases. |
| Caspase-Glo 3/7 Assay | Luminescent assay to measure caspase-3/7 activity [25] | Provides quantitative, functional data to complement Western blot results. |
Troubleshooting FAQs
FAQ 1: I see strong cleaved caspase-3 signal in my blot, but my cell viability assay shows no significant cell death. What could explain this discrepancy?
- Answer: This is a recognized phenomenon, especially during bacterial infections or in specific cellular contexts. Research has shown that host cells can exhibit significant caspase-3/7 activity (measured by DEVDase activity) without immediately undergoing lytic cell death, as indicated by assays measuring membrane integrity (LDH release) or metabolic activity (MTS) [25].
- Solution:
- Probe additional markers: Check for cleaved PARP to confirm downstream apoptotic signaling.
- Assess morphology: Use microscopy to look for classic apoptotic morphology (membrane blebbing, chromatin condensation) which may occur without immediate lysis.
- Consider non-canonical roles: Executioner caspases can have functions beyond inducing rapid cell lysis. For example, they can directly cleave bacterial virulence factors to inhibit intracellular bacterial growth, a process that may not immediately kill the host cell [25].
FAQ 2: How can I tell if cell death in my model is purely apoptotic or if there is pyroptotic crosstalk?
- Answer: The pathways are not mutually exclusive. A single stimulus can trigger components of both, and there is documented bidirectional crosstalk [1].
- Solution: Implement the multilateral blotting strategy from Figure 2.
- If GSDMD is absent, caspase-1 can cleave and activate caspase-3, leading to apoptosis. In this case, you would detect cleaved caspase-1, cleaved caspase-3, and cleaved PARP, but not cleaved GSDMD [1].
- Conversely, during apoptosis, activated caspase-3 and -7 can cleave GSDMD at a distinct site, which inactivates it and prevents pyroptosis. Here, you would see cleaved caspase-3 and cleaved GSDMD (likely a different fragment on a Western blot), but the cell would still die via apoptosis [1].
FAQ 3: My Western blot for cleaved caspase-3 shows high background or non-specific bands. How can I improve specificity?
- Answer: This is a common issue that can often be resolved by optimizing antibody incubation conditions.
- Solution:
- Titrate your antibody: Use the lowest concentration that gives a clear specific signal.
- Adjust blocking conditions: Switch from milk to BSA (or vice versa) as the blocking agent, as this can reduce non-specific binding in some cases.
- Increase wash stringency: Add a mild detergent like 0.1% SDS to your TBST wash buffer or increase the number of washes.
- Verify the result: Use a positive control (e.g., lysate from cells treated with Staurosporine) to confirm the identity of the correct band. Knockout cell lysates, if available, are excellent negative controls.
Fluorescent-Based Assays for Real-Time Caspase Activity Monitoring
For researchers investigating programmed cell death, distinguishing between apoptosis and pyroptosis is crucial, as these pathways have profoundly different impacts on tissue health, immune responses, and disease outcomes. While both processes involve caspase family proteases, the specific caspases activated and their subsequent functions create a clear divergence. Apoptosis is a non-inflammatory, programmed cell death essential for development and homeostasis, primarily mediated by initiator caspases (e.g., caspase-8, -9) and executor caspases (e.g., caspase-3, -7) that dismantle the cell neatly for phagocytosis [26] [27]. In contrast, pyroptosis is a lytic, highly inflammatory form of cell death triggered in response to pathogens or danger signals. It is primarily mediated by inflammatory caspases (caspase-1, -4, -5 in humans) that cleave gasdermin proteins (e.g., GSDMD), forming pores in the plasma membrane and leading to the release of pro-inflammatory cytokines like IL-1β and IL-18 [26] [28].
Fluorescent-based real-time caspase activity assays provide a powerful tool to differentiate these pathways in live cells. By using probes specific for different caspase classes, you can determine the cell death mechanism as it unfolds, providing invaluable insights for drug discovery and basic research.
The Scientist's Toolkit: Key Reagents and Assays
The following table summarizes the core reagent solutions used in real-time caspase activity monitoring.
| Reagent/Assay Name | Caspase Target | Mechanism of Action | Primary Application |
|---|---|---|---|
| CellEvent Caspase-3/7 [29] | Caspase-3 & -7 | Cell-permeant, fluorogenic substrate (DEVD peptide) bound to nucleic acid dye; cleavage yields bright nuclear fluorescence. | Real-time, no-wash imaging in live cells. |
| Image-iT LIVE Kits [29] | Caspase-3/7 or Poly-caspases | Fluorescently-labeled caspase inhibitors (e.g., FAM-DEVD-FMK) covalently bind active enzymes. | End-point, fixable assay for microscopy/HCS. |
| Caspase-Glo 3/7 Assay [30] | Caspase-3 & -7 | Lytic, luminogenic DEVD substrate; cleavage generates luminescent signal. | High-throughput, end-point screening. |
| Fluorescent Activity-Based Probes (ABPs) [31] [32] | Broad or specific (e.g., Caspase-3) | Irreversibly bind active caspases with a fluorophore; allows biochemical analysis via SDS-PAGE. | In vitro profiling & in vivo imaging; target identification. |
| FRET-Based Probe (CFP-LEVD-YFP) [33] | Primarily Caspase-6 & -8 | Caspase cleavage separates CFP and YFP, eliminating FRET signal. | Real-time monitoring of caspase activity in live, transfected cells via flow cytometry. |
| ApoAlert Caspase Assay Kits [34] | Various (e.g., Caspase-3, -9/6) | Fluorometric or colorimetric detection of cleaved substrates (e.g., DEVD-AFC, LEHD-AMC). | End-point, plate-reader based quantification from cell lysates. |
Frequently Asked Questions (FAQs)
Q1: My real-time caspase-3/7 assay shows no signal, even though cell death is evident. What could be wrong?
This is a common issue and often relates to assay timing. Caspase-3/7 activity is transient. If you measure too late in the cell death process, the cells may have progressed to secondary necrosis, and the caspases are no longer active [30]. To troubleshoot:
- Perform a time-course experiment: Use a kinetic cytotoxicity assay (e.g., CellTox Green) to monitor the onset of cell death. The peak caspase activity typically coincides with the initial increase in cytotoxicity signal [30].
- Confirm the death mechanism: The compound you are using might induce primary necrosis (e.g., with digitonin) or pyroptosis, which may not involve significant caspase-3/7 activation. Primary necrosis shows high cytotoxicity without caspase-3/7 signal, while pyroptosis may involve caspase-1 instead [30] [26].
- Check inhibitor specificity: If studying pyroptosis, note that caspase-3/7 inhibitors will not block this pathway, which relies on inflammatory caspases [26].
Q2: How can I specifically distinguish between apoptotic and pyroptotic caspase activation in my model?
The key is to use assays that differentiate between executioner caspases (apoptosis) and inflammatory caspases (pyroptosis).
- Use Target-Specific Probes:
- Combine with Pyroptosis Markers: Multiplex your caspase assay with a probe for Gasdermin D (GSDMD) pore formation or measure the release of IL-1β, which is a hallmark of pyroptosis [26] [28].
- Employ Activity-Based Probes (ABPs): These covalent probes allow you to biochemically identify the specific active caspases present in your sample via fluorescent SDS-PAGE, providing unambiguous identification [31].
Q3: I am seeing high background fluorescence in my no-wash, live-cell assay. How can I reduce it?
High background can obscure specific signal.
- Optimize Staining Concentration and Time: Titrate your probe to find the lowest concentration that gives a robust signal upon induction. Incubate for the recommended time (typically 30-60 minutes); over-incubation can increase non-specific background [29].
- Verify Cell Permeability and Health: The probe is designed to be non-fluorescent until cleaved and bound to DNA. Excessive background can sometimes occur in cells with compromised membranes. Ensure your control cells are healthy [29].
- Include a Caspase Inhibitor Control: Always run a parallel sample pre-treated with a specific caspase-3/7 inhibitor (e.g., Z-DEVD-FMK). A reduction in fluorescence confirms the signal is specific to caspase activity [29].
Q4: Can I multiplex a caspase activity assay with other cell health indicators?
Yes, multiplexing is highly recommended for an accurate interpretation of cell death.
- Viability and Cytotoxicity: The CellEvent Caspase-3/7 assay can be combined with viability stains (e.g., TMRM for mitochondrial membrane potential) and cytotoxicity dyes (e.g., CellTox Green for membrane integrity) [29] [30].
- Immunofluorescence: A major advantage of some probes (e.g., CellEvent) is that the signal survives formaldehyde fixation, allowing you to subsequently stain for other intracellular targets, such as cleaved Gasdermin D or phospho-proteins [29].
- Annexin V Caution: Be aware that phosphatidylserine (PS) exposure is a feature of both apoptosis and pyroptosis. Therefore, Annexin V staining alone cannot distinguish between them [28].
Experimental Protocols
Protocol 1: Real-Time, No-Wash Imaging of Caspase-3/7 Activity in Live Cells
This protocol uses reagents like CellEvent Caspase-3/7 Green to monitor apoptosis kinetics in live cells, ideal for distinguishing the rapid dynamics of apoptotic versus pyroptotic death [29].
Workflow Overview:
Detailed Steps:
- Cell Preparation: Seed cells into an appropriate imaging chamber (e.g., 96-well plate). Apply your apoptotic or pyroptotic stimulus. Include a control well pre-treated with a caspase-3/7 inhibitor (e.g., 20 µM Z-DEVD-FMK) for 1 hour before stimulus to confirm signal specificity [29].
- Staining Solution: Prepare a working solution of the CellEvent reagent (e.g., 2-5 µM) in pre-warmed culture medium or PBS.
- Staining: At the desired time point post-stimulation, remove the existing medium and replace it with the staining solution.
- Incubation: Incubate the cells for 30-60 minutes at 37°C, protected from light. Do not wash the cells, as this can dislodge fragile apoptotic cells.
- Imaging: Visualize the cells immediately using a fluorescence microscope with a standard FITC filter set. Apoptotic cells will display bright green nuclear fluorescence.
Protocol 2: Profiling Active Caspases Using Fluorescent Activity-Based Probes (ABPs)
This protocol describes how to use ABPs like AB50 or LE22 to directly label and identify specific active caspases in cell lysates, which is powerful for confirming which caspases are engaged in your death pathway [31].
Workflow Overview:
Detailed Steps:
- Prepare Cell Lysate:
- Induce apoptosis/pyroptosis in cells (e.g., in a 6-well plate).
- Harvest cells by scraping and pellet by centrifugation.
- Wash cell pellet with cold PBS.
- Lyse cells in a hypotonic lysis buffer (e.g., 50 mM PIPES, pH 7.4, 10 mM KCl, 5 mM MgCl₂, 1% NP-40) supplemented with fresh DTT [31].
- Clarify the lysate by centrifugation and determine protein concentration using a BCA assay.
Labeling Reaction:
- Incubate a sample of the lysate (e.g., 50 µg of protein) with the fluorescent ABP (e.g., 1 µM final concentration of LE22 or AB50) for 30-60 minutes at 37°C [31].
- Stop the reaction by adding 4X Laemmli sample buffer and heating at 95°C for 5 minutes.
Detection:
- Separate the proteins by SDS-PAGE on a 15% gel.
- Scan the gel directly using a flatbed laser scanner (e.g., Typhoon scanner) with the appropriate fluorescence channel (e.g., Cy5 for AB50/LE22). The labeled caspases will appear as distinct bands. For example, AB50 labels active caspase-3 and -7, while LE22 labels caspase-3, -6, and -7 [31].
Troubleshooting Guide
The table below outlines common problems, their potential causes, and recommended solutions.
| Problem | Potential Causes | Suggested Solutions |
|---|---|---|
| Weak or No Signal | 1. Incorrect timing.2. Wrong caspase target.3. Over-fixed cells (for fixed assays).4. Probe degradation. | 1. Perform a kinetic time-course with a cytotoxicity dye [30].2. Use a poly-caspase probe (e.g., FAM-VAD-FMK) or ABPs to identify active caspases [29] [31].3. Optimize fixation protocol; some probes are fixable [29].4. Prepare fresh probe solution. |
| Excessive Background Fluorescence | 1. Probe concentration too high.2. Incubation time too long.3. High cell death causing non-specific binding. | 1. Titrate the probe to optimal concentration [29].2. Reduce incubation time.3. Include a healthy cell control; use inhibitor to confirm specificity [29]. |
| Signal in Untreated Controls | 1. Basal caspase activity.2. Spontaneous cell death due to poor culture conditions. | 1. This can be normal; quantify and establish a baseline. Use the inhibitor control.2. Ensure cells are healthy, not over-confluent, and use low-passage numbers. |
| Inconsistent Results Between Replicates | 1. Uneven cell seeding or treatment.2. Edge effects in microplates.3. Inconsistent assay reagent addition. | 1. Ensure uniform cell seeding and compound dispensing.2. Use edge-well controls or plate seals to prevent evaporation.3. Use automated dispensers for reagent addition. |
Gasdermin Cleavage as a Definitive Marker for Pyroptosis Detection
Pyroptosis is a lytic and pro-inflammatory type of programmed cell death that plays crucial roles in host defense and inflammatory diseases [35] [36]. Unlike apoptosis, which is generally non-inflammatory, pyroptosis is characterized by cell swelling, plasma membrane rupture, and release of pro-inflammatory cytokines and cellular contents [35] [36]. The discovery of Gasdermin D (GSDMD) as the key executor of pyroptosis has provided a definitive molecular marker for detecting this form of cell death [37] [38].
Gasdermin D belongs to the gasdermin protein family, which includes six members in humans (GSDMA, GSDMB, GSDMC, GSDMD, GSDME, and DFNB59) [39] [38]. Under normal conditions, full-length GSDMD remains inactive in the cytoplasm through autoinhibition, where the C-terminal domain (GSDMD-CTD) folds back and inhibits the N-terminal domain (GSDMD-NTD) [38] [40]. When cells receive specific inflammatory signals, inflammatory caspases cleave GSDMD, releasing this autoinhibition and triggering pyroptosis [37] [38].
Molecular Mechanisms of Gasdermin D Activation
Canonical Inflammasome Pathway
In the canonical pathway, inflammasome sensors (such as NLRP3, NLRC4, or AIM2) detect pathogen-associated molecular patterns (PAMPs) or damage-associated molecular patterns (DAMPs) [37] [38]. These sensors recruit the adapter protein ASC, which in turn recruits and activates caspase-1 [35] [38]. Activated caspase-1 then cleaves GSDMD at Asp275 (in humans) or Asp276 (in mice), separating the N-terminal domain from the C-terminal domain [37] [38]. The cleaved GSDMD-N-terminal fragment (GSDMD-NT) oligomerizes and forms pores in the plasma membrane, leading to pyroptosis [37] [38] [40]. Caspase-1 also processes pro-IL-1β and pro-IL-18 into their mature forms, which are released through GSDMD pores [35].
Non-Canonical Inflammasome Pathway
The non-canonical pathway is triggered by intracellular lipopolysaccharide (LPS) from Gram-negative bacteria [35] [38]. In mice, caspase-11 directly binds to cytosolic LPS, becomes activated, and cleaves GSDMD [35] [38]. In humans, caspase-4 and caspase-5 serve this function [35] [38]. The cleavage of GSDMD by these inflammatory caspases similarly results in GSDMD-NT pore formation and pyroptosis [35].
Alternative Activation Pathways
Beyond the canonical and non-canonical pathways, other enzymes can cleave and activate GSDMD under specific conditions. Caspase-8, typically associated with apoptosis, can cleave GSDMD at D275 during extrinsic apoptosis triggered by TNF or death receptor signaling [39]. Additionally, granzymes from cytotoxic T cells and natural killer cells, as well as certain bacterial proteases, can cleave GSDMD [39].
Figure 1: Gasdermin D Activation Pathways in Pyroptosis. This diagram illustrates the canonical and non-canonical pathways leading to GSDMD cleavage and pyroptosis.
Experimental Detection of Gasdermin D Cleavage
Western Blot Analysis
Western blotting is the most common method for detecting GSDMD cleavage. Researchers can monitor the appearance of the cleaved N-terminal fragment (approximately 31 kDa) and/or the disappearance of full-length GSDMD (approximately 53 kDa).
Protocol:
- Cell Lysis: Lyse cells in RIPA buffer supplemented with protease and phosphatase inhibitors.
- Protein Quantification: Determine protein concentration using BCA or Bradford assay.
- Gel Electrophoresis: Separate 20-50 μg of protein on 4-20% gradient SDS-PAGE gels.
- Membrane Transfer: Transfer proteins to PVDF or nitrocellulose membranes.
- Blocking: Block membranes with 5% non-fat milk or BSA in TBST for 1 hour.
- Antibody Incubation:
- Incubate with primary antibodies against GSDMD (specific for full-length and/or N-terminal fragment) overnight at 4°C.
- Wash membranes 3 times with TBST.
- Incubate with HRP-conjugated secondary antibodies for 1 hour at room temperature.
- Detection: Develop blots using enhanced chemiluminescence (ECL) substrate and visualize with a chemiluminescence imager.
Troubleshooting Tips:
- If bands are weak, try different antibody dilutions or increase protein loading.
- For better separation of full-length and cleaved fragments, use longer electrophoresis times.
- Include both positive and negative controls in each experiment.
Immunofluorescence and Microscopy
Immunofluorescence can visualize GSDMD cleavage and pore formation in fixed cells.
Protocol:
- Cell Culture: Plate cells on glass coverslips and apply treatments.
- Fixation: Fix cells with 4% paraformaldehyde for 15 minutes at room temperature.
- Permeabilization: Permeabilize with 0.1% Triton X-100 for 10 minutes.
- Blocking: Block with 3% BSA in PBS for 1 hour.
- Antibody Staining:
- Incubate with anti-GSDMD-NT primary antibody diluted in blocking buffer overnight at 4°C.
- Wash 3 times with PBS.
- Incubate with fluorophore-conjugated secondary antibody for 1 hour at room temperature.
- Nuclear Staining: Stain with DAPI or Hoechst for 5 minutes.
- Mounting: Mount coverslips with antifade mounting medium.
- Imaging: Image using a fluorescence or confocal microscope.
Expected Results: In cells undergoing pyroptosis, GSDMD-NT localizes to the plasma membrane, forming distinct puncta or ring-like structures. Full-length GSDMD shows diffuse cytoplasmic staining in resting cells.
ELISA-Based Detection
Commercial ELISA kits are available for quantifying GSDMD cleavage fragments in cell culture supernatants and lysates.
Protocol (using mouse GSDMD ELISA kit as example):
- Sample Preparation: Collect cell culture supernatants (centrifuge to remove cells) and prepare cell lysates.
- Standard Preparation: Reconstitute and dilute standards according to kit instructions.
- Plate Setup: Add standards and samples to appropriate wells.
- Incubation: Incubate at room temperature for 2 hours with gentle shaking.
- Washing: Wash wells 4 times with wash buffer.
- Detection Antibody: Add detection antibody and incubate for 1-2 hours.
- Substrate Addition: Add substrate solution and incubate for 30 minutes.
- Stop Solution: Add stop solution and read absorbance immediately at 450 nm.
Performance Characteristics:
- Detection Range: 15.6 to 1000 pg/ml
- Sensitivity: 14 pg/ml
- Specificity: Detects C-terminal part of GSDMD as well as full-length protein [37]
Flow Cytometry
Flow cytometry can detect GSDMD cleavage and pore formation in single cells using specific antibodies.
Protocol:
- Cell Harvesting: Harvest cells and wash with PBS.
- Fixation and Permeabilization: Fix and permeabilize cells using commercial fixation/permeabilization buffers.
- Antibody Staining: Stain with anti-GSDMD-NT antibody followed by fluorophore-conjugated secondary antibody.
- Analysis: Analyze cells using flow cytometry, gating on the population of interest.
Troubleshooting Guides
Common Experimental Issues and Solutions
Table 1: Troubleshooting Guide for GSDMD Cleavage Detection
| Problem | Possible Causes | Solutions |
|---|---|---|
| Weak or no GSDMD cleavage signal | Insufficient inflammasome activation; Incorrect caspase activation; Low expression of GSDMD | Optimize stimulant concentration and duration; Verify caspase activation with specific inhibitors; Check GSDMD expression levels by qPCR or Western blot |
| High background in Western blot | Non-specific antibody binding; Incomplete blocking | Optimize antibody concentrations; Include no-primary antibody control; Use different blocking buffer (BSA instead of milk) |
| Unexpected band sizes | Non-specific cleavage; Proteasome degradation; Alternative splicing | Include positive control; Use protease inhibitor cocktail; Validate with multiple antibodies targeting different epitopes |
| No pore formation despite cleavage | Impaired GSDMD-NT oligomerization; Compensatory mechanisms | Check phospholipid binding conditions; Verify membrane composition; Assess other gasdermin family members |
| Inconsistent results between replicates | Variable cell density; Inconsistent treatment; Edge effects in culture plates | Standardize cell counting; Use master mixes for treatments; Avoid using edge wells in plate |
Controls for GSDMD Cleavage Experiments
Proper controls are essential for interpreting GSDMD cleavage experiments:
Negative Controls:
- Unstimulated cells
- Cells treated with caspase inhibitors (e.g., VX-765 for caspase-1)
- Cells with GSDMD knockdown/knockout
Positive Controls:
- Cells treated with nigericin (for NLRP3 activation)
- Cells transfected with LPS (for non-canonical pathway)
- Cells treated with known pyroptosis inducers specific to your system
Specificity Controls:
- Use multiple antibodies targeting different GSDMD epitopes
- Include molecular weight markers to verify fragment sizes
- Validate with genetic approaches (overexpression, knockdown)
Frequently Asked Questions
Q1: Can GSDMD cleavage occur without leading to pyroptosis? A: Yes, emerging evidence suggests that GSDMD can be cleaved without immediate cell lysis. Limited pore formation may allow for cytokine release without full pyroptosis, and cellular repair mechanisms can potentially reseal membranes after minor GSDMD pore formation [39] [40].
Q2: How specific is GSDMD cleavage for pyroptosis versus other cell death types? A: GSDMD cleavage is highly specific for pyroptosis when mediated by inflammatory caspases (caspase-1/4/5/11). However, certain apoptotic stimuli can cause GSDMD cleavage via caspase-8, and cytotoxic lymphocyte granzymes can also cleave GSDMD, blurring this distinction in specific contexts [39].
Q3: What is the typical timeframe for GSDMD cleavage after stimulation? A: The kinetics depend on the stimulus and cell type. For strong NLRP3 activators like nigericin, cleavage can be detected within 15-30 minutes, peaking at 1-2 hours. For non-canonical activators, cleavage typically occurs within 1-4 hours post-stimulation.
Q4: Why do I detect GSDMD cleavage but no IL-1β release? A: This could indicate limited pore formation that's insufficient for cytokine release, the presence of alternative IL-1β secretion pathways, or that GSDMD pores are selectively permeable and may not efficiently release all cytokines under certain conditions [39] [40].
Q5: Can other gasdermin family members compensate for GSDMD function? A: Yes, particularly GSDME can be cleaved by caspase-3 and execute pyroptosis when GSDMD is absent or non-functional. This represents an important compensatory mechanism in some experimental systems [39].
Q6: How does GSDMD pore formation lead to cell lysis? A: GSDMD pores disrupt ionic gradients, leading to water influx, cell swelling, and eventual membrane rupture. Recent research has identified NINJ1 as a protein that mediates plasma membrane rupture downstream of GSDMD pore formation [39] [40].
Research Reagent Solutions
Table 2: Essential Reagents for Studying GSDMD Cleavage and Pyroptosis
| Reagent Category | Specific Examples | Application Notes |
|---|---|---|
| GSDMD Antibodies | Anti-GSDMD (full-length), Anti-GSDMD-NT, Anti-GSDMD-CT | Validate antibodies for specific applications (WB, IF, FC); Different species may require specific validations |
| Caspase Inhibitors | VX-765 (caspase-1), Z-VAD-FMK (pan-caspase), Wedelolactone (caspase-11) | Use appropriate concentrations and pretreatment times; Verify specificity for intended caspases |
| Inflammasome Activators | Nigericin (NLRP3), Poly(dA:dT) (AIM2), Flagellin (NLRC4), LPS transfection (non-canonical) | Optimize concentrations for specific cell types; Include cell death controls |
| Genetic Tools | GSDMD siRNA/shRNA, GSDMD knockout cells, GSDMD overexpression plasmids | Validate knockdown/knockout efficiency; Use multiple constructs for confirmation |
| Detection Kits | GSDMD ELISA kits, LDH release assays, IL-1β/IL-18 ELISA | Choose species-compatible kits; Establish standard curves for quantification |
| Positive Controls | Recombinant cleaved GSDMD-NT, Pyroptosis-inducer compounds | Use as standards for assay validation and quantification |
Distinguishing Pyroptosis from Apoptosis
A key application of monitoring GSDMD cleavage is distinguishing pyroptosis from apoptosis in experimental systems. The table below summarizes the key differences:
Table 3: Differentiation Between Pyroptosis and Apoptosis
| Feature | Pyroptosis | Apoptosis |
|---|---|---|
| Morphology | Cell swelling, plasma membrane rupture, pore formation | Cell shrinkage, membrane blebbing, apoptotic bodies |
| Inflammation | Highly inflammatory | Generally non-inflammatory |
| Key Executors | GSDMD pore formation | Caspase-3/6/7, cytochrome c, apoptosome |
| Caspases Involved | Caspase-1/4/5/11 (inflammatory caspases) | Caspase-8/9/10 (initiators), Caspase-3/6/7 (effectors) |
| Biomarkers | GSDMD cleavage, IL-1β/IL-18 release | Caspase-3 cleavage, PARP cleavage, phosphatidylserine exposure |
| Membrane Integrity | Early disruption with pore formation | Maintained until late stages |
| Physiological Role | Host defense against pathogens, inflammatory diseases | Development, homeostasis, elimination of damaged cells |
Advanced Technical Considerations
Post-Translational Modifications of GSDMD
Beyond cleavage, GSDMD activity is regulated by various post-translational modifications:
- Ubiquitination: Regulates GSDMD stability and degradation
- Succination: Can inhibit GSDMD pore formation [40]
- ADP-riboxanation: May modulate GSDMD function in specific contexts [40]
- Phosphorylation: Potential regulatory mechanism under investigation
Non-Canonical Functions of GSDMD
Recent research has revealed that GSDMD has functions beyond pyroptosis execution:
- GSDMD pores can facilitate selective cytokine release without cell lysis [39] [40]
- GSDMD can associate with intracellular organelles including mitochondria, peroxisomes, and endosomes [39]
- GSDMD may play roles in cellular homeostasis independent of cell death [39]
Species-Specific Considerations
When studying GSDMD across species, important differences include:
- Mice have multiple GSDMA paralogs but lack GSDMB
- Caspase-11 in mice functions similarly to caspase-4/5 in humans
- Some ELISA and antibodies may not cross-react between species (e.g., the mouse GSDMD ELISA kit mentioned does not cross-react with human GSDMD) [37]
Gasdermin D cleavage serves as a definitive marker for pyroptosis detection when properly contextualized within experimental systems. The methodologies outlined in this technical guide provide researchers with comprehensive tools for detecting and quantifying GSDMD cleavage, troubleshooting common experimental issues, and distinguishing pyroptosis from other cell death modalities. As research in this field advances, monitoring GSDMD cleavage remains essential for understanding inflammatory cell death in host defense, inflammatory diseases, and cancer biology.
For researchers in drug development and immunology, accurately distinguishing between apoptosis and pyroptosis is crucial, as these pathways have profoundly different impacts on the immune response and therapeutic outcomes. Apoptosis is generally considered an immunologically "silent" process, whereas pyroptosis is highly inflammatory, acting as a "whistle blower" by releasing pro-inflammatory alarmins [24]. While both processes involve caspase activation, the specific caspases involved and the subsequent morphological changes differ. Multiparametric flow cytometry provides a powerful tool to dissect this complexity by simultaneously measuring caspase activation alongside key phenotypic markers of cell death in individual cells. This technical guide provides troubleshooting support and detailed methodologies to enhance the reliability of your experiments in this critical area of research.
Troubleshooting Guide & FAQs
This section addresses common experimental challenges encountered when configuring multiparametric flow cytometry assays for cell death.
FAQ 1: Why is my caspase signal weak or absent?
| Possible Cause | Recommendation |
|---|---|
| Insufficient Induction | Optimize treatment conditions (e.g., stimulus concentration, duration) to ensure measurable caspase activation. Include a positive control (e.g., camptothecin for apoptosis) for validation [41]. |
| Suboptimal Fluorochrome | For weakly expressed targets like early caspase activation, use a bright fluorochrome conjugate (e.g., PE). Save dimmer fluorochromes (e.g., FITC) for highly abundant targets [41]. |
| Instrument Settings | Verify that the laser wavelength and PMT settings on your cytometer match the excitation and emission wavelengths of your chosen caspase substrate (e.g., PhiPhiLux G1D2 for 488 nm laser) [41]. |
| Analysis Timing | If using PhiPhiLux substrates, analyze samples promptly. The fluorescent fragments diffuse out of the cell over several hours, leading to signal loss [42]. |
FAQ 2: How can I reduce high background or non-specific staining?
| Possible Cause | Recommendation |
|---|---|
| Fc Receptor Binding | Block cells with Bovine Serum Albumin, an Fc receptor blocking reagent, or normal serum from the host species of your antibodies prior to staining [41]. |
| Antibody Concentration | Titrate your antibodies. Using too much antibody is a common cause of high background [41]. |
| Dead Cells | Dead cells can non-specifically bind antibodies. Use a viability dye (e.g., PI, 7-AAD, or a fixable dye) to gate out dead cells during analysis [41]. |
| Cell Autofluorescence | For cell types with high autofluorescence (e.g., neutrophils), use fluorochromes that emit in red-shifted channels (e.g., APC) or very bright fluorochromes to overcome the background [41]. |
FAQ 3: My data shows high variability from day to day. What could be wrong?
| Possible Cause | Recommendation |
|---|---|
| Inconsistent Sample Prep | Adhere strictly to a standardized protocol for cell washing, fixation, and permeabilization. Inconsistent permeabilization, in particular, can greatly affect results [41]. |
| Unstable Reagents | Check the storage conditions and shelf life of reagents. For example, opened PhiPhiLux aliquots should be stored at -20°C and avoided repeated freeze-thaw cycles [42] [43]. |
| Instrument Performance | Ensure proper instrument settings are loaded and that the flow cell is not clogged. Run control samples to standardize instrument performance across days [44] [41]. |
Research Reagent Solutions
The table below lists key reagents for configuring a multiparametric assay to detect caspase activation and cell death.
| Reagent | Function in the Assay | Key Considerations |
|---|---|---|
| PhiPhiLux G1D2 | Fluorogenic substrate for caspases-3/7. Becomes fluorescent upon cleavage, marking early apoptosis [42] [43]. | Cell-permeable; analysis should be prompt as fragments diffuse out. Incompatible with fixation. Signal-to-noise ratio is ~40:1 [42]. |
| FLICA | Fluorogenic substrate that covalently binds active caspases, immobilizing the signal [42]. | Compatible with subsequent fixation and permeabilization steps, allowing for more flexible experimental timing [42]. |
| Annexin V (PE or APC) | Binds to phosphatidylserine (PS) exposed on the outer leaflet of the cell membrane, a marker for early/mid-stage apoptosis [42] [43]. | Requires calcium-containing buffer. Always pair with a viability dye to exclude late apoptotic/necrotic cells [43]. |
| Propidium Iodide (PI) | DNA-binding dye that is impermeant to live and early apoptotic cells. Labels cells with compromised membrane integrity (late apoptosis/necrosis) [43]. | Inexpensive; excited by 488 nm laser. More cell-permeable than 7-AAD [43]. |
| 7-AAD | DNA-binding dye that is less cell-permeable than PI. Also excited by 488 nm laser but emits in the far-red [43]. | A good spectral alternative to PI, especially when combining with FITC and PE conjugates [43]. |
Experimental Protocols
Protocol 1: Multiparametric Staining for Apoptosis (Caspase-3/7, PS Exposure, Membrane Integrity)
This protocol is adapted for a flow cytometer equipped with a single 488 nm laser [42] [43].
Materials:
- Complete culture medium (e.g., RPMI-1640 with 10% FBS) [45].
- Wash Buffer: Dulbecco's PBS (with calcium and magnesium) supplemented with 2% fetal bovine serum [43].
- PhiPhiLux G1D2 substrate (OncoImmunin, Inc.) [42].
- PE-conjugated Annexin V.
- Propidium Iodide (PI) stock solution (1 mg/mL in water) or 7-AAD [43].
Method:
- Induce and Harvest Cells: After applying your apoptotic stimulus, harvest cells (e.g., by gentle pipetting for adherent cells) and wash once in Wash Buffer.
- Stain for Caspase Activity: Resuspend the cell pellet (~1 x 10^6 cells) in 50 μL of a diluted PhiPhiLux G1D2 working solution (prepared as per manufacturer's instructions in complete medium). Incubate for 60 minutes at 37°C in the dark [42].
- Wash Cells: Add 1 mL of Wash Buffer to the cells, centrifuge, and carefully decant the supernatant.
- Stain for PS Exposure and Viability: Resuspend the cell pellet in 100 μL of Wash Buffer containing a pre-optimized concentration of PE-conjugated Annexin V and PI (e.g., 1 μg/mL final concentration). Incubate for 15 minutes at room temperature in the dark [43].
- Acquire Data: Without further washing, add 400 μL of Wash Buffer to the tubes and analyze immediately on the flow cytometer. Use unstained and single-stained controls for compensation.
Protocol 2: Distinguishing Apoptotic from Pyroptotic Caspase Activation
The key to this distinction lies in the combination of caspase substrates and membrane integrity dyes, as pyroptosis involves rapid plasma membrane pore formation.
Materials:
- Fluorogenic substrate for inflammatory caspases (e.g., PhiPhiLux E2D2 for caspase-1) or executioner caspases (e.g., PhiPhiLux G1D2 for caspase-3/7) [42] [43].
- A cell-impermeable DNA dye like Propidium Iodide (PI) or 7-AAD [43].
- (Optional) Gasdermin D (GSDMD) antibody to detect pyroptosis-specific pores.
Method:
- Stain for Caspase Activity: Follow steps 1-3 from Protocol 1, using your caspase substrate of choice.
- Critical Step - Stain for Membrane Integrity: Resuspend the cell pellet in buffer containing PI. Note: In pyroptosis, GSDMD pore formation allows PI uptake concurrently with or immediately after inflammatory caspase activation. In contrast, in classical apoptosis, PI uptake is a late event, occurring well after caspase-3/7 activation [24] [46].
- Acquire and Analyze Data: Analyze by flow cytometry. Co-localization of inflammatory caspase activity (Caspase-1) with rapid PI uptake strongly indicates pyroptosis. Executioner caspase activity (Caspase-3/7) with delayed or no PI uptake is indicative of apoptosis.
Signaling Pathway Diagrams
Apoptosis vs Pyroptosis Pathways
Multiparametric Flow Cytometry Workflow
Advanced Imaging Techniques for Visualizing Plasma Membrane Pores and Morphological Changes
Troubleshooting Guide: Common Experimental Issues & Solutions
FAQ 1: My membrane staining is too weak for clear visualization of pores. What could be wrong?
Issue: Faint FM4-64 or propidium iodide staining fails to resolve plasma membrane details, especially in thick maternal tissues or early-stage embryos.
Solutions:
- Optimize dye concentration and incubation time: For FM4-64 staining of plant reproductive cells, use an established in vitro ovule cultivation system with optimized dye concentration and imaging parameters [47].
- Confirm dye compatibility with your sample type: FM4-64 effectively permeates living ovules and highlights boundaries of both zygotic and maternal tissues in Arabidopsis thaliana, while propidium iodide (PI) may only show weak signals in outer ovule layers and endosperm [47].
- Use appropriate microscopy techniques: Combine staining with two-photon excitation microscopy (2PEM) or confocal microscopy for deeper tissue penetration and clearer visualization [47].
- Check developmental stage: Penetration efficiency decreases in later developmental stages. FM4-64 successfully stained 74% of 1-cell stage embryos but only 16% of 8-cell stage embryos [47].
FAQ 2: How can I distinguish between apoptosis and pyroptosis based on plasma membrane morphology?
Issue: Difficulty differentiating between apoptotic membrane blebbing and pyroptotic pore formation in cellular assays.
Solutions:
- Use multiple morphological markers: Apoptosis features cell shrinkage, membrane blebbing, and apoptotic body formation, while pyroptosis involves plasma membrane pore formation, swelling, and eventual lysis [3].
- Employ caspase-specific fluorescent reporters: Implement a novel fluorescent reporter that detects caspase-3 activation (a key apoptosis executioner) through fluorescence switch-off mechanism [48].
- Combine with lytic cell death markers: For pyroptosis, monitor release of inflammatory mediators like HMGB1, LDH, and IL-1β, which indicate gasdermin-mediated pore formation [3].
- Utilize imaging flow cytometry: This technology integrates high-throughput analysis with morphological imaging, enabling direct visualization of cell morphology and detection of subtle differences in membrane changes [49].
FAQ 3: What controls should I include when studying PANoptosis in an inflammatory model?
Issue: Uncertain validation strategies for distinguishing concurrent pyroptosis, apoptosis, and necroptosis (PANoptosis) in TNF-α-induced inflammatory models.
Solutions:
- Include pathway-specific inhibitors: Use NLRP3 inhibitors (e.g., CY-09) to block pyroptosis pathway and validate rescue from PANoptosis [50].
- Perform multi-modal staining: Combine cell death staining (e.g., PI for membrane integrity) with LDH quantification and specific molecular markers for each pathway [50].
- Employ scanning electron microscopy: This reveals multiple PANoptosis morphologies appearing in the same field of view, providing visual confirmation of concurrent cell death pathways [50].
- Validate with protein interaction studies: Use immunofluorescence and co-immunoprecipitation of closely related proteins to verify the possible presence of PANoptosome complexes [50].
Research Reagent Solutions
Table: Essential Reagents for Plasma Membrane Pore and Cell Death Imaging
| Reagent/Method | Primary Function | Application Context | Key Considerations |
|---|---|---|---|
| FM4-64 [47] | Membrane-specific fluorescent dye | Visualizing living reproductive cell morphology in plants; labels cells lacking complete walls | Permeates ovules; compatible with live imaging; superior to PI and S4B for zygote outlining |
| Caspase-3 Fluorescent Reporter [48] | Apoptosis detection via caspase-3 activity | Real-time visualization of apoptosis in human/animal cells; drug efficacy evaluation | Fluorescence switch-off upon caspase-3 cleavage; high sensitivity and simplicity |
| Imaging Flow Cytometry (IFC) [49] | High-throughput morphological imaging | Simultaneous multi-parameter analysis and visual cell death classification | Integrates FC with microscopy; enables rare cell detection and subcellular dynamics |
| PANoptosis Analysis Panel [50] | Detect concurrent PCD pathways | TNF-α-induced inflammatory models; osteogenic differentiation studies | Combines RNA sequencing, WB, cell death staining, LDH assay, and SEM |
| Gasdermin Family Probes [3] | Pyroptosis-specific detection | Identifying gasdermin-mediated pore formation | Monitor GSDMD cleavage and pore formation; different caspases cleave specific GSDMs |
| ClearSeeAlpha [47] | Tissue clearing method | Structural imaging of fixed ovules and reproductive tissues | incompatible with FM4-64; works with cell wall stains like CFW |
Table: Membrane Staining Efficiency Across Developmental Stages
| Developmental Stage | Staining Efficiency | Optimal Technique | Visualization Challenges |
|---|---|---|---|
| Egg Cell/Synergid Cells [47] | High (complete outlines) | FM4-64 with confocal/2PEM | Conventional cell wall stains (CFW) fail due to incomplete walls |
| Zygote [47] | High | FM4-64 with 2PEM | PI shows only weak signals in outer layers |
| 1-Cell Embryo [47] | 74% (20/27 samples) | FM4-64 staining | Some samples show incomplete staining |
| 8-Cell Embryo [47] | 16% (3/19 samples) | FM4-64 staining | Reduced dye penetration; weak division plane staining |
| PANoptosis Model [50] | Multiple morphologies detectable | SEM + multiplex staining | Requires multiple validation methods |
Table: Caspase Functions in Programmed Cell Death Pathways
| Caspase | Primary PCD Pathway | Key Effectors/Substrates | Regulatory Role |
|---|---|---|---|
| Caspase-1 [3] | Pyroptosis | Cleaves GSDMD; activates IL-1β/IL-18 | Can induce apoptosis in absence of GSDMD |
| Caspase-3 [3] [48] | Apoptosis (Executioner) | Cleaves PARP, lamin; activates DNA fragmentation | Also cleaves GSDME to induce pyroptosis |
| Caspase-8 [3] | Extrinsic Apoptosis | Cleaves BID, GSDMC; inhibits necroptosis | Molecular switch between apoptosis, necroptosis, and pyroptosis |
| Caspase-9 [3] | Intrinsic Apoptosis | Activates caspases-3/7 via apoptosome | Indirectly activates GSDME via caspase-3 |
| Caspase-4/5/11 [3] | Pyroptosis | Cleave GSDMD directly | Trigger pore formation in plasma membrane |
Experimental Protocols
Application: Visualizing plasma membrane dynamics in living plant ovules, egg cells, zygotes, and early embryos.
Materials:
- FM4-64 dye (N-(3-triethylammoniumpropyl)-4-(p-diethylaminophenyl-hexatrienyl) pyridinium dibromide)
- In vitro ovule cultivation system
- Confocal or two-photon excitation microscope
- Arabidopsis thaliana ovules at desired developmental stage
Procedure:
- Establish in vitro ovule cultivation system following established protocols
- Apply FM4-64 dye directly to living ovules in cultivation medium
- Optimize dye concentration and incubation time for specific tissue type
- Image using confocal microscopy or 2PEM for deeper penetration
- For quantitative live imaging, combine with 2PEM and time-lapse imaging
Technical Notes:
- FM4-64 outperforms PI and Pontamine Fast Scarlet 4B for outlining zygote boundaries
- Method applicable to broad phylogenetic range including Marchantia polymorpha and Ceratopteris richardii
- Does not require transgenic fluorescent reporters
- Incompatible with ClearSeeAlpha clearing method
Application: Real-time visualization of apoptosis in human and animal cells for drug discovery and therapeutic evaluation.
Materials:
- Engineered GFP-based apoptosis reporter containing caspase-3 cleavage motif (DEVDG)
- Cell culture system (cancer cell lines or animal cell models)
- Fluorescence microscopy or imaging flow cytometry
- Apoptosis-inducing agents (toxic substances, anticancer drugs)
Procedure:
- Transfert cells with GFP-based reporter containing caspase-3 cleavage site
- Expose to experimental conditions (toxic substances, drug treatments)
- Monitor fluorescence in real-time using time-lapse imaging
- Detect apoptosis as fluorescence "switch-off" upon caspase-3 cleavage
- Quantify apoptosis rate based on fluorescence loss kinetics
Technical Notes:
- Provides greater sensitivity and simplicity than conventional methods
- Enables high-throughput screening of drug efficacy
- Applicable to neurodegenerative disease and cancer research
- Does not require complex sample preparation or additional staining steps
Application: Comprehensive analysis of concurrent pyroptosis, apoptosis, and necroptosis in inflammatory bone disease models.
Materials:
- MC3T3-E1 cells or primary osteoblasts
- TNF-α (10-50 ng/ml)
- NLRP3 inhibitor CY-09 (10 μM)
- Cell death staining reagents, LDH quantification kit
- RNA sequencing facilities, Western blot equipment
- Scanning electron microscope
Procedure:
- Culture MC3T3-E1 cells in osteogenic differentiation medium
- Stimulate with TNF-α (10-50 ng/ml) for 72 hours
- For inhibition studies, pre-treat with CY-09 for 30 minutes before TNF-α stimulation
- Assess cell death using multiple methods:
- Cell death staining
- LDH quantification
- Propidium iodide staining
- Validate PANoptosis-related genes via RNA sequencing and Western blot
- Visualize multiple cell death morphologies using scanning electron microscopy
- Confirm protein interactions via immunofluorescence and co-immunoprecipitation
Technical Notes:
- Multiple PANoptosis morphologies appear in the same field of view under SEM
- NLRP3 inhibition rescues cells from PANoptosis and osteogenic differentiation inhibition
- Requires multimodal validation approach for conclusive results
Signaling Pathway & Experimental Workflow Diagrams
Caspase Activation in Programmed Cell Death Pathways: This diagram illustrates the complex roles of caspases as central regulators across pyroptosis, apoptosis, and necroptosis pathways, highlighting how specific caspases activate distinct effectors and can influence multiple cell death modalities [3].
PANoptosis Detection Experimental Workflow: This workflow outlines the comprehensive approach for detecting concurrent pyroptosis, apoptosis, and necroptosis in TNF-α-induced inflammatory models, incorporating multimodal validation from transcriptional analysis to morphological confirmation [50].
Membrane Imaging Technique Selection Guide: This decision workflow helps researchers select appropriate imaging methodologies based on their specific sample type and research objectives, connecting various advanced techniques to their optimal applications [47] [48] [49].
Navigating Experimental Complexities: Resolving Ambiguity in Cell Death Classification
Frequently Asked Questions
FAQ 1: My experiment shows caspase-8 activation with an NLRP3 trigger. Is this a sign of a failed experiment or a biologically relevant mixed phenotype? Answer: This can be a biologically relevant switch, not necessarily a failed experiment. In models of impaired canonical NLRP3 signaling (e.g., caspase-1 deficiency or delayed trigger delivery), the NLRP3/ASC platform can divert from recruiting caspase-1 (pyroptosis) to recruiting caspase-8, initiating an apoptotic cascade [51]. You should confirm the experimental conditions and use multiple assays to characterize the cell death modality fully [52].
FAQ 2: How can I distinguish between reduced cell proliferation and increased cell death when my viability assay shows a positive result? Answer: Viability assays alone are insufficient to make this distinction, as identical viability readouts can mask opposing effects on growth and death rates [53]. To resolve this, use complementary methods:
- Real-time imaging with dyes for apoptosis (Annexin V) and cell death (membrane-impermeable DNA dyes) [53].
- Proliferation assays (e.g., CFSE-based) to directly measure cell division rates [53].
- Endpoint analysis of apoptotic markers (e.g., cleaved caspase-3) to confirm death mechanisms [53].
FAQ 3: My cells show mixed morphological features. What is the gold standard for confirming the primary cell death pathway? Answer: There is no single gold standard. The most robust approach is to perform multiple, methodologically unrelated assays [52]. Relying on a single parameter (e.g., morphology alone) can be misleading. A hierarchical approach is recommended:
- General techniques to identify dead cells (e.g., plasma membrane integrity dyes like trypan blue) [52].
- Specific techniques to classify the death subroutine (e.g., caspase activation assays, analysis of specific substrates like GSDMD or GSDME) [52] [51].
FAQ 4: The inhibitor VX-765 (Belnacasan) is suppressing caspase-8 and -3 in my model. Is it acting as a pan-caspase inhibitor? Answer: Potentially, yes. Research has shown that in specific experimental models, VX-765 can exhibit a "pan-caspase inhibitor-like effect," dose-dependently suppressing the activation of caspase-8, -9, and -3 [51]. It is crucial to verify the specificity of pharmacological inhibitors in your specific system, as off-target effects can occur.
The Scientist's Toolkit: Essential Research Reagents
The following table details key reagents used in experiments investigating apoptotic and pyroptotic caspase activation.
| Research Reagent | Primary Function / Application |
|---|---|
| VX-765 (Belnacasan) | A caspase-1 inhibitor; however, it can show pan-caspase inhibitory effects in some models, suppressing caspase-8/-9/-3 activation [51]. |
| Lipopolysaccharide (LPS) | A pathogen-associated molecular pattern (PAMP) used to "prime" innate immune cells (e.g., macrophages), inducing the expression of NLRP3 and pro-IL-1β [51]. |
| Nigericin | A potassium ionophore commonly used as a potent trigger for NLRP3 inflammasome activation after cellular priming [51]. |
| ATP | An endogenous danger signal (DAMP) that activates the P2X7 receptor, serving as a trigger for NLRP3 inflammasome assembly [51]. |
| Annexin V | A fluorescent probe used to detect phosphatidylserine exposure on the outer leaflet of the plasma membrane, an early marker of apoptosis [53]. |
| YOYO-3 / Propidium Iodide (PI) | Membrane-impermeable DNA dyes used to identify cells with compromised plasma membranes (a late apoptotic or necrotic feature) [53] [51]. |
| Antibodies (Cleaved Caspases, GSDMD, GSDME) | Essential for Western blotting and immunofluorescence to detect the activation (cleavage) of key executioners in apoptosis (caspase-3, -8) and pyroptosis (caspase-1, GSDMD) [51] [5]. |
Quantitative Data & Experimental Protocols
Table 1: Key Caspase Classes and Their Roles in Cell Death
| Caspase Class | Example Members | Primary Role in Cell Death | Key Features / Substrates |
|---|---|---|---|
| Initiator | Caspase-8, -9, -10 | Initiate apoptotic pathways; caspase-8 can also engage with inflammasomes [51] [5]. | Activated by upstream death platforms (e.g., DISC, apoptosome). |
| Inflammatory | Caspase-1, -4, -5, -11 | Mediate pyroptosis and cytokine maturation (IL-1β, IL-18) [5]. | Activated by inflammasomes; cleave Gasdermin D (GSDMD). |
| Executioner | Caspase-3, -6, -7 | Carry out the final stages of the apoptotic program [5]. | Cleave numerous cellular substrates (e.g., PARP, ICAD); can also cleave GSDME to induce secondary necrosis/pyroptosis [51]. |
Table 2: Core Methodologies for Distinguishing Cell Death Pathways
| Method Category | Specific Technique | Key Readout | Advantage | Potential Pitfall |
|---|---|---|---|---|
| Vital Staining | Trypan Blue Exclusion | Plasma membrane integrity; identifies dead cells [52]. | Simple, inexpensive, rapid. | Does not differentiate between death subroutines [52]. |
| Morphology | Light & Electron Microscopy | Cellular and nuclear morphology (e.g., shrinkage, blebbing, swelling) [52]. | Direct visualization; can identify mixed morphotypes. | Operator-dependent; may miss early-stage cells [52]. |
| Flow Cytometry | Annexin V/PI Staining | Phosphatidylserine exposure (Apoptosis) & membrane integrity [53]. | Quantifies populations of live, early apoptotic, and late apoptotic/dead cells. | Cannot definitively classify non-apoptotic death. |
| Immunoblotting | Western Blot | Cleavage of caspases, GSDMD, GSDME, PARP [51] [5]. | Confirms specific biochemical events; widely accessible. | Semi-quantitative; requires cell lysis. |
| Live-Cell Imaging | Kinetic Analysis with Fluorescent Dyes | Real-time changes in cell number, apoptosis, and death markers [53]. | Provides dynamic, high-content data on growth vs. death rates. | More complex setup and data analysis. |
Experimental Protocol 1: Kinetic Analysis of Cell Growth and Death This protocol is adapted from methods used to reveal that compounds with identical viability effects can have divergent impacts on growth and death rates [53].
- Cell Seeding: Seed cells in a multi-well plate suitable for live-cell imaging.
- Staining and Treatment: Treat cells with the compound of interest. Include a staining solution containing:
- A fluorescent Annexin V conjugate to mark apoptotic cells.
- A membrane-impermeable DNA dye (e.g., YOYO-3) to mark dead cells.
- Hoechst 33342 or similar to label all nuclei.
- Image Acquisition: Place the plate in a live-cell imager. Acquire images from multiple sites per well at regular intervals (e.g., every 4-6 hours) for 48-72 hours.
- Data Analysis: Use image analysis software to quantify the total area of cells (phase contrast), Annexin V signal, and YOYO-3 signal over time. Fit this kinetic data to a mathematical model that infers cell division and death rates.
Experimental Protocol 2: Assessing Caspase Switching in NLRP3-Impaired Models This protocol is based on research showing diversion from pyroptosis to apoptosis when canonical NLRP3 signaling is impaired [51].
- Cell Models: Use primary macrophages or macrophage cell lines. Compare wild-type (WT) cells with genetically modified models (e.g., caspase-1 deficient) or experimental models of impairment (e.g., delayed delivery of the NLRP3 trigger).
- Inflammasome Activation:
- Priming: Prime cells with LPS (e.g., 100 ng/ml for 3-4 hours) to induce pro-IL-1β and NLRP3 expression.
- Triggering: Activate the NLRP3 inflammasome by adding ATP or nigericin.
- For the "delayed trigger" model, add the trigger after an extended period post-priming (e.g., 16 hours).
- Sample Collection: Collect cell culture supernatants and lysates at various time points post-triggering.
- Downstream Analysis:
- Western Blotting: Probe lysates for cleavage of caspase-1, caspase-8, caspase-3, GSDMD, and GSDME.
- ELISA: Measure the release of mature IL-1β from supernatants to confirm pyroptotic activity.
- Cell Viability Assay: Use a method like PI uptake or LDH release to quantify cell death.
Signaling Pathways and Experimental Workflows
Welcome to this technical support center, dedicated to the investigation of cell death pathway switching. Within the broader context of distinguishing apoptotic versus pyroptotic caspase activation, a critical phenomenon has been identified: impaired canonical pyroptotic signaling can divert the cellular response to apoptotic caspase activation [54]. This guide provides detailed troubleshooting and methodological support for researchers studying this complex crossover, which is crucial in host defense, chronic inflammation, and the response to pathogen evasion strategies [54] [55].
The core concept is that the NLRP3 inflammasome platform, which typically recruits and activates caspase-1 to drive pyroptosis, demonstrates remarkable plasticity. When the canonical pathway is disrupted—whether by genetic deficiency of caspase-1, pharmacological inhibition, or delayed triggering—the platform can pivot to recruit apoptotic caspases like caspase-8, initiating an apoptotic response [54] [9].
Core Signaling Pathways and Molecular Switches
Visualizing the Pathway Switch
The diagram below illustrates the key molecular decision points between pyroptosis and apoptosis, and how impaired signaling diverts the pathway.
Key Molecular Regulators of the Switch
The transition from pyroptosis to apoptosis is governed by specific molecular interactions and regulators, as detailed in the table below.
Table 1: Key Molecular Regulators in the Pyroptosis-to-Apoptosis Switch
| Molecule/Pathway | Function in Canonical Pyroptosis | Role in Pathway Diversion |
|---|---|---|
| NLRP3/ASC Platform | Recruits and activates caspase-1 [54] | Recruits caspase-8 when caspase-1 is deficient or delayed [54] |
| Caspase-1 | Inflammatory caspase; cleaves GSDMD and pro-IL-1β [56] | Its inhibition or deficiency is the primary switch signal [54] |
| Caspase-8 | Apoptotic initiator caspase; not typically involved [56] | Activated by the NLRP3/ASC platform; initiates apoptotic cascade [54] [55] |
| Caspase-3 | Apoptotic executioner caspase [56] | Activated downstream of caspase-8; cleaves GSDME [54] [9] |
| GSDMD | Executes pyroptosis; forms plasma membrane pores [28] | Activity is suppressed when pathway is diverted [54] |
| GSDME | Generally inactive [9] | Cleaved by caspase-3, leading to secondary necrosis [54] [9] |
| Mitochondria | Not directly involved in canonical pathway [56] | Upstream damage occurs, contributing to caspase-9/-3 activation [54] |
Experimental Models & Quantitative Data
Established Cell Models for Studying Pathway Switching
Researchers can employ several well-characterized cellular models to induce and study the diversion from pyroptosis to apoptosis.
Table 2: Experimental Models for Inducing Pathway Diversion
| Experimental Model | Method of Induction | Key Readouts of Apoptotic Diversion |
|---|---|---|
| Caspase-1 Deficient Macrophages | Genetic knockout (e.g., Casp1-/- BMDMs) [54] | Activation of caspase-8/-9/-3; GSDME cleavage; reduced LDH/GSDMD cleavage [54] |
| Pharmacologic Inhibition | Pre-treatment with VX-765 (Belnacasan) prior to nigericin/ATP [54] | Dose-dependent suppression of caspase-8/-9/-3; inhibition of GSDME cleavage [54] |
| Delayed Triggering Model | LPS priming followed by delayed addition of nigericin (> several hours) [54] | Shift from caspase-1 to caspase-8 recruitment on the ASC speck [54] |
| TNF-α Induction | Stimulation of MC3T3-E1 osteoblasts with TNF-α (10-50 ng/mL) [21] | Co-activation of pyroptotic, apoptotic, and necroptotic markers (PANoptosis) [21] |
| Methylglyoxal (MGO) Model | Treatment of HUVECs with MGO (e.g., 0.4-1.6 mM) [9] | Caspase-3/GSDME-mediated pyroptosis; can be shifted to apoptosis by agents like DT-13 [9] |
Quantitative Assessment of Cell Death Markers
Accurately distinguishing between pyroptosis and apoptosis requires a multi-parametric approach, as key markers can overlap.
Table 3: Quantitative and Morphological Distinctions Between Pyroptosis and Apoptosis
| Parameter | Pyroptosis | Apoptosis (Diversion Pathway) |
|---|---|---|
| Primary Caspase | Caspase-1 [56] [57] | Caspase-8 and Caspase-3 [54] [56] |
| Gasdermin Cleavage | GSDMD [56] [57] | GSDME (cleaved by caspase-3) [54] [9] |
| Plasma Membrane | Pore formation, followed by lytic rupture [56] [28] | Intact until late stage (secondary necrosis) [56] |
| Morphology | Cell swelling, large bubbles [56] [28] | Cell shrinkage, membrane blebbing [56] |
| Nuclear Morphology | Nuclear condensation, intact [28] | Nuclear fragmentation (karyorrhexis) [56] |
| Inflammation | Highly inflammatory (IL-1β, IL-18 release) [56] [28] | Generally non-inflammatory [56] |
| PI / 7-AAD Staining | Positive (pores allow dye uptake) [28] | Negative until secondary necrosis [56] |
| Key Metric: LDH Release | High Release (full cytolysis) [58] | Low/Moderate Release (only after secondary necrosis) [54] |
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Reagents for Investigating Pyroptosis-to-Apoptosis Switching
| Reagent / Kit | Specific Example (Catalog Number) | Primary Application in This Context |
|---|---|---|
| Caspase-1 Inhibitor | VX-765 (Belnacasan) [54] | To chemically impair the canonical pyroptosis pathway and induce diversion. |
| Caspase-8 Inhibitor | Z-IETD-FMK | To confirm the role of caspase-8 in the diverted apoptotic pathway. |
| Caspase-3/7 Assay | CellEvent Caspase-3/7 Green ReadyProbes [59] | Live-cell detection of apoptotic executioner caspase activity. |
| LDH Cytotoxicity Kit | Sigma #11644793001 [59] | Quantify plasma membrane rupture, distinguishing lytic (pyroptosis) from non-lytic (apoptosis) death. |
| ELISA for IL-1β | R&D Systems DY401 [59] | Measure inflammasome-specific output; levels decrease upon diversion to apoptosis. |
| Antibody: Cleaved Caspase-3 | Cell Signaling Technology #9664 [54] | Western blot confirmation of apoptotic pathway engagement. |
| Antibody: GSDMD | Abcam #ab209845 [54] | Detect full-length and pyroptosis-associated cleaved GSDMD. |
| Antibody: GSDME | Abcam #ab215191 [54] | Detect apoptosis-associated, caspase-3-cleaved GSDME. |
| Cell Death Stains Propidium Iodide (PI), Annexin V kits [54] [59] | Flow cytometry to distinguish early apoptosis (Annexin V+/PI-) from lytic death (Annexin V+/PI+). |
Frequently Asked Questions (FAQs) & Troubleshooting
Q1: I used VX-765 to inhibit caspase-1, but I do not see a clear activation of apoptotic caspases. What could be wrong?
- Check Inhibitor Potency and Timing: Ensure VX-765 is used at an effective concentration (e.g., 20-50 µM) and is added to cells before the NLRP3 triggering stimulus (e.g., nigericin or ATP) [54].
- Verify Model Suitability: Confirm your cell model robustly expresses the apoptotic machinery. Primary macrophages are often more reliable than some immortalized lines [58].
- Optimize Triggering Signal: The switch may be dependent on the strength and duration of the NLRP3 stimulus. Titrate your trigger (e.g., nigericin concentration) to find the optimal window for observing diversion [54].
Q2: How can I definitively prove that the cell death I'm observing after caspase-1 inhibition is apoptosis and not another form of death?
- Multi-Parametric Analysis: Do not rely on a single assay. Combine techniques:
- Western Blot: Look for cleaved, active fragments of caspase-8, caspase-3, and PARP, alongside a reduction in GSDMD cleavage [54].
- Flow Cytometry: Use Annexin V/PI co-staining. Apoptotic cells will be Annexin V+/PI- early in the process, while pyroptotic cells become PI+ rapidly due to membrane pores [56] [28].
- Morphology: Use live-cell imaging to observe characteristic apoptotic morphology like cell shrinkage and blebbing, which contrasts with the cell swelling and large bubbles of pyroptosis [56] [28].
Q3: My results show markers for both pyroptosis and apoptosis. Is this possible?
- Yes, this is a recognized phenomenon. Cells can simultaneously engage multiple death pathways, a concept known as PANoptosis [55] [21]. This is particularly common under strong inflammatory stimuli like TNF-α or specific infections [55] [21]. Your observation may be valid. Focus on the dominant pathway by quantifying the relative levels of key markers (e.g., GSDMD vs. GSDME cleavage) and functional outputs (e.g., IL-1β release).
Q4: What is the role of GSDME in this process?
- GSDME acts as a molecular switch downstream of caspase-3 activation [9]. When caspase-3 cleaves GSDME, the N-terminal fragments form pores in the plasma membrane, causing the apoptotic cells to undergo lytic death, which can be characterized as secondary necrosis [54] [9] [28]. Therefore, in the context of pathway diversion, GSDME cleavage is a key event that blurs the lines between apoptotic and pyroptotic morphology at the terminal stage.
Detailed Experimental Protocol: Inducing and Validating the Switch
This protocol outlines the key steps for inducing the diversion from pyroptosis to apoptosis in macrophages using caspase-1 inhibition, based on methodologies from the search results [54] [58].
Workflow Visualization
Step-by-Step Procedure
Step 1: Cell Seeding and Priming
- Seed wild-type (WT) bone marrow-derived macrophages (BMDMs) or THP-1-derived macrophages in appropriate plates 24 hours before stimulation.
- Prime cells with ultrapure LPS (e.g., 100 ng/mL) for 3-4 hours. This provides "signal 1," upregulating NLRP3 and pro-IL-1β [54] [60].
Step 2: Pathway Inhibition
- Pre-treat cells with the caspase-1 inhibitor VX-765 (Belnacasan, e.g., 20-50 µM) or vehicle control (DMSO) for 30-60 minutes before adding the NLRP3 trigger [54].
Step 3: Inflammasome Activation
- Activate the NLRP3 inflammasome by adding the trigger nigericin (e.g., 5-10 µM) or ATP (e.g., 5 mM) for 1-2 hours. This provides "signal 2" [54] [58].
Step 4: Sample Collection
- Collect cell culture supernatants to analyze released LDH, IL-1β (by ELISA), and other DAMPs.
- Harvest cell lysates for Western blot analysis.
- For flow cytometry, gently detach cells (if adherent) and combine with supernatant to collect all cells.
Step 5: Downstream Analysis (Perform in Parallel)
- Western Blot: Probe lysates for:
- Pyroptosis Markers: Cleaved caspase-1 (p20), cleaved GSDMD.
- Apoptosis Markers: Cleaved caspase-8, cleaved caspase-3, cleaved PARP, cleaved GSDME.
- LDH Release Assay: Quantify cytotoxicity in supernatants. Expect high LDH in the LPS+Nigericin (vehicle) control (pyroptosis) and lower but significant LDH in the LPS+Nigericin+VX-765 group (apoptosis with secondary necrosis) [54] [58].
- ELISA for IL-1β: Measure mature IL-1β in supernatants. Levels should be significantly reduced in the VX-765 treated group compared to the pyroptosis control [54].
- Flow Cytometry: Stain cells with Annexin V and PI to distinguish apoptotic (Annexin V+/PI-) from lytic (Annexin V+/PI+) populations [59].
Frequently Asked Questions (FAQs)
Q1: Why can the same caspase, like caspase-8, trigger different types of cell death in different cell types? Caspase-8 acts as a key molecular switch whose function is highly dependent on cellular context. It can initiate extrinsic apoptosis, inhibit necroptosis, or, under specific conditions such as in macrophages where caspase-1 is inhibited or certain chemotherapeutic agents are present, cleave gasdermins (GSDMC, GSDMD) to induce pyroptosis [3] [28] [51]. The outcome is determined by the specific proteins available in the cell, including the expression levels of gasdermins and the activation status of other signaling pathways.
Q2: How reliable are caspase activity assays for identifying a specific cell death pathway? Use these assays with caution. Caspase cleavage specificities often overlap, meaning a single substrate is not exclusive to one caspase. For example, the IETD substrate used for caspase-8 can also be cleaved by caspases-3, -6, and -10 [61]. To confirm specific caspase activation, best practice is to use a combination of methods, such as an activity assay alongside western blot detection of the cleaved (active) caspase form [61].
Q3: What happens to the cell death pathway when a key pyroptosis component is missing? Cells can exhibit remarkable plasticity. Research shows that in GSDMD-knockout monocytes and macrophages, caspase-1 activation diverts from pyroptosis to apoptosis, leading to the activation of caspases-3 and -7 and PARP cleavage [1]. Similarly, in caspase-1-deficient macrophages, NLRP3 inflammasome activation can switch to caspase-8/-9/-3 activation and GSDME-mediated secondary necrosis [51]. This demonstrates the existence of robust backup cell death mechanisms.
Q4: What is a key morphological difference between apoptosis and pyroptosis? A key difference lies in plasma membrane integrity. In apoptosis, the membrane remains intact but undergoes scrambling and blebbing, leading to the formation of apoptotic bodies. In pyroptosis, the membrane is compromised by gasdermin pore formation, leading to cell swelling, osmotic lysis, and the release of inflammatory contents [62] [1].
Troubleshooting Guides
Issue 1: Unexpected or Inconclusive Caspase Activation Results
Problem: Your caspase activity assay shows positive signal, but other cell death markers do not align with the expected pathway, or results are inconsistent with your experimental manipulation.
| Potential Cause | Explanation & Diagnostic Steps | Recommended Solution |
|---|---|---|
| Caspase Assay Cross-Reactivity | The synthetic substrate peptide is being cleaved by multiple caspases [61]. | Confirm with orthogonal methods. Use western blot to detect caspase-specific cleavage events (e.g., pro-caspase-3 to cleaved caspase-3) [61]. |
| Cell Type-Specific Caspase Expression | The abundance and expression of caspases vary significantly between different cell types [61]. | Profile baseline caspase expression in your specific cell model using western blot or RNA-seq data before designing experiments. |
| Kinetics of Activation | You may be measuring caspase activity at a time point that is too early or too late for the peak of activation of your target caspase [61]. | Perform a time-course experiment to establish the kinetics of caspase activation under your specific treatment conditions. |
| Pathway Switching | Inhibition or deficiency in one PCD pathway may cause the signal to divert to another. For example, impaired NLRP3 signaling can divert pyroptosis to apoptosis [51]. | Analyze multiple PCD markers simultaneously. In cases of suspected switching, check for gasdermin cleavage (pyroptosis) and PARP cleavage (apoptosis) [1]. |
Issue 2: Differentiating Between Apoptosis and Pyroptosis in a Mixed Population
Problem: Your cell population appears to be undergoing cell death, but you are unable to conclusively determine if it is apoptotic, pyroptotic, or a mixture of both.
| Feature | Apoptosis | Pyroptosis |
|---|---|---|
| Primary Caspases | Initiator: Caspase-8, -9; Executioner: Caspase-3, -6, -7 [3] [62] | Inflammatory: Caspase-1, -4, -5, -11; Also: Caspase-3, -8 (via Gasdermin cleavage) [3] [28] |
| Key Executioner Protein | None (caspases cleave hundreds of substrates) | Gasdermin Family (GSDMD, GSDME, etc.) [28] |
| Membrane Integrity | Maintained until late stages [62] | Disrupted by gasdermin pores [62] |
| Morphology | Cell shrinkage, membrane blebbing, apoptotic bodies [62] | Cell swelling, formation of large bubbles, eventual lysis [28] [62] |
| Inflammatory Response | Generally non-inflammatory [3] | Highly inflammatory (release of IL-1β, IL-18, HMGB1) [3] [28] |
| DNA Fragmentation | Ordered, internucleosomal cleavage ("DNA ladder") [63] | Random, TUNEL-positive but with less intensity [28] |
Solution: Employ a multi-parametric approach:
- Morphology: Use real-time imaging to observe for swelling/lysis (pyroptosis) vs. shrinkage/blebbing (apoptosis).
- Membrane Integrity: Perform a propidium iodide (PI) uptake assay. Pyroptotic cells, with their membrane pores, will be PI-positive early, while apoptotic cells maintain membrane integrity until late stages [28].
- Biochemical Markers: By western blot, analyze for:
Experimental Protocols
Protocol 1: Differentiating Caspase-1-Mediated Pyroptosis from Apoptosis via Western Blot
This protocol is designed to help determine if cell death is proceeding via canonical pyroptosis or has been diverted to apoptosis, particularly useful when studying knockout models or inhibitory conditions [51] [1].
Key Reagents:
- LPS (for priming inflammasome)
- NLRP3 activator (e.g., Nigericin, ATP)
- Caspase-1 inhibitor (e.g., VX-765/Belnacasan) [51]
- RIPA Lysis Buffer
- Antibodies: Pro/cleaved caspase-1, GSDMD (full length and N-terminal), Pro/cleaved caspase-3, PARP, β-Actin
Methodology:
- Cell Stimulation: Seed and differentiate your immune cells (e.g., primary macrophages or THP-1 cells). Prime cells with LPS (e.g., 100 ng/mL, 3-4 hours). Stimulate with an NLRP3 activator like Nigericin (e.g., 5-10 µM) for a predetermined time (e.g., 30-90 mins). Include conditions with a caspase-1 inhibitor (e.g., VX-765, 10-20 µM) added prior to Nigericin.
- Protein Extraction: Lyse cells in RIPA buffer supplemented with protease inhibitors. Centrifuge at high speed (e.g., 12,000-14,000 x g) for 10 minutes at 4°C to collect total protein lysates. Determine protein concentration.
- Western Blot: Load 20-30 µg of protein per lane on an SDS-PAGE gel. Transfer to a PVDF membrane and block. Probe with the key antibodies.
- Interpretation of Results:
- Canonical Pyroptosis: Cleavage of caspase-1 (p10/p20 fragments) and GSDMD (appearance of GSDMD-NT fragment). Little to no cleavage of caspase-3 or PARP.
- Diverted Apoptosis: In GSDMD-KO or Casp1-KO cells, or with caspase-1 inhibition: Absence of GSDMD-NT and active caspase-1, but presence of cleaved caspase-3 and cleaved PARP [51] [1].
- Mixed Death: Presence of both GSDMD-NT and cleaved caspase-3/PARP.
Protocol 2: DNA Fragmentation Analysis to Support Apoptosis Identification
This protocol provides a semi-quantitative method to detect the characteristic internucleosomal DNA laddering of apoptosis, which differs from the more random DNA cleavage in pyroptosis [28] [63].
Key Reagents:
- Cell Lysis Buffer: 10 mM Tris-HCl (pH 7.4), 5 mM EDTA, 0.2% Triton X-100
- DNase-free RNase A
- Proteinase K
- Phenol/Chloroform/Isoamyl Alcohol (25:24:1)
- Ethanol and Sodium Acetate (3 M, pH 5.2)
- Agarose gel electrophoresis equipment
Methodology:
- Harvest and Lyse Cells: Pellet ~1x10^6 cells. Resuspend in 0.5 mL of ice-cold lysis buffer. Vortex and incubate on ice for 30 minutes.
- Separate Fragmented DNA: Centrifuge the lysate at 27,000 x g for 30 minutes at 4°C. The supernatant contains the fragmented DNA.
- Precipitate DNA: Transfer the supernatant to a new tube. Add 50 µL of 5 M NaCl, 600 µL ethanol, and 150 µL 3 M sodium acetate. Mix and incubate at -80°C for 1 hour. Pellet the DNA by centrifugation at 20,000 x g for 20 minutes.
- Digest RNA and Proteins: Dissolve the DNA pellet. Add DNase-free RNase A and incubate at 37°C for several hours. Then add Proteinase K and incubate overnight at 65°C.
- Purify and Visualize DNA: Re-extract DNA with phenol/chloroform/isoamyl alcohol and precipitate with ethanol. After a final wash, air-dry the pellet and resuspend in TE buffer. Separate the DNA on a 2% agarose gel containing ethidium bromide and visualize under UV light [63].
- Interpretation: A positive result for apoptosis is a "ladder" of DNA fragments in multiples of ~180-200 base pairs. Pyroptotic cells may show a more diffuse "smear" or less intense, random fragmentation [28].
Signaling Pathway Diagrams
Caspase Interplay in Cell Death
Experimental Workflow for Cell Death Pathway Analysis
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Assay | Primary Function | Key Considerations & Cross-Reactivity |
|---|---|---|
| Caspase Activity Assays | Measure protease activity via cleavage of colorimetric/fluorogenic substrates. | High cross-reactivity: IETD (Casp-8) is also cleaved by Casp-3, -6, -10. DEVD (Casp-3) is also cleaved by Casp-7. Always confirm with western blot [61]. |
| Gasdermin Antibodies | Detect cleavage and activation of pyroptosis executioners (e.g., GSDMD, GSDME). | Crucial for definitive pyroptosis identification. Look for the appearance of the N-terminal fragment [28] [51]. |
| Cell Death Inhibitors | Pharmacologically inhibit specific caspases or pathways to establish mechanism. | VX-765 (Belnacasan): A caspase-1 inhibitor that may show pan-caspase effects at higher doses [51]. Use dose-response studies. |
| Propidium Iodide (PI) | Fluorescent dye that stains DNA in cells with compromised membranes. | Pyroptotic cells are PI+ early due to GSDMD pores. Apoptotic cells are PI- until late/secondary necrosis [28] [51]. |
| Western Blot Antibodies | Detect specific protein expression and cleavage events. | Essential panel: Pro/cleaved Casp-1, Pro/cleaved Casp-3, GSDMD (FL/NT), PARP. Provides a multi-parametric view of activation [51] [1]. |
FAQs: Addressing Common Experimental Challenges
FAQ 1: How can I determine if cell death in my experiment is apoptosis or pyroptosis? Morphological and biochemical hallmarks can distinguish these pathways. Apoptotic cells maintain membrane integrity until late stages, form apoptotic bodies, and exhibit ordered DNA fragmentation. Pyroptotic cells undergo plasma membrane pore formation, cellular swelling, release inflammatory cytokines like IL-1β and IL-18, and show random DNA fragmentation with lower intensity than apoptosis [28] [64] [24]. Key biochemical markers include caspase-3/7 activation and PARP cleavage for apoptosis, versus GSDMD cleavage and caspase-1 activation for pyroptosis.
FAQ 2: My caspase inhibitor isn't producing the expected effect. What could be wrong? This could be due to several factors. The inhibitor may lack specificity for your target caspase; for example, VX-765, a caspase-1 inhibitor, can exhibit pan-caspase inhibitory effects at higher concentrations [51]. Check the inhibitor's mechanism of action (reversible/irreversible, competitive/non-competitive) and cellular context, which can cause pathway diversion (e.g., from pyroptosis to apoptosis) [9] [51]. Always validate inhibition of the intended target and use multiple orthogonal probes with different chemotypes to confirm phenotype specificity [65].
FAQ 3: How do I validate that my inhibitor is acting specifically on the intended target? Perform comprehensive selectivity profiling against related targets, ideally demonstrating >10-100-fold potency for your target versus other family members [65]. Use negative controls like inactive structural analogs (R/S stereoisomers) and positive controls with known effects [65]. Conduct washout experiments to assess phenotypic consequences after inhibitor removal, which provides insight into target vulnerability [65]. Complementary experiments with RNAi or genetic mutants can build consensus about the target's role [65].
Troubleshooting Guides
Table 1: Troubleshooting Inhibitor Specificity Issues
| Problem | Possible Causes | Recommended Solutions |
|---|---|---|
| Unexpected cell death phenotype | Off-target effects; Pathway diversion; Incorrect potency | Use lowest effective concentration; Profile selectivity in biochemical assays; Include multiple cell death markers [65] [51] |
| No phenotypic effect | Insufficient cellular permeability; Rapid metabolism; Incorrect mechanism of action | Verify cellular permeability assays; Check metabolic stability; Confirm mechanism of action (competitive vs. allosteric) [65] |
| Inconsistent results between assays | Different cellular contexts; Varied substrate concentrations; Assay timeframe mismatches | Standardize physiological context; Account for substrate concentration in potency calculations (Cheng-Prusoff); Align assay timeframe with expected phenotypic effect [65] |
| High background or toxic effects | Chemical reactivity; Pan-assay interference; Membrane destabilization | Check for toxic moieties and PAINS; Ensure solubility in relevant media; Balance lipophilicity for permeability vs. solubility [65] |
Table 2: Distinguishing Apoptotic vs. Pyroptotic Caspase Activation
| Feature | Apoptosis | Pyroptosis |
|---|---|---|
| Key Initiator Caspases | Caspase-2, -8, -9, -10 [3] | Caspase-1, -4, -5, -11 [3] [64] |
| Key Executioner Caspases | Caspase-3, -6, -7 [3] | Caspase-1, -3 (via GSDME), -8 (via GSDMC) [3] |
| Key Effector Proteins | PARP, Lamin proteins [3] | GSDMD, GSDME, GSDMB [3] [28] |
| Morphological Outcome | Membrane blebbing, apoptotic bodies, phagocytic clearance [3] | Plasma membrane pores, cell swelling, osmotic lysis [28] [64] |
| Inflammatory Response | Generally non-inflammatory [3] | Highly inflammatory (IL-1β, IL-18 release) [64] |
| Primary Molecular Triggers | Death receptors, mitochondrial perturbation [3] | Inflammasome activation, cytoplasmic LPS [64] |
Research Reagent Solutions
Table 3: Essential Reagents for Cell Death Pathway Dissection
| Reagent Category | Specific Examples | Function & Application |
|---|---|---|
| Caspase Inhibitors | Z-VAD-FMK (pan-caspase), Z-DEVD-FMK (caspase-3/7), VX-765 (caspase-1) [9] [51] | Pharmacological inhibition to establish caspase dependency in cell death pathways |
| IAP Antagonists | SMAC mimetics [66] | Promote apoptosis by counteracting IAP-mediated caspase inhibition |
| Gasdermin Cleavage Reporters | Anti-GSDMD, Anti-GSDME antibodies [9] [51] | Detect gasdermin cleavage as specific marker for pyroptosis |
| Cell Death Detection Assays | Propidium iodide, TUNEL staining, LDH release, IL-1β/IL-18 ELISAs [28] [51] | Differentiate membrane integrity loss, DNA damage patterns, and inflammatory cytokine release |
| Pathway-Specific Activators | Nigericin (NLRP3), ATP (P2X7), LPS (caspase-4/5/11) [64] [51] | Specific stimulation of pyroptotic pathways |
Experimental Protocols
Protocol 1: Assessing Pyroptosis-to-Apoptosis Transition This protocol is adapted from research on DT-13-mediated cell death transition [9]:
- Culture HUVECs in ECM medium with 5% FBS and passage at 70-80% confluence
- Treat cells with Methylglyoxal (0.4-1.6 mM) for 24h to induce pyroptosis via Caspase-3/GSDME pathway
- Pre-treat with DT-13 (1 μM) for 6h prior to MGO exposure to promote apoptosis transition
- Assess cell death morphology: pyroptosis shows swelling and large bubbles; apoptosis shows membrane blebbing and apoptotic bodies
- Validate by Western blotting for GSDME cleavage (pyroptosis marker) and PARP cleavage (apoptosis marker)
- Confirm mechanism by examining MLC2 phosphorylation and NMMHC IIA cleavage
Protocol 2: Evaluating Caspase Diversion in Impaired Inflammasome Signaling This protocol models caspase switching when canonical pathways are disrupted [51]:
- Differentiate THP-1 macrophages with PMA (100 nM, 24h) or use primary mouse BMDMs
- Prime cells with LPS (100-500 ng/mL, 3-4h) to induce NLRP3 and pro-IL-1β expression
- For delayed triggering model: Add nigericin (10 μM) after extended LPS priming (e.g., 12-24h)
- For caspase-1 deficiency model: Use caspase-1-/- macrophages or inhibit with VX-765 (10-50 μM)
- Assess caspase-8 recruitment to NLRP3-ASC complex by immunoprecipitation/Western blot
- Monitor downstream activation of caspase-9/-3 and GSDME cleavage
- Measure mitochondrial damage via JC-1 staining (membrane potential) and ROS production
Signaling Pathway Visualizations
Caspase Activation Pathways and Inhibitor Targets
Cell Death Pathway Identification
Fundamental Concepts
What is PANoptosis and how does it differ from other cell death pathways?
PANoptosis is a novel and complex inflammatory programmed cell death (PCD) pathway that was first conceptualized by Kanneganti's research team in 2019. It is characterized by the simultaneous activation of key features from three major PCD pathways: pyroptosis, apoptosis, and necroptosis [67]. The term "PANoptosis" was coined to describe a cell death phenomenon that possesses the essential characteristics of these three pathways but cannot be fully explained by any one of them alone [67] [68].
Unlike traditional views that treated these death pathways as independent, PANoptosis represents a "death continuum" with extensive crosstalk among the molecular components. This integrated cell death pathway is governed by the assembly of large multiprotein complexes called PANoptosomes, which serve as activation platforms that synchronize all three death mechanisms under specific stimuli [67].
What are the key morphological and biochemical features that distinguish PANoptosis from isolated cell death pathways?
Recognizing PANoptosis requires understanding both the shared and distinct features of its component pathways. The table below summarizes the key characteristics:
Table 1: Comparative Analysis of Cell Death Pathways in PANoptosis
| Feature | Apoptosis | Pyroptosis | Necroptosis | PANoptosis |
|---|---|---|---|---|
| Morphology | Cell shrinkage, membrane blebbing, apoptotic bodies [27] | Cell swelling, bubble-like protrusions, membrane rupture [28] | Cytoplasmic swelling, organelle dysfunction, membrane rupture [27] | Combined features that cannot be explained by any single pathway alone [67] |
| Membrane Integrity | Maintained until late stages [27] | Compromised by gasdermin pores [28] | Lost [27] | Typically lost due to combined pore formation and membrane disruption |
| Inflammation | Minimal or anti-inflammatory [27] | Strongly inflammatory [28] | Inflammatory [67] | Highly inflammatory; enhances immunogenic cell death [67] |
| Key Executors | Caspase-3/7, CAD/DFF40 [27] | Gasdermin family proteins, caspase-1 [28] | RIPK3, MLKL [67] | Multiple executors simultaneously active [67] |
| DNA Damage | Ordered fragmentation (DNA ladder) [27] | Random fragmentation, TUNEL-positive [28] | Not well characterized | Complex pattern reflecting multiple cleavage activities |
| Caspase Involvement | Caspase-8/9 (initiators), caspase-3/6/7 (effectors) [69] [27] | Caspase-1/4/5/11 (inflammatory), caspase-3/8 (alternative) [28] | Caspase-independent [67] | Multiple caspases simultaneously (e.g., caspase-1/3/6/8) [67] |
How is PANoptosis experimentally detected and confirmed?
Confirming PANoptosis requires a multiparameter approach that simultaneously assesses markers for all three death pathways. No single assay can definitively identify PANoptosis; instead, researchers must combine multiple techniques to build compelling evidence [70].
Essential detection methodologies include:
- Western blotting for simultaneous detection of key effectors: cleaved caspase-3 (apoptosis), phosphorylated MLKL (necroptosis), and cleaved gasdermin D or E (pyroptosis) [67] [28]
- Cell viability assays with inhibitors specific to each pathway: Z-VAD-FMK (pan-caspase inhibitor for apoptosis), necrostatin-1 (RIPK1 inhibitor for necroptosis), and disulfiram or specific gasdermin inhibitors for pyroptosis [67]
- Morphological analysis using live-cell imaging to observe combined characteristics like swelling with apoptotic body formation [67]
- Flow cytometry with Annexin V/PI staining, though note that Annexin V cannot differentiate apoptotic from pyroptotic cells [28]
- ELISA for inflammatory cytokines IL-1β and IL-18, which are released during pyroptotic component activation [67] [28]
PANoptosome Complexes: Identification and Characterization
What are the major types of PANoptosomes and their key components?
Table 2: Major PANoptosome Complexes and Their Components
| PANoptosome Type | Key Sensors | Adapters | Effectors | Primary Stimuli/Context |
|---|---|---|---|---|
| ZBP1-PANoptosome | ZBP1, NLRP3 [67] | ASC, FADD [67] | Caspase-1/6/8, RIPK1/3, MLKL [67] | Influenza A virus infection, antiviral defense [67] [68] |
| AIM2-PANoptosome | AIM2, pyrin, ZBP1 [67] | ASC, FADD [67] | Caspase-1/8, RIPK1/3 [67] | Cytosolic dsDNA, HSV-1 infection, autoimmune disorders [67] |
| NLRP3-PANoptosome | NLRP3 [67] | ASC, RIPK3 [67] | Caspase-1/8 complexes [67] | Diverse stimuli (ATP, crystals, mitochondrial ROS) [67] |
| RIPK1-PANoptosome | RIPK1 [67] | NLRP3, ASC [67] | RIPK3, caspase-1/8 complexes [67] | TAK1 deficiency, TNF-α stimulation [67] |
| NLRP12-PANoptosome | NLRP12 [68] | Not specified | Not specified | Yersinia pestis infection [68] |
What is the experimental evidence for PANoptosome assembly?
The initial evidence for PANoptosome formation came from studies of influenza A virus (IAV) infection in macrophages. Kuriakose et al. demonstrated that IAV-infected macrophages undergo simultaneous pyroptosis, apoptosis, and necroptosis, characterized by concurrent activation of caspase-1/3/8 and MLKL phosphorylation [68]. This cell death could not be blocked by inhibitors targeting any single pathway, suggesting an integrated process.
Key experimental approaches for PANoptosome validation:
- Co-immunoprecipitation to demonstrate physical interactions between components from different death pathways [67]
- Genetic ablation studies showing that deletion of individual pathway components only partially reduces cell death [67]
- Confocal microscopy with fluorescently tagged proteins to visualize complex formation [67]
- Proteomic analysis of immunoprecipitated complexes to identify all associated components [67]
Troubleshooting Common Experimental Challenges
How can researchers distinguish PANoptosis from other death pathways when caspase activation is detected?
This challenge is particularly relevant in the context of distinguishing apoptotic vs. pyroptotic caspase activation. The key is recognizing that caspases can play multiple roles across different death pathways. For example, caspase-8 can participate in apoptosis, necroptosis regulation, and even pyroptosis under certain conditions [67] [28].
Troubleshooting guide for ambiguous caspase activation:
Problem: Caspase-3 activation is detected, but cell morphology suggests pyroptosis. Solution: Check for gasdermin cleavage (GSDMD or GSDME). Caspase-3 can cleave GSDME to induce pyroptosis rather than apoptosis [28].
Problem: Mixed morphological features make classification difficult. Solution: Use live-cell imaging with multiple fluorescent probes simultaneously (e.g., Annexin V, PI, caspase activity probes) to track death progression in real-time.
Problem: Pathway-specific inhibitors only partially reduce cell death. Solution: This is a key indicator of PANoptosis. Use combination inhibitor treatments (e.g., Z-VAD-FMK + necrostatin-1 + disulfiram) to assess if death can be fully blocked.
Problem: Inconsistent results between different detection methods. Solution: Ensure you're using multiple orthogonal techniques. For example, combine Western blotting for key effectors with functional assays using pathway-specific inhibitors.
What are the common pitfalls in PANoptosis research and how can they be avoided?
Critical considerations for experimental design:
- Temporal dynamics: PANoptosis involves sequential and parallel activation of multiple pathways. Single timepoint analyses may miss important transitions; include multiple timepoints in your experiments [67].
- Inhibitor specificity: Many small molecule inhibitors have off-target effects at higher concentrations. Use multiple inhibitors with different mechanisms and include genetic approaches (knockdown/knockout) where possible [67].
- Cell type variation: PANoptosis components show significant cell-type specific expression. For example, NLRP3 is primarily expressed in myeloid cells [67]. Validate findings in multiple relevant cell types.
- Stimulus optimization: PANoptosis is highly context-dependent. Titrate your stimuli carefully, as excessive stimulation may cause overwhelming necrosis that masks regulated death pathways [67].
Research Reagent Solutions
What essential tools and reagents are needed for PANoptosis research?
Table 3: Essential Research Reagents for PANoptosis Studies
| Reagent Category | Specific Examples | Function/Application | Key Considerations |
|---|---|---|---|
| Pathway Inhibitors | Z-VAD-FMK (pan-caspase), Necrostatin-1 (RIPK1), Disulfiram (GSDMD), GSK'872 (RIPK3) [67] | Functional dissection of pathway contributions | Use multiple concentrations; combine inhibitors to test for additivity; verify specificity |
| Antibodies | Anti-cleaved caspase-3, anti-pMLKL, anti-cleaved GSDMD, anti-cleaved GSDME [67] [28] | Detection of activated effectors by Western blot, IF, IHC | Validate specificity with knockout controls; check species reactivity |
| Cell Death Assays | Annexin V/PI kits, LDH release assays, real-time cell imaging systems [70] [28] | Quantification and characterization of cell death | Use multiple assays in parallel; establish baseline for each cell type |
| Cytokine Detection | IL-1β and IL-18 ELISA kits [67] [28] | Measurement of inflammatory output | Correlate with cell death markers; consider multiplex approaches |
| Genetic Tools | siRNA/shRNA libraries, CRISPR/Cas9 systems for key PANoptosis genes (ZBP1, AIM2, NLRP3, etc.) [67] | Functional validation of component roles | Use multiple targeting constructs; rescue with wild-type genes |
What emerging technologies are advancing PANoptosis research?
Recent technological advances are enabling more sophisticated analysis of PANoptosis:
- Machine learning approaches for identifying PANoptosis-related gene signatures and predicting therapeutic responses [71]
- Advanced imaging techniques including super-resolution microscopy to visualize PANoptosome assembly in real-time
- Multiplexed proteomics to comprehensively characterize PANoptosome composition under different conditions
- Single-cell RNA sequencing to understand heterogeneous cellular responses to PANoptotic stimuli [71]
- Nanomedicine approaches for targeted modulation of PANoptosis in specific tissues or cell types [67]
Advanced Experimental Protocols
Protocol: Comprehensive PANoptosis Detection and Validation
Objective: To definitively confirm PANoptosis activation in response to a specific stimulus.
Materials:
- Cells of interest (primary or cell line)
- Stimulus (pathogen, cytokine, chemical, etc.)
- Pathway-specific inhibitors: Z-VAD-FMK (20-50 μM), Necrostatin-1 (10-30 μM), Disulfiram (5-20 μM)
- Antibodies: cleaved caspase-3, cleaved caspase-8, phosphorylated MLKL (Ser358), cleaved GSDMD (Asp275), cleaved GSDME (Asp270)
- Cell culture reagents, lysis buffers, Western blot equipment
- Live-cell imaging system or flow cytometer with Annexin V/PI staining capability
- ELISA kits for IL-1β and IL-18
Procedure:
Stimulus Optimization:
- Seed cells in appropriate culture vessels and allow to adhere overnight
- Titrate stimulus concentration and time course to establish submaximal death conditions (aim for 40-60% death at maximum timepoint)
- Include untreated controls and positive controls for individual death pathways if available
Inhibitor Treatment:
- Pre-treat cells with individual inhibitors or combinations 1-2 hours before stimulus application
- Maintain inhibitors in media throughout stimulus exposure
- Include DMSO vehicle controls for all inhibitor conditions
Cell Death Assessment:
- Collect supernatant and cells at multiple timepoints (e.g., 6, 12, 24 hours)
- Analyze cell death by: a) Annexin V/PI flow cytometry b) LDH release assay c) Real-time cell imaging if available
- Note: PANoptosis typically shows only partial inhibition with single pathway blockers
Molecular Analysis:
- Lyse cells for Western blotting analysis of key effectors
- Probe for simultaneous activation of apoptotic (cleaved caspase-3/7), necroptotic (pMLKL), and pyroptotic (cleaved GSDMD/GSDME) markers
- Measure inflammatory cytokine release (IL-1β, IL-18) in supernatant by ELISA
Morphological Assessment:
- Fix cells for electron microscopy or perform live-cell imaging to document combined morphological features
- Look for simultaneous appearance of apoptotic bodies, pyroptotic bubble-like protrusions, and necroptotic swelling
Genetic Validation:
- If possible, confirm findings with genetic approaches (CRISPR knockout, siRNA) targeting key PANoptosis components like ZBP1 or AIM2
Interpretation: Strong evidence for PANoptosis includes: (1) simultaneous activation of effectors from multiple death pathways, (2) only partial protection with individual pathway inhibitors but near-complete protection with combination treatment, (3) inflammatory cytokine release, and (4) mixed morphological features that cannot be classified as a single death type.
Protocol: PANoptosome Complex Immunoprecipitation
Objective: To isolate and identify components of PANoptosome complexes.
Materials:
- Cells undergoing PANoptosis and appropriate controls
- Lysis buffer (e.g., RIPA buffer with protease and phosphatase inhibitors)
- Antibodies for immunoprecipitation (anti-ZBP1, anti-AIM2, anti-ASC, etc.)
- Protein A/G beads
- Western blot equipment and antibodies for potential complex components
Procedure:
- Induce PANoptosis in cells and harvest at optimal timepoint
- Lyse cells with gentle lysis buffer to preserve protein complexes
- Pre-clear lysate with protein A/G beads
- Incubate lysate with immunoprecipitation antibody or control IgG overnight at 4°C
- Add protein A/G beads and incubate 2-4 hours
- Wash beads extensively with lysis buffer
- Elute bound proteins with SDS sample buffer
- Analyze by Western blotting for suspected PANoptosome components
Expected Results: Successful PANoptosome immunoprecipitation should pull down components from multiple death pathways simultaneously, such as ZBP1 with RIPK3, caspase-8, and ASC in the case of ZBP1-PANoptosome.
Confirming the Pathway: Orthogonal Validation and Biomarker Correlation
Caspases are cysteine proteases that play a central role in executing programmed cell death. Your research into distinguishing apoptotic versus pyroptotic signaling hinges on identifying specific caspase activation patterns and their downstream substrates. Apoptosis, mediated by caspases-3, -6, and -7, is generally non-inflammatory and characterized by specific protein cleavage events, while pyroptosis, mediated by caspase-1 or caspase-11/4/5, is highly inflammatory and forms a key part of innate immunity [72] [73]. This guide will help you troubleshoot the experimental challenges in correctly identifying the active pathway in your models.
Troubleshooting Guides
FAQ: Determining Cell Death Type
Q1: My experiments show caspase activation and cell death, but I cannot determine if it's apoptotic or pyroptotic. What are the key differentiators I should measure?
A1: You should establish a multi-parameter approach to distinguish these pathways confidently. The table below summarizes the core characteristics to analyze.
Table 1: Key Characteristics of Apoptosis vs. Pyroptosis
| Characteristic | Apoptosis | Pyroptosis |
|---|---|---|
| Initiator Caspases | Caspase-8, -9, -10 [74] | Caspase-1, -4, -5, -11 [72] |
| Effector Caspases | Caspase-3, -6, -7 [72] [74] | (None; initiator also acts as effector) [72] |
| Key Downstream Substrates | PARP (Cleaved) [72], ICAD (Cleaved) [72] | pro-IL-1β (Cleaved), pro-IL-18 (Cleaved) [72] [75], GSDMD (Cleaved) |
| Cell Membrane Phenotype | Remains intact; forms apoptotic bodies [73] | Pore formation (1.1-2.4 nm); becomes permeable to dyes like PI [72] |
| Inflammatory Outcome | Non-inflammatory [24] [73] | Highly inflammatory [24] [73] |
| Nuclear Morphology | Nuclear fragmentation; high-intensity TUNEL stain [72] [76] | Chromatin condensation; intact nucleus; lower-intensity TUNEL stain [72] |
Troubleshooting Tips:
- Inconclusive Western Blots: If you only see cleaved caspase-3, it strongly suggests apoptosis. However, note that caspase-3 can also cleave the pyroptotic effector GSDME/DFNA5, potentially causing a secondary pyroptotic-like phenotype [77]. Always check multiple markers.
- Unexpected Inflammation in "Apoptotic" Model: If your cell death model is expected to be apoptotic but you detect significant inflammation, check for GSDME cleavage via immunoblotting and measure LDH release, a marker of lytic cell death [78] [77].
Q2: I've detected PARP cleavage in my model, does this automatically confirm apoptosis?
A2: No, this is a critical and common misconception. PARP cleavage is a hallmark of apoptosis, executed by effector caspases like caspase-3 [72]. However, during pyroptosis, PARP is not cleaved [72]. Therefore, the presence of cleaved PARP is a strong indicator of apoptotic activity, while its absence in a scenario of active cell death points towards pyroptosis or other non-apoptotic pathways. Be aware that PARP1 itself can be a trigger for cell death; PARP1 inhibitors have been shown to induce pyroptosis in BRCA-deficient cells via caspase-3-mediated GSDME cleavage [78].
Q3: I see mature IL-1β in my supernatant, but cannot detect active caspase-1. Is this possible?
A3: Yes, these events can be separable. Research shows that the activation of caspase-1 (and the subsequent processing of pro-IL-1β) can be distinct from the release of the mature cytokine. Using human α-defensin-5 (HD-5) as a tool, studies demonstrated that the release of IL-1β can be completely inhibited without affecting the intracellular processing of pro-IL-1β by caspase-1 [79]. This suggests that your detection method for active caspase-1 may need optimization, or that an alternative secretion pathway is involved.
FAQ: Technical Challenges
Q4: My flow cytometry data for Annexin V and PI is confusing when distinguishing late apoptosis from pyroptosis. What is the explanation?
A4: This confusion arises because both late apoptotic and pyroptotic cells will be Annexin V and PI positive, but for different biological reasons.
- In apoptosis, phosphatidylserine (PS) is externalized to the outer membrane leaflet early in the process, making cells Annexin V+/PI-. In late stages, the membrane becomes permeable, allowing PI to stain the cell [76].
- In pyroptosis, pore formation in the plasma membrane happens earlier, allowing PI to enter. The pores also expose the inner leaflet PS to the extracellular environment, where Annexin V can bind it, resulting in Annexin V+/PI+ staining [72].
Solution: You cannot rely on Annexin V/PI alone. You must incorporate additional assays, such as:
- Caspase-specific FLICA assays: Use FAM-VAD-FMK (pan-caspase) or more specific inhibitors to differentiate caspase-1 from caspase-3/7 activity via flow cytometry [76].
- LDH Release Assay: Quantify lactate dehydrogenase release as a robust measure of lytic cell death, which is characteristic of pyroptosis [78].
- Immunoblotting: Confirm the cleavage of pathway-specific substrates like PARP (apoptosis) or IL-1β/GSDMD (pyroptosis).
Q5: Why does my caspase inhibition not completely block cell death?
A5: Commitment to cell death can occur upstream of caspase activation. Even when caspases are inhibited, the cell death signal may still proceed through caspase-independent pathways.
- The Bcl-2 family proteins (e.g., Bax, Bak) can induce mitochondrial damage and cell death even in the absence of caspase activity [80].
- Other programmed necrosis pathways, such as necroptosis, can be triggered when caspases (particularly caspase-8) are inhibited [24]. Necroptosis is dependent on RIPK1, RIPK3, and MLKL.
Troubleshooting Tip: If a pan-caspase inhibitor like z-VAD-FMK does not block death, consider investigating necroptosis. You can use specific inhibitors like Necrostatin-1 (RIPK1 inhibitor) or GSK'872 (RIPK3 inhibitor) to test this possibility [24] [77].
Experimental Protocols & Workflows
Protocol 1: Differentiating Apoptosis and Pyroptosis via Immunoblotting
This protocol allows you to simultaneously analyze key markers from a single sample.
Workflow Diagram:
Detailed Steps:
- Cell Lysis: Lyse 1-2 million cells in 100-200 µL of RIPA lysis buffer supplemented with a protease inhibitor cocktail. Incubate on ice for 15-30 minutes, then centrifuge at >12,000 x g for 15 minutes at 4°C to pellet debris. Collect the supernatant [78].
- Protein Quantification: Use the BCA assay per manufacturer's instructions to determine the protein concentration of your lysates. Dilute all samples to the same concentration using lysis buffer [78].
- Sample Preparation: Mix equal volumes of protein lysate with 2X reducing Laemmli buffer (containing β-mercaptoethanol). Heat the samples at 95-100°C for 5-10 minutes to denature proteins [78].
- Gel Electrophoresis and Western Blotting: Load equal amounts of protein (e.g., 20-30 µg) onto an SDS-PAGE gel. After separation, transfer proteins to a PVDF or nitrocellulose membrane. Block the membrane with 5% non-fat milk or BSA in TBST for 1 hour at room temperature.
- Antibody Probing: Incubate with primary antibodies diluted in blocking buffer overnight at 4°C. The next day, wash the membrane and incubate with an appropriate HRP-conjugated secondary antibody. Detect signals using an enhanced chemiluminescence (ECL) substrate and imaging system [78].
Protocol 2: Multiparameter Flow Cytometry for Cell Death
This protocol enables single-cell analysis of multiple death parameters simultaneously.
Workflow Diagram:
Detailed Steps:
- Cell Preparation: Harvest both adherent and non-adherent cells to avoid selection bias. Wash the cells once with 1X PBS [76].
- Caspase Staining (FLICA): Resuspend the cell pellet in 100 µL of PBS. Add 3 µL of the FLICA working solution. Incubate for 60 minutes at 37°C in the dark, gently agitating the cells every 20 minutes. After incubation, wash the cells by adding 2 mL of PBS and centrifuging. Repeat this wash step one more time [76].
- Membrane Integrity Staining: After the final wash, resuspend the cell pellet in 100 µL of Annexin V Binding Buffer. Add Annexin V-APC and Propidium Iodide (PI) according to the manufacturer's recommended concentrations. Incubate for 15 minutes at room temperature in the dark [76].
- Analysis: Add 400 µL of Annexin V Binding Buffer to the tubes and analyze the samples immediately on a flow cytometer. Use 488 nm laser for FLICA and PI, and 640 nm laser for Annexin V-APC.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Differentiating Cell Death Pathways
| Reagent / Assay | Primary Function | Key Application & Interpretation |
|---|---|---|
| z-VAD-FMK (Pan-caspase inhibitor) | Irreversibly inhibits a broad range of caspases. | Used to confirm caspase-dependence of cell death. If death persists, suggests caspase-independent pathways (e.g., necroptosis) [80]. |
| Ac-YVAD-CHO (Caspase-1 inhibitor) | Selectively inhibits caspase-1 activity. | Confirms the role of caspase-1 in pyroptosis. Should block IL-1β/IL-18 maturation and GSDMD cleavage [73]. |
| DEVD-FMK (Caspase-3 inhibitor) | Selectively inhibits caspase-3/7 activity. | Confirms the role of caspase-3 in apoptosis. Should block PARP cleavage and other apoptotic hallmarks. |
| FLICA Assay Kits (e.g., FAM-VAD-FMK) | Fluorescently labels active caspases in live cells. | Flow cytometry-based detection of caspase activity. FAM-VAD-FMK is pan-caspase; specific probes exist for caspase-1 (FLICA-1) or caspase-3/7 (DEVD-FLICA) [76]. |
| LDH Release Assay | Measures lactate dehydrogenase enzyme released upon membrane rupture. | Quantifies lytic cell death (pyroptosis, oncosis, secondary necrosis). Correlate with caspase activity to define the pathway [78]. |
| Annexin V / PI Staining Kit | Detects phosphatidylserine exposure (Annexin V) and membrane integrity (PI). | Distinguishes early apoptotic (Annexin V+/PI-) from late apoptotic/pyroptotic (Annexin V+/PI+) and necrotic (Annexin V-/PI+) cells [72] [76]. |
| Antibody: Cleaved PARP | Detects the signature 89 kDa fragment of PARP generated by caspase-3/7. | A positive result is a strong indicator of apoptotic execution [72]. |
| Antibody: Cleaved IL-1β (p17) | Detects the mature, active 17 kDa form of IL-1β generated by caspase-1. | A positive result is a definitive marker of inflammasome and caspase-1 activation [79] [75]. |
| Antibody: Cleaved GSDMD | Detects the active N-terminal pore-forming fragment of GSDMD. | A positive result is a direct marker of pyroptotic execution downstream of caspase-1/4/5/11 [24]. |
| Necrostatin-1 (Nec-1) | Inhibits RIPK1 kinase activity. | Used to confirm or rule out necroptosis, especially when cell death is caspase-independent [24]. |
Frequently Asked Questions (FAQs)
Q1: My CRISPR knockout in THP-1 cells has failed; what are the most critical steps to optimize? A1: For hard-to-transfect immune cells like THP-1, the delivery method is paramount. Traditional transfection methods often fail. An optimized protocol uses lentiviral delivery of the CRISPR-Cas9 system to achieve stable gene delivery with high efficiency. Furthermore, ensure proper validation through a combination of colony PCR, sequencing, and western blotting to confirm successful knockout at the DNA, RNA, and protein levels [81].
Q2: After generating a caspase knockout, how can I determine if the cell death pathway has switched from pyroptosis to apoptosis? A2: This is a key consideration in pathway validation. Research shows that impaired canonical signaling can divert cell death. For example, in caspase-1-deficient macrophages, the NLRP3 inflammasome platform can recruit caspase-8 instead, leading to apoptotic caspase activation (caspase-8/-9/-3) and GSDME-mediated secondary necrosis instead of pyroptosis. To confirm this switch, you should monitor [51] [54]:
- Key Effectors: Assess the activation (cleavage) of apoptotic caspases (e.g., caspase-3, -8) versus pyroptotic caspases (e.g., caspase-1).
- Gasdermin Cleavage: Determine which gasdermin protein is cleaved (e.g., GSDMD for pyroptosis, GSDME for apoptosis).
- Morphological Features: Use imaging techniques like scanning electron microscopy to identify mixed morphological features of cell death in the same field of view [50].
Q3: What is the most comprehensive way to validate my CRISPR knockout cell line? A3: Relying on a single method is insufficient. A multi-layered validation strategy is considered best practice [81] [82]:
- Genomic Level: Use Sanger sequencing or next-generation sequencing to verify the edit at the target locus and check for potential off-target effects.
- Protein Level: Perform western blotting or mass spectrometry to confirm the absence of the target protein. This is crucial for confirming a functional knockout.
- Functional Level: Employ immunohistochemistry, immunocytochemistry, or FACS to assess the functional consequence of the knockout, such as the loss of a specific marker or a change in cell surface proteins [82].
Troubleshooting Guides
Common CRISPR Workflow Challenges
The table below outlines frequent pitfalls in generating CRISPR knockout cell lines and their solutions.
Table 1: Troubleshooting Common Issues in CRISPR Knockout Generation
| Workflow Step | Common Challenge | Potential Solution |
|---|---|---|
| Guide Design & Production | Low on-target efficiency or high off-target effects [82] | Use multiple sgRNAs to increase desired editing. Consider synthetic sgRNAs to reduce off-target effects compared to plasmid systems [82]. |
| Transfection | Low efficiency, especially in hard-to-transfect cells (e.g., THP-1, primary immune cells) [81] [82] | Switch to lentiviral transduction, which offers higher efficiency for suspension and immune cell lines [81]. |
| Enrichment & Isolation | Low cell viability during single-cell cloning; time-consuming process [82] | Fully optimize growth conditions and use enrichment strategies to reduce the number of passages needed. Screen multiple clones to ensure homogeneity [82]. |
| Validation & Confirmation | Incomplete knockout or unintended edits missed [82] | Implement a multi-pronged validation approach: DNA sequencing to confirm edits and western blotting to verify protein ablation [81] [82]. |
Interpreting Cell Death Pathway Outcomes
When studying caspase knockouts, a key challenge is interpreting the resulting cell death phenotype. The following table summarizes key markers to distinguish between apoptosis and pyroptosis in your models.
Table 2: Key Markers to Distinguish Apoptosis and Pyroptosis
| Feature | Apoptosis | Pyroptosis |
|---|---|---|
| Primary Initiator Caspases | Caspase-8, -9, -10 [3] | Caspase-1, -4, -5, -11 [3] |
| Primary Executioner Caspases | Caspase-3, -6, -7 [3] | Caspase-1 [3] |
| Key Gasdermin Protein | GSDME (cleaved by caspase-3) [3] | GSDMD (cleaved by caspase-1, -4, -5, -11) [3] |
| Key Morphological & Biochemical Hallmarks | Cell shrinkage, membrane blebbing, nuclear fragmentation, PARP cleavage [3] | Cell swelling, plasma membrane pore formation, release of IL-1β and IL-18, LDH release [3] |
| Inflammatory Response | Generally non-inflammatory [3] | Highly inflammatory [3] |
Experimental Protocols
Detailed Protocol: Single-Gene Knockout in THP-1 Cells
This protocol is optimized for hard-to-transfect suspension immune cell lines like THP-1, as used in pyroptosis research [81].
1. sgRNA Design and Cloning:
- Design specific single-guide RNAs (sgRNAs) targeting your gene of interest (e.g., GSDMD or a caspase gene).
- Clone the sgRNA into a CRISPR vector, such as a lentiviral plasmid containing the Cas9 nuclease.
2. Viral Packaging and Transduction:
- Package the constructed lentiviral vector into viral particles using a suitable packaging cell line (e.g., HEK293T).
- Transduce the target THP-1 cells with the packaged lentivirus. Lentiviral delivery is critical for achieving high efficiency in these cells.
3. Selection and Expansion:
- After transduction, select successfully transduced cells using an appropriate selection agent (e.g., puromycin).
- Expand the selected cell population and perform single-cell cloning to isolate homogeneous knockout clones.
4. Validation:
- Genomic DNA Level: Perform colony PCR and Sanger sequencing on the target locus to confirm the presence of indels (insertions or deletions).
- Protein Level: Use western blotting to confirm the absence of the target protein, which is essential for validating a successful knockout [81].
Protocol: Rapidly Screening CRISPR Editing Outcomes
This protocol uses a fluorescent reporter system for high-throughput assessment of gene editing efficiency [83].
1. Generate eGFP-Positive Cells:
- Create a stable cell line expressing enhanced Green Fluorescent Protein (eGFP) via lentiviral transduction.
2. Transfection with Editing Reagents:
- Transfect the eGFP-positive cells with your CRISPR-Cas9 reagents designed to target and disrupt the eGFP gene.
3. Analysis via FACS:
- After transfection, measure cell fluorescence using Fluorescence-Activated Cell Sorting (FACS).
- Successful non-homologous end joining (NHEJ)-mediated knockout of eGFP will result in a loss of green fluorescence and, depending on the edit, may result in a shift to a Blue Fluorescent Protein (BFP) phenotype or non-fluorescence. This provides a rapid, quantifiable readout of editing efficiency [83].
Signaling Pathways and Experimental Workflows
Caspase Interconnectivity in Cell Death Pathways
This diagram illustrates the complex roles of different caspases as master regulators bridging apoptosis, pyroptosis, and necroptosis, which is central to interpreting knockout model phenotypes.
CRISPR-Cas9 Knockout Workflow for Immune Cells
This workflow visualizes the key steps for generating and validating a knockout in hard-to-transfect immune cells, highlighting critical validation points.
Research Reagent Solutions
The following table lists essential materials and reagents used in the featured experiments for generating and analyzing CRISPR knockouts in cell death pathway research.
Table 3: Essential Research Reagents for CRISPR-based Cell Death Studies
| Item | Function/Application | Example from Protocols |
|---|---|---|
| Lentiviral CRISPR Vector | Delivery of Cas9 and sgRNA into hard-to-transfect cells [81]. | Used for generating GSDMD knockout in THP-1 cells [81]. |
| THP-1 Cell Line | Human monocytic cell line; common model for immunology and cell death studies [81] [54]. | Used in protocol for single-gene knockout [81]. |
| Caspase-1 Inhibitor (VX-765) | Pharmacological inhibitor used to probe caspase-1 function; can have pan-caspase effects at higher doses [51] [54]. | Used to study diversion from pyroptosis to apoptosis [51] [54]. |
| Antibody: Cleaved Caspase-3 | Detects activation of the key executioner caspase in apoptosis [51] [54]. | Used in western blotting to confirm apoptotic activation [51] [54]. |
| Antibody: GSDMD | Detects expression and cleavage (into GSDMD-NT) of the pyroptosis executioner protein [51] [54]. | Used in western blotting to confirm pyroptotic pathway activation [51] [54]. |
| NLRP3 Inflammasome Activator (Nigericin) | Potent activator of the NLRP3 inflammasome, used to induce pyroptosis in primed macrophages [51] [54]. | Used to stimulate inflammasome formation in macrophage models [51] [54]. |
| Flow Cytometer | Instrument for analyzing and sorting cells based on fluorescence; used for validating knockout efficiency and assessing cell death [82] [83]. | Used in the rapid eGFP-to-BFP screening protocol [83]. |
Within caspase activation research, distinguishing between the nuanced pathways of apoptosis and pyroptosis is a common experimental challenge. Apoptosis is a immunologically silent, non-inflammatory form of programmed cell death, whereas pyroptosis is a lytic, pro-inflammatory process [84] [85]. This technical support center provides targeted guidance for researchers assembling biomarker panels that integrate Lactate Dehydrogenase (LDH) release, cytokine profiling, and gasdermin fragment detection to accurately differentiate these pathways. The core of this approach lies in interpreting the unique biomarker signatures each death modality leaves behind. By combining multiple assays, you can overcome the limitations of single-parameter tests and generate robust, publishable data on caspase functions in cell death.
Core Biomarker Profiles: Apoptosis vs. Pyroptosis
The table below summarizes the key differential biomarkers used to distinguish apoptotic and pyroptotic caspase activation.
Table 1: Key Biomarkers for Differentiating Apoptosis and Pyroptosis
| Biomarker Category | Specific Marker | Apoptosis Signature | Pyroptosis Signature | Primary Function & Significance |
|---|---|---|---|---|
| Cell Membrane Integrity | LDH Release | Low/Moderate (late stage) | High & Rapid [86] | Cytoplasmic enzyme; key indicator of plasma membrane rupture. |
| Propidium Iodide (PI) Staining | PI-negative (early), PI-positive (late) | Rapidly PI-positive [87] | DNA dye; stains cells after membrane pore formation/rupture. | |
| Inflammatory Cytokines | IL-1β & IL-18 | Not Released | Significantly Released [84] [88] | Mature forms released through GSDMD pores; indicate inflammatory caspase activity. |
| Gasdermin Protein Cleavage | GSDMD-NT fragment | Typically absent or inactivated | Present (Key Executor) [28] [85] | Pore-forming fragment; definitive marker for pyroptosis execution. |
| GSDME-NT fragment | Can be present (caspase-3 mediated) | Can be present (caspase-3 mediated) [28] | Indicates caspase-3-mediated switch from apoptosis to secondary pyroptosis. | |
| Effector Caspase Activity | Cleaved Caspase-3 | Present (Executioner) | Typically absent | Primary executioner caspase in apoptosis. |
| Cleaved Caspase-1 | Absent | Present (Inflammatory) [84] | Inflammatory caspase; cleaves GSDMD and pro-IL-1β/18 in canonical pyroptosis. |
Essential Research Reagent Solutions
The following table catalogs critical reagents required for experiments focused on distinguishing cell death mechanisms.
Table 2: Essential Research Reagents for Apoptosis and Pyroptosis Studies
| Reagent / Kit Type | Specific Examples | Critical Function in Experiments |
|---|---|---|
| LDH Detection Kits | Colorimetric, Fluorometric, Bioluminescent Kits [86] | Quantify cell membrane integrity and lytic cell death by measuring released LDH enzyme. |
| Cytokine ELISA/Kits | IL-1β, IL-18 ELISA Kits [88] | Detect and quantify mature, released inflammatory cytokines to confirm pyroptotic inflammation. |
| Gasdermin Antibodies | Anti-GSDMD (full length & N-terminal), Anti-GSDME [84] [89] | Detect full-length and cleaved, active gasdermin fragments via Western blot; key for identifying the pyroptosis executor. |
| Caspase Antibodies & Assays | Anti-Caspase-1, Cleaved Caspase-3, Caspase Activity Assays | Differentiate between inflammatory (caspase-1) and apoptotic (caspase-3) caspase activation. |
| Cell Death Stains | Propidium Iodide (PI), Annexin V [90] [87] | Assess membrane integrity (PI) and phosphatidylserine exposure (Annexin V) via flow cytometry or microscopy. |
| Inflammasome Activators | NLRP3 Agonists (Nigericin, ATP), LPS [84] [85] | Positive control stimuli to induce canonical (NLRP3) and non-canonical (LPS) pyroptosis pathways. |
| Selective Inhibitors | Caspase-1 Inhibitors (YVAD), Caspase-3/8 Inhibitors (Z-VAD), NLRP3 Inhibitors (CY-09) [21] | Tool compounds to chemically genetically dissect specific contributions of caspases and inflammasomes to cell death. |
Detailed Experimental Protocols
LDH Release Assay for quantifying Lytic Cell Death
Principle: This assay measures the activity of the cytosolic enzyme LDH released into the culture supernatant upon plasma membrane rupture, a hallmark of pyroptosis and other lytic cell death types [86].
Materials:
- LDH assay kit (typically containing assay buffer, substrate mix, NAD+, and lysis solution)
- 96-well flat-bottom microplate
- Microplate reader (spectrophotometer, fluorometer, or luminometer)
- Centrifuge with plate adaptors
Step-by-Step Protocol (Colorimetric):
- Cell Seeding and Treatment: Seed cells in a 96-well plate at a density of 1-5 x 10⁴ cells/well in 100 μL complete medium. Incubate overnight. Apply your experimental treatments, ensuring to include essential controls in triplicate [86].
- Prepare Controls:
- Spontaneous LDH Control: Untreated cells.
- Maximum LDH Control: Cells treated with lysis solution (e.g., 1% Triton X-100) at the end of the incubation to lyse all cells.
- Culture Medium Background: Medium without cells.
- Supernatant Collection: After treatment incubation, centrifuge the plate at 1,500-2,000 rpm for 5 minutes. Carefully transfer 50 μL of supernatant from each well to a new clear 96-well assay plate.
- Reaction Setup: Add the prepared reaction mixture from the kit (e.g., containing INT, lactate, NAD+) to each supernatant sample. Incubate in the dark for 15-30 minutes at room temperature.
- Signal Measurement: Add the stop solution (e.g., 1M acetic acid). Measure the absorbance at 490 nm (for formazan dye) using a microplate reader.
- Data Calculation:
- Subtract the background signal (medium-only control) from all values.
- Calculate cytotoxicity using the formula:
% Cytotoxicity = (Experimental LDH - Spontaneous LDH) / (Maximum LDH - Spontaneous LDH) × 100
Troubleshooting:
- High Background in Medium Control: Fetal Bovine Serum (FBS) contains LDH. Use a low-serum or serum-free medium during the treatment and assay period, or ensure this background is accurately subtracted.
- Low Signal Intensity: Extend the reaction incubation time. Verify that the maximum LDH control gives a strong signal, confirming the assay is working.
Immunoblotting for Gasdermin Activation
Principle: This method detects the cleavage of gasdermin proteins (e.g., GSDMD), which is a definitive molecular event in pyroptosis. The cleavage generates an N-terminal fragment (GSDMD-NT) that forms plasma membrane pores [28] [89] [85].
Materials:
- RIPA lysis buffer with protease inhibitors
- SDS-PAGE gel system and Western blot transfer apparatus
- Antibodies: Anti-GSDMD (full-length and N-terminal specific), Anti-Caspase-1, Anti-Caspase-3, and corresponding loading control antibodies (e.g., GAPDH, β-Actin)
Step-by-Step Protocol:
- Cell Lysis and Protein Extraction: Lyse treated and control cells on ice using RIPA buffer. Centrifuge at 12,000 rpm for 15 minutes at 4°C to pellet debris. Collect the supernatant (whole cell lysate).
- Protein Quantification: Determine protein concentration using a BCA or Bradford assay.
- Gel Electrophoresis and Transfer: Dilute protein lysates in Laemmli buffer, denature at 95-100°C for 5-10 minutes. Load equal amounts of protein (e.g., 20-30 μg) onto an SDS-PAGE gel (10-15% gradient gels are suitable for detecting gasdermin fragments). Run the gel and transfer proteins to a PVDF membrane.
- Antibody Incubation:
- Blocking: Block the membrane with 5% non-fat milk or BSA in TBST for 1 hour.
- Primary Antibody: Incubate with primary antibodies (e.g., anti-GSDMD, ~53-55 kDa for FL, ~31 kDa for NT; anti-Caspase-1) diluted in blocking buffer overnight at 4°C.
- Secondary Antibody: Wash membrane and incubate with HRP-conjugated secondary antibody for 1 hour at room temperature.
- Detection: Use enhanced chemiluminescence (ECL) substrate and image the membrane with a chemiluminescence imager.
Troubleshooting:
- No GSDMD Cleavage Signal: Confirm the efficiency of your pyroptosis induction stimulus (e.g., check with a known NLRP3 inflammasome activator like nigericin). Ensure your antibody is specific for the N-terminal fragment. Overexpose the blot if necessary, as the cleaved fragment may be less abundant.
- High Background: Increase the number and duration of washes with TBST after antibody incubations. Optimize the concentration of the primary antibody.
Signaling Pathways & Experimental Workflows
Caspase Activation and Biomarker Signaling Pathways
Caspase Activation Pathways and Biomarker Outputs: This diagram illustrates the distinct signaling pathways in apoptosis and pyroptosis, highlighting the specific caspases involved and the unique biomarkers generated at key steps, which form the basis of the differential assay panel.
Integrated Experimental Workflow for Cell Death Discrimination
Integrated Workflow for Cell Death Discrimination: This workflow outlines the parallel execution of key assays on samples from the same experiment, leading to a consolidated data analysis that allows for confident classification of the cell death modality, including complex scenarios like PANoptosis.
Frequently Asked Questions (FAQs) & Troubleshooting
Q1: My LDH assay shows high cytotoxicity, but I cannot detect GSDMD cleavage by Western blot. What could explain this discrepancy? A: This is a common puzzle with several possible explanations:
- Alternative Lytic Death: The cell death might be driven by another lytic process, such as necroptosis (MLKL pores) or oncosis [90]. Perform additional assays, like using the necroptosis inhibitor Necrostatin-1 or staining for MLKL oligomerization.
- Non-canonical Pyroptosis: The pyroptosis could be mediated by other gasdermin family members (e.g., GSDME or GSDMB) that are cleaved by different enzymes (e.g., caspase-3 or granzymes) [28] [88]. Probe your Western blots for other cleaved gasdermins.
- Sensitivity Issues: The antibody for GSDMD-NT might have low affinity, or the cleaved fragment could be unstable or present in low abundance. Try a different, validated antibody and overexpose your blot.
Q2: I detect significant IL-1β release in my model, but other pyroptosis markers are weak. Is this sufficient to conclude pyroptosis is occurring? A: No, IL-1β release alone is not definitive proof of pyroptosis. While GSDMD pores are a major release mechanism for IL-1β, alternative pathways exist [85]. It is crucial to confirm pyroptosis by demonstrating:
- Gasdermin Pore Formation: Directly detect GSDMD-NT or other active gasdermin fragments via Western blot.
- Lytic Cell Death: Corroborate with an LDH release assay showing significant membrane rupture.
- Caspase-1 Activation: Show cleavage/activation of caspase-1. Without these confirmatory steps, IL-1β release could be from other secretory mechanisms or from cells undergoing other forms of inflammatory death.
Q3: My data shows mixed features of both apoptosis (cleaved caspase-3) and pyroptosis (GSDMD cleavage). Is this possible, or is my experiment contaminated? A: This is not only possible but an area of cutting-edge research. The simultaneous activation of multiple cell death pathways is now recognized as PANoptosis [21]. This is a coordinated inflammatory cell death process regulated by complex PANoptosomes. A mixed biomarker profile in your data may be a valid biological finding, not a technical artifact. You can investigate this further by using specific caspase inhibitors (e.g., Z-VAD for pan-caspase, VX-765 for caspase-1) to dissect the contribution of each pathway.
Q4: What are the critical controls for a rigorous LDH release assay? A: Always include these triplicate controls to ensure accurate interpretation [86]:
- Spontaneous LDH Control: Untreated cells. Represents the baseline LDH release from healthy cells.
- Maximum LDH Control: Cells treated with a lysis solution (e.g., 1-2% Triton X-100) at the end of the experiment. Represents the total LDH content and is used to calculate 100% release.
- Culture Medium Background Control: Medium alone, without cells. Accounts for any LDH activity present in the serum or medium components.
- Treatment Control: Your treatment compounds/drugs in medium without cells. Rules out any interference of the treatment with the assay chemistry.
Frequently Asked Questions (FAQs)
FAQ 1: Why is there a fundamental difference in how immune and non-immune cells respond to the same cell death stimulus? The difference arises from the divergent biological roles these cells play. Innate immune cells, such as macrophages, are professional first responders to infection and damage. Consequently, they express higher basal levels of key cell death proteins, including caspase-1, gasdermins, caspase-8, and RIPKs, equipping them to mount robust and rapid lytic cell death responses like pyroptosis and PANoptosis. In contrast, non-immune cells like fibroblasts have a lower innate expression of these molecules, leading to weaker, delayed, or altogether different death outcomes to the same trigger [58].
FAQ 2: My immunoblot shows cleaved caspase-3 in my cells. Does this definitively indicate apoptosis is occurring? Not necessarily. While cleaved caspase-3 is a classic executioner caspase for apoptosis, it can also contribute to other cell death pathways. For instance, active caspase-3 can cleave gasdermin E (GSDME), switching the cell death mode from non-lytic apoptosis to lytic pyroptosis [3]. To confirm apoptosis, you should assay for multiple hallmarks, such as the absence of plasma membrane rupture (e.g., by measuring LDH release) and the presence of other apoptotic markers like PARP cleavage, alongside caspase-3 activity [3] [24].
FAQ 3: When stimulating cells with LPS, how can I tell if the resulting cell death is pyroptosis versus necroptosis? Distinguishing between these lytic pathways requires examining specific molecular signatures. The table below outlines the key differentiators.
Table: Key Differentiators between Pyroptosis and Necroptosis
| Feature | Pyroptosis | Necroptosis |
|---|---|---|
| Key Executor | Gasdermin D (GSDMD) N-terminal pores [3] | Phosphorylated MLKL forming membrane pores/disruptions [24] |
| Key Initiating Caspase | Caspase-1 (canonical) or Caspase-4/5/11 (non-canonical) [3] | Caspase-8 inhibition [24] |
| Critical Kinase | Not typically required | RIPK1 and RIPK3 (forms necrosome) [24] |
| Key Readout | GSDMD cleavage (immunoblot); IL-1β maturation (ELISA) [91] | Phospho-MLKL (immunoblot); RIPK3 autophosphorylation [24] |
FAQ 4: Why might my positive control for caspase activation work in immune cells but fail in my non-immune cell line? This is a common issue rooted in the cell-type-specific expression of cell death machinery. Your chosen stimulus (e.g., a specific inflammasome activator) might rely on a sensor or adaptor protein that is poorly expressed or absent in your non-immune cells [58]. Always check the literature for the expression of the required pathway components (e.g., ZBP1, AIM2, specific NLRs) in your cell type of choice. You may need to use a different, more appropriate positive control stimulus for your non-immune cells.
Troubleshooting Guides
Issue 1: Lack of Expected Cell Death in Non-Immune Cells
Problem: After applying a canonical cell death stimulus (e.g., TNF-α + z-VAD for necroptosis), your immune cells (e.g., BMDMs) die robustly, but your non-immune cells (e.g., fibroblasts) show little to no death.
Solutions:
- Confirm Protein Expression: Use immunoblotting to verify that the non-immune cells express the necessary executioner proteins (e.g., RIPK3 and MLKL for necroptosis; GSDMD and inflammatory caspases for pyroptosis). It is common for non-immune cells to have low or absent expression of key molecules like RIPK3 [58].
- Titrate the Stimulus: Non-immune cells may require a higher concentration or longer duration of the stimulus to initiate death. Perform a dose-response and time-course experiment.
- Prime the Cells: Some cell types require a priming signal (e.g., low-dose IFN-γ or TNF-α) to upregulate components of the cell death machinery before they can respond to the cytotoxic trigger [58].
- Use a Redundant Stimulus: If one pathway is absent, target another. For example, if a fibroblast line is resistant to TNF-α-induced death, try a chemotherapeutic agent like etoposide to engage the intrinsic apoptosis pathway [92].
Issue 2: High Background or Non-Specific Staining in Immunohistochemistry (IHC) for Cell Death Markers
Problem: When staining tissue sections (e.g., from an infected mouse) for markers like cleaved caspase-3, you experience high background staining, making specific signal interpretation difficult.
Solutions:
- Optimize Blocking: Ensure adequate blocking is performed. Use 1-3% blocking agent (e.g., BSA or normal serum from the secondary antibody host) and increase the blocking time [93].
- Titer Antibodies: A high concentration of primary or secondary antibody is a common cause of background. Systematically titrate both antibodies to find the optimal concentration that provides a specific signal with minimal noise [94].
- Increase Washing Stringency: After primary and secondary antibody incubations, wash slides 3 times for 5 minutes with TBST or a similar buffer containing a mild detergent like Tween-20 [94].
- Include Controls: Always run a control slide stained without the primary antibody. This will reveal if the background is caused by non-specific binding of the secondary antibody [94].
Issue 3: Weak Fluorescent Signal in Flow Cytometry for Intracellular Antigens
Problem: When staining for intracellular proteins like active caspases or cytokines, the fluorescent signal is weak, and the positive population is indistinct from the negative control.
Solutions:
- Check Antibody Quality: Ensure antibodies have not degraded or expired. Aliquot and store them according to the manufacturer's instructions, protected from light [95].
- Titrate Antibodies: The antibody concentration may be too low for detection. Titrate the primary antibody to find the optimal amount [95].
- Optimize Permeabilization: The intracellular antigen may not be accessible. Ensure you are using an effective permeabilization buffer (e.g., saponin-based) and that the protocol has been optimized for your specific cell type [95].
- Use a Brighter Fluorochrome: For low-abundance antigens, pair them with bright fluorochromes like PE or APC to enhance the signal-to-noise ratio [95].
- Prevent Signal Loss: Keep cells at 4°C during staining and acquire data immediately after staining is complete to prevent fluorescence bleaching and epitope degradation [95].
Comparative Data Across Cell Types
The table below summarizes quantitative and qualitative differences in cell death responses between human and mouse immune versus non-immune cells, as identified in key studies.
Table: Comparative Cell Death Responses Across Cell Types
| Cell Type | Stimulus | Key Findings & Molecular Markers | Experimental Notes |
|---|---|---|---|
| Human Macrophages (Hu-mΦ) | Influenza A Virus (IAV) | Robust PANoptosis: Activation of caspase-1 (inflammasome), caspase-3/7 (apoptosis), & MLKL (necroptosis). High basal expression of death pathway components [58]. | Requires siRNA electroporation for gene knockdown (e.g., ZBP1, RIPK3) [58]. |
| Mouse Macrophages (BMDMs) | LPS + ATP (Pyroptosis) TNF-α + z-VAD (Necroptosis) | Robust Pyroptosis: Cleavage of caspase-1 and GSDMD. Robust Necroptosis: Phosphorylation of RIPK3 and MLKL [58]. | Canonical stimuli work effectively. Bone marrow cells differentiated with L929-conditioned media for 6 days [58]. |
| Normal Human Lung Fibroblasts (NHLF) | TNF-α + z-VAD | Weak/Resistant: Minimal cell death due to low expression of key necroptosis proteins (e.g., RIPK3) [58]. | Fibroblasts may require priming or alternative stimuli to induce death [58]. |
| Mouse Fibroblasts (L929) | TNF-α + z-VAD | Variable Response: Some lines are sensitive due to endogenous RIPK3 expression, but responses are generally less robust than in macrophages [58]. | Cell line-specific validation of protein expression is critical. |
| Human Leukemia (K562) | Etoposide (Apoptosis) | Delayed Apoptosis: Delayed cytochrome c release, caspase activation (DEVDase activity), and PARP cleavage compared to HL-60 cells [92]. | Requires higher doses (68 μM) and longer observation (24-48h) to see apoptotic changes [92]. |
Experimental Protocols
Protocol 1: Generating and Stimulating Bone Marrow-Derived Macrophages (BMDMs)
This protocol is essential for obtaining a primary, apoptosis-competent innate immune cell population from mice [58].
- Harvest Bone Marrow: Isolate bone marrow from the femurs and tibias of wild-type mice.
- Differentiate Macrophages: Culture primary bone marrow cells for 6 days in IMDM growth media supplemented with:
- 10% heat-inactivated fetal bovine serum (HI-FBS)
- 30% L929 conditioned media (as a source of M-CSF)
- 1% non-essential amino acids
- 1% penicillin/streptomycin
- Seed Cells: On day 6, harvest BMDMs and seed them at a density of 0.5-1.0 x 10^6 cells per well in appropriate plates.
- Stimulate: The next day, stimulate cells with canonical triggers:
- Pyroptosis: 100 ng/mL LPS for 3-4 hours, followed by 5 mM ATP for 30 minutes.
- Necroptosis: 50 ng/mL TNF-α combined with 25 μM pan-caspase inhibitor z-VAD for 4-8 hours.
- Apoptosis: 5 μM Staurosporine for 4-6 hours.
Protocol 2: Distinguishing Apoptosis from Pyroptosis via Multiparameter Assay
Use this combined assay workflow to dissect overlapping cell death pathways.
- Treat Cells: Apply your experimental stimulus to adherent or suspended cells.
- Harvest Supernatant and Lysate: Collect culture supernatant and cell lysate at predetermined time points.
- Run Parallel Assays:
- Membrane Integrity (Lytic Death): Use the supernatant to measure lactate dehydrogenase (LDH) release. High LDH indicates lytic death (pyroptosis/necroptosis) [58] [3].
- Caspase Activation: Use the cell lysate to measure caspase activity (e.g., fluorogenic substrates for caspase-1, -3/7, -8) or detect cleaved caspases and their substrates (like PARP) by immunoblotting [58] [92].
- Gasdermin Cleavage: Use immunoblotting on cell lysates to detect the cleavage of GSDMD or GSDME, a hallmark of pyroptosis [3].
- Interpret Combined Results:
- Apoptosis Signature: Caspase-3/7 activation + PARP cleavage + Low LDH release.
- Pyroptosis Signature: Caspase-1 (or caspase-4/5/11) activation + GSDMD cleavage + High LDH release.
Signaling Pathways and Experimental Workflows
Caspase-Mediated Cell Death Pathways
Experimental Workflow for Cell Death Analysis
The Scientist's Toolkit
Table: Key Research Reagents for Cell Death Studies
| Reagent / Material | Primary Function | Example Application |
|---|---|---|
| z-VAD(OMe)-FMK (z-VAD) | Broad-spectrum, cell-permeable caspase inhibitor. | Inhibiting apoptotic caspase activity to unmask or promote necroptosis (e.g., in TNF-α treatment) [58]. |
| Recombinant TNF-α | Potent cytokine that activates the extrinsic apoptosis pathway. | Inducing apoptosis or, when combined with z-VAD, triggering necroptosis in sensitive cells [58]. |
| LPS (Lipopolysaccharide) | TLR4 agonist and potent macrophage activator. | Priming for inflammasome activation or inducing non-canonical pyroptosis (with transfection) [91] [58]. |
| ATP | Endogenous DAMP and activator of the P2X7 receptor. | Triggering K+ efflux and NLRP3 inflammasome activation when added to LPS-primed cells [58]. |
| Staurosporine | Broad-spectrum protein kinase inhibitor. | Inducing intrinsic apoptosis across a wide range of cell types [58]. |
| SignalStain Boost IHC Detection Reagents | Polymer-based detection system for IHC. | Enhancing sensitivity and reducing background in immunohistochemistry staining of tissue sections [94]. |
| Fc Receptor Blockers (e.g., Normal Serum) | Block non-specific antibody binding. | Reducing high background in flow cytometry and IHC, especially in immune cells expressing Fc receptors [94] [95]. |
| Viability Dyes (e.g., PI, 7-AAD) | Stain DNA in cells with compromised membranes. | Distinguishing live cells from dead cells during flow cytometry analysis [95]. |
Frequently Asked Questions (FAQs)
Q1: What are the key functional outcomes that distinguish apoptotic from pyroptotic cell death? Apoptosis and pyroptosis lead to fundamentally different functional outcomes, primarily defined by the presence or absence of inflammation. The table below summarizes the core differences.
Table 1: Key Characteristics of Apoptosis vs. Pyroptosis
| Feature | Apoptosis | Pyroptosis |
|---|---|---|
| Primary Caspases Involved | Initiator: Caspase-8, -9; Executioner: Caspase-3, -6, -7 [3] | Inflammatory: Caspase-1, -4, -5, -11; Can involve Caspase-3 via GSDME [3] [96] |
| Key Molecular Executioner | Caspase-mediated substrate cleavage (e.g., PARP, lamins) [3] | Gasdermin protein (e.g., GSDMD, GSDME) cleavage and pore formation [3] [96] |
| Cell Membrane Integrity | Maintained (cell shrinks into apoptotic bodies) [3] | Compromised (due to gasdermin pores) [3] |
| Release of Cellular Content | No; contents are neatly packaged for phagocytosis [3] [97] | Yes; release of DAMPs, PAMPs, and inflammatory cytokines (IL-1β, IL-18) [3] |
| Phagocytic Clearance | Rapid, anti-inflammatory efferocytosis [98] [99] [100] | Typically lytic, but debris is still cleared by phagocytes [3] |
| Overall Immune Outcome | Anti-inflammatory, promotes immune tolerance and tissue homeostasis [99] [100] | Pro-inflammatory, amplifies immune responses and alerts the system to danger [3] |
Q2: My data shows simultaneous activation of caspase-8 and caspase-1. Is this possible, and what does it mean? Yes, this is a recognized complexity in cell death pathways. Caspase-8 acts as a central molecular switch that can participate in extrinsic apoptosis, inhibit necroptosis, and under certain conditions, also contribute to pyroptosis by cleaving gasdermin proteins like GSDMC [3]. Furthermore, research shows that when the canonical pyroptosis pathway (e.g., NLRP3-caspase-1) is impaired, the inflammasome platform can divert to recruit and activate caspase-8, leading to apoptotic signaling instead [51]. The specific cellular context, timing of signaling, and the status of other proteins (e.g., GSDMD) determine the ultimate cell fate.
Q3: I have observed significant cell death, but no release of classic inflammatory cytokines. Does this rule out pyroptosis? Not necessarily. While the release of IL-1β and IL-18 is a hallmark of canonical pyroptosis, cell lysis and the release of other DAMPs (Damage-Associated Molecular Patterns) like HMGB1 and LDH are also key indicators [3]. Furthermore, caspase-3-mediated cleavage of GSDME can switch an apoptotic signal to a pyroptotic outcome, which may not always involve the same cytokine profile as caspase-1-mediated pyroptosis [96]. You should assess multiple endpoints, including gasdermin cleavage, DAMP release, and membrane integrity.
Q4: Why is the clearance of apoptotic cells anti-inflammatory, while clearance of pyroptotic cells is not? The anti-inflammatory nature of apoptotic cell clearance ("efferocytosis") is an active process. Apoptotic cells display "eat-me" signals like phosphatidylserine (PS) on their surface. When phagocytes engage these signals, it actively suppresses the production of pro-inflammatory cytokines (e.g., IL-12) and induces the release of anti-inflammatory mediators like TGF-β and IL-10 [99] [100]. In contrast, pyroptotic cells release potent inflammatory mediators before and during their clearance, which dominates the immunological environment and drives inflammation [3].
Troubleshooting Guides
Problem: Differentiating Caspase-3-Mediated Apoptosis from Pyroptosis
Background: Caspase-3 is a key executioner caspase in apoptosis but can also contribute to pyroptosis by cleaving GSDME. Determining the primary cell death mode is crucial for data interpretation.
Potential Causes & Solutions:
Cause 1: Unrecognized GSDME activation.
- Solution: Perform western blotting to detect GSDME cleavage (appearance of GSDME-N terminal fragment) in addition to caspase-3 activation [96].
- Experimental Protocol:
- Cell Lysis: Lyse cells in RIPA buffer supplemented with protease inhibitors.
- Western Blot: Resolve proteins via SDS-PAGE and transfer to a PVDF membrane.
- Antibody Probing: Probe the membrane with antibodies against:
- Cleaved Caspase-3 (Asp175)
- GSDME / DFNA5 (Full length and N-terminal fragment)
- β-Actin (loading control).
- Interpretation: Cleaved caspase-3 with full-length GSDME suggests apoptosis. Cleaved caspase-3 plus the GSDME-N fragment indicates a shift towards GSDME-mediated pyroptosis [96].
Cause 2: Over-reliance on a single assay (e.g., TUNEL).
- Solution: Use a multi-parametric approach.
- Experimental Protocol:
- Membrane Integrity Assay: Use Propidium Iodide (PI) staining in live cells. Apoptotic cells exclude PI until late stages, while pyroptotic cells become PI-positive early due to membrane pores [3] [51].
- Morphology Assessment: Use live-cell imaging with high-content analysis. Look for apoptotic morphology (cell shrinkage, blebbing) versus pyroptotic morphology (cell swelling, plasma membrane ballooning) [101].
- * cytokine Release:* Measure extracellular LDH as a marker of lytic cell death and IL-1β via ELISA to specifically indicate inflammatory caspase activity [3].
Problem: Inconsistent or Weak Phagocytic Clearance of Apoptotic Cells In Vitro
Background: Efficient efferocytosis requires proper "find-me" and "eat-me" signals on apoptotic cells and functional receptors on phagocytes. Failures can skew experimental outcomes.
Potential Causes & Solutions:
Cause 1: Suboptimal induction of apoptosis.
- Solution: Standardize and validate your apoptosis induction method. Use a positive control (e.g., Staurosporine) and confirm apoptosis by Annexin V/PI flow cytometry and caspase activation western blot. Ensure PS exposure is occurring [99].
Cause 2: Inadequate "find-me" signals or phagocyte recruitment.
Cause 3: Poor recognition/engulfment due to reagent interference.
- Solution:
- Serum: Use heat-inactivated FBS, as active complement can opsonize cells and cause non-specific uptake [101].
- Blocking: If using specific receptor-blocking antibodies, confirm their efficacy and use appropriate isotype controls.
- Phagocyte Type: Use professional phagocytes like primary macrophages (bone marrow-derived or monocyte-derived) rather than cell lines for more physiologically relevant results [101].
- Cell Density: Ensure the correct phagocyte:target cell ratio (often between 1:1 to 1:3) and that phagocytes are at an appropriate confluence (e.g., 80%) for live imaging [101].
- Solution:
Problem: Interpreting Caspase Activation Patterns in Complex Models
Background: In disease models or with specific inhibitors, cross-talk between cell death pathways can lead to unexpected caspase activation.
Potential Causes & Solutions:
Cause 1: Pathway diversion due to genetic or pharmacological inhibition.
- Solution: If you inhibit one pathway (e.g., caspase-1 with VX-765), monitor for compensatory activation of other pathways. For instance, in caspase-1-deficient macrophages, NLRP3 activation can divert to caspase-8/-9/-3 activation and GSDME-driven secondary necrosis [51].
- Experimental Protocol:
- Treat your model with the specific inhibitor (e.g., VX-765 for caspase-1).
- Analyze cleavage of not only the target caspase but also other key caspases (e.g., caspase-8, caspase-3) and gasdermins (GSDMD, GSDME) by western blot [51].
- Be aware that some inhibitors, like VX-765, can have off-target effects and exhibit pan-caspase inhibition at higher doses [51].
Cause 2: Simultaneous initiation of multiple death pathways.
- Solution: Employ a comprehensive detection panel. The table below outlines key markers to dissect complex cell death.
Table 2: Key Markers for Differentiating Cell Death Pathways
| Target | Technique | Interpretation of Positive Result |
|---|---|---|
| Cleaved Caspase-3 | Western Blot, IF [102] | Indicates activation of apoptosis or GSDME-mediated pyroptosis. |
| Cleaved Caspase-1 | Western Blot | Indicates inflammasome activation and canonical pyroptosis. |
| GSDMD-N Terminal | Western Blot | Specific for pyroptosis execution (caspase-1/4/5/11-mediated). |
| GSDME-N Terminal | Western Blot | Indicates pyroptosis execution via caspase-3. |
| Phosphatidylserine (PS) Exposure | Annexin V Staining | Early marker of apoptosis. Can be ambiguous in late stages/other deaths. |
| Plasma Membrane Permeability | Propidium Iodide (PI) Uptake | Late apoptosis/secondary necrosis and pyroptosis are PI+. |
| HMGB1 Release | ELISA (extracellular) | Indicates lytic cell death (e.g., pyroptosis, necrosis). |
| IL-1β Release | ELISA (extracellular) | Specific for inflammatory caspase activation (pyroptosis). |
Signaling Pathways & Experimental Workflows
Caspase Cross-Talk in Cell Death Decision Making
This diagram illustrates the complex interplay and key diversion points between apoptotic and pyroptotic pathways, as described in the research [3] [51].
Efferocytosis: Phagocytic Clearance of Apoptotic Cells
This workflow details the step-by-step process of apoptotic cell clearance, from recruitment of phagocytes to the anti-inflammatory outcome [98] [99] [97].
The Scientist's Toolkit: Essential Research Reagents
This table lists key reagents and their functions for studying phagocytic clearance and inflammatory cell death.
Table 3: Key Reagents for Cell Death and Clearance Research
| Reagent / Assay | Primary Function / Application | Key Considerations |
|---|---|---|
| VX-765 (Belnacasan) | Pharmacological inhibitor of caspase-1. Used to inhibit canonical pyroptosis. | At higher doses, can exhibit pan-caspase inhibitory effects; always titrate and monitor for off-target pathway activation [51]. |
| Z-VAD-FMK | Broad-spectrum, cell-permeable pan-caspase inhibitor. Used to confirm caspase-dependent cell death. | Does not distinguish between apoptotic and pyroptotic caspases. May not fully inhibit inflammatory caspases. |
| Annexin V (FITC/APC) | Binds to externalized Phosphatidylserine (PS). Flow cytometry or fluorescence microscopy marker for early apoptosis. | PS exposure is not exclusive to apoptosis. Must be used with a viability dye (e.g., PI) to distinguish early apoptotic (Annexin V+/PI-) from late apoptotic/necrotic (Annexin V+/PI+) cells. |
| Propidium Iodide (PI) | DNA intercalating dye that is impermeable to live and early apoptotic cells. Labels cells with compromised membranes. | Standard marker for lytic cell death (pyroptosis, necrosis). Also labels cells in late-stage apoptosis (secondary necrosis). |
| LysoTracker Dyes | Fluorescent probes that accumulate in acidic compartments. Used to label lysosomes and phagolysosomes. | Useful for visualizing the final stages of efferocytosis and phagolysosomal maturation during cellular processing [101]. |
| Anti-Cleaved Caspase-3 Antibody | Detects the active (cleaved) fragment of caspase-3 via western blot or immunofluorescence (IF). Key marker for apoptosis execution. | Also activated in GSDME-mediated pyroptosis. Must be paired with gasdermin cleavage assays for definitive interpretation [102]. |
| Anti-GSDMD Antibody | Detects full-length and cleaved (N-terminal) GSDMD via western blot. Specific marker for pyroptosis execution. | Crucial for confirming pyroptosis downstream of caspase-1, -4, -5, -11. The appearance of the N-terminal fragment is diagnostic [51]. |
| Recombinant MFG-E8 | A bridging molecule that enhances PS-dependent phagocytosis. Used to boost efferocytosis efficiency in vitro. | Can be added to co-culture assays to rescue potential clearance defects and study the effects of enhanced efferocytosis [98]. |
| Lactate Dehydrogenase (LDH) Release Assay | Measures the release of the cytosolic enzyme LDH into the supernatant. A colorimetric assay for lytic cell death. | Correlates with plasma membrane rupture in pyroptosis and secondary necrosis. A simple, quantitative measure of cellular lysis [3]. |
Conclusion
Accurately distinguishing between apoptotic and pyroptotic caspase activation is paramount for both basic research and therapeutic development. This distinction extends beyond academic interest, as the immunological consequences of these pathways—silent clearance versus inflammatory alarm—directly impact disease pathogenesis and treatment strategies. The emerging understanding of caspase plasticity, exemplified by pathway diversion under impaired signaling and the integrated nature of PANoptosis, reveals a complex regulatory network that demands sophisticated experimental approaches. Future research must focus on developing more specific chemical tools, standardized assay protocols, and single-cell technologies to fully capture the dynamics of cell death decisions. For drug development, selectively modulating these pathways—potently inducing pyroptosis in cancer cells while suppressing it in inflammatory diseases—represents a promising frontier. Mastering these distinctions will ultimately enable more precise therapeutic interventions for cancer, neurodegenerative disorders, and autoinflammatory conditions.
References
- 1. Pyroptosis and apoptosis pathways engage in bidirectional ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5467448/]
- 2. The caspase -3/GSDME signal pathway as a switch between... [https://www.nature.com/articles/s41420-020-00349-0?error=cookies_not_supported&code=52d41f4e-61e4-41cf-93ea-da154777f6ef]
- 3. Caspases as master regulators of programmed cell death [https://www.nature.com/articles/s12276-025-01470-9]
- 4. Caspases as master regulators of programmed cell death [https://pmc.ncbi.nlm.nih.gov/articles/PMC12229594/]
- 5. A Comprehensive Exploration of Caspase Detection Methods [https://pmc.ncbi.nlm.nih.gov/articles/PMC11121653/]
- 6. Frontiers | Cell death in acute lung injury: caspase -regulated... [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2025.1559659/full]
- 7. What is the difference between pyroptosis and apoptosis ? [https://www.ptglab.com/news/blog/what-is-the-difference-between-pyroptosis-and-apoptosis/]
- 8. A long way to go: caspase inhibitors in clinical use [https://www.nature.com/articles/s41419-021-04240-3]
- 9. Promoting the transition from pyroptosis to apoptosis in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11809013/]
- 10. Pyroptosis - Wikipedia [https://en.wikipedia.org/wiki/Pyroptosis]
- 11. of the executioner Activation -3 and -7 promotes microglial... caspases [https://jneuroinflammation.biomedcentral.com/articles/10.1186/s12974-020-01902-5]
- 12. Studying cell death | Abcam [https://www.abcam.com/en-us/technical-resources/guides/cell-death-guide/studying-cell-death]
- 13. Comprehensive landscape of cell death mechanisms [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2025.1611055/full]
- 14. Chapter Eight Caspases: Determination of Their Activities ... [https://www.sciencedirect.com/science/article/abs/pii/S0076687908014080]
- 15. Optogenetic activators of apoptosis , necroptosis, and pyroptosis - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9014795/]
- 16. Caspases: structural and molecular mechanisms and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12052993/]
- 17. Balance Cell Apoptosis and Pyroptosis of Caspase-3 ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9817729/]
- 18. PANoptosis: bridging apoptosis, pyroptosis, and ... [https://www.nature.com/articles/s41417-024-00765-9]
- 19. What is Pyroptosis? | A Drug Development Perspective [https://biospective.com/resources/what-is-pyroptosis]
- 20. Identification and validation of PANoptosis-related biomarkers ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12649045/]
- 21. Identification of the pyroptosis, apoptosis, and necroptosis ... [https://eurjmedres.biomedcentral.com/articles/10.1186/s40001-025-02702-4]
- 22. Determination of Caspase Activation by Western Blot - PubMed [https://pubmed.ncbi.nlm.nih.gov/34033089/]
- 23. What is the difference between pyroptosis and apoptosis? [https://www.ptglab.com/news/blog/what-is-the-difference-between-pyroptosis-and-apoptosis/?srsltid=AfmBOopO8BAvwRPBG7IuzAvS8FVBcJdjaRkmBneCd3ch86sAlNhS9kiF]
- 24. Necroptosis, pyroptosis and apoptosis: an intricate game ... [https://www.nature.com/articles/s41423-020-00630-3]
- 25. Executioner caspases degrade essential mediators of ... [https://www.nature.com/articles/s41419-025-07365-x]
- 26. What is the difference between pyroptosis and apoptosis? [https://www.ptglab.com/news/blog/what-is-the-difference-between-pyroptosis-and-apoptosis/?srsltid=AfmBOoqqnBITwXk2V4gcVuYtEFoxR8CzoyNqvFQRZpokieeYCmy69twX]
- 27. Apoptosis, Pyroptosis, and Necrosis: Mechanistic Description ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1087413/]
- 28. Pyroptosis: mechanisms and diseases [https://www.nature.com/articles/s41392-021-00507-5]
- 29. Caspase Assays | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/life-science/cell-analysis/cell-viability-and-regulation/apoptosis/caspase-assays.html]
- 30. Using a Real Time Kinetic Cytotoxicity Assay to Determine ... [https://www.promega.com/resources/pubhub/using-a-real-time-kinetic-cytotoxicity-assay-to-determine-when-to-detect-apoptosis/]
- 31. Detection of Active Caspases During Apoptosis Using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6091214/]
- 32. A novel quenched fluorescent activity-based probe reveals ... [https://pubs.rsc.org/en/content/articlelanding/2016/sc/c5sc03207e]
- 33. Monitoring Caspase Activity in Living Cells Using Fluorescent ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1615789/]
- 34. Caspase assays—colorimetric and fluorescent kits [https://www.takarabio.com/products/gene-function/cell-biology-assays/apoptosis-detection-kits/apoalert-caspase-assays?srsltid=AfmBOorVK0Lo6w-SjhQPg5ly4tIrtwY-jNEKmKkhFZJnEvDt6-CtROtZ]
- 35. What is the difference between pyroptosis and apoptosis? [https://www.ptglab.com/news/blog/what-is-the-difference-between-pyroptosis-and-apoptosis/?srsltid=AfmBOoo1Dya9zR7Y5_LXX9fa3CVLRmJ2j2UaAEPznQZMJcwXGMGoQ5Sx]
- 36. Targeting regulated cell death: Apoptosis, necroptosis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11921819/]
- 37. Inflammasomes and Gasdermin D Signaling Pathways [https://resources.biomol.com/biomol-blog/inflammasomes-and-gasdermin-d-signaling-pathways]
- 38. GSDMD - Wikipedia [https://en.wikipedia.org/wiki/GSDMD]
- 39. new roles for GSDMs in regulating cellular homeostasis [https://pmc.ncbi.nlm.nih.gov/articles/PMC10611448/]
- 40. The Regulation and Modification of GSDMD Signaling in ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2022.893912/full]
- 41. Flow Cytometry Troubleshooting Guide [https://www.cellsignal.com/learn-and-support/troubleshooting/flow-cytometry-troubleshooting-guide?srsltid=AfmBOorHhHP5kPVKwGaBzNPnebkVlwxhIv-hDYa2FPlC0uRr3KewmdH4]
- 42. Multiparametric Analysis of Apoptosis by Flow Cytometry [https://pmc.ncbi.nlm.nih.gov/articles/PMC8063493/]
- 43. Multiparametric Analysis of Apoptosis by Flow Cytometry ... [https://www.creative-diagnostics.com/multiparametric-analysis-of-apoptosis-by-flow-cytometry-protocol.htm]
- 44. Flow Cytometry Troubleshooting Guide [https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/protein-biology/flow-cytometry/flow-cytometry-troubleshooting-guide?srsltid=AfmBOoowel95CDSxWBFWGSL7Jn3kWx6JwqNcWoUGE0DJ8Dt_kqKMPKGU]
- 45. Multi-parametric imaging of cell heterogeneity in apoptosis ... [https://www.sciencedirect.com/science/article/abs/pii/S1046202316302055]
- 46. What is the difference between pyroptosis and apoptosis? [https://www.ptglab.com/news/blog/what-is-the-difference-between-pyroptosis-and-apoptosis/?srsltid=AfmBOoqxqiuWg0KfqlEMd-a_w8-X3NdZ2p1BXLatFwpCrcJUzX1uTS3A]
- 47. A simple and versatile plasma membrane staining method for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12625368/]
- 48. "Reading cell death with light: Real-time visualization of ... [https://www.eurekalert.org/news-releases/1096590]
- 49. Imaging flow cytometry: from high - resolution ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1580749/full]
- 50. Identification of the pyroptosis, apoptosis, and necroptosis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12211273/]
- 51. Impaired NLRP3 inflammasome signaling diverts ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1631152/full]
- 52. Guidelines for the use and interpretation of assays ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2757140/]
- 53. Measurement and models accounting for cell death ... [https://www.nature.com/articles/s41419-020-2462-8]
- 54. Impaired NLRP3 inflammasome signaling diverts pyroptotic to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12591955/]
- 55. From pyroptosis, apoptosis and necroptosis to PANoptosis [https://pmc.ncbi.nlm.nih.gov/articles/PMC8405902/]
- 56. What is the difference between pyroptosis and apoptosis? [https://www.ptglab.com/news/blog/what-is-the-difference-between-pyroptosis-and-apoptosis/?srsltid=AfmBOorDhYQmgrcz79Ysi_XV_ECC27z3Le19CybHaoh1eVTcPJshzrx7]
- 57. Mechanisms of Cell Death: Pyroptosis | CST Blog [https://blog.cellsignal.com/mechanisms-of-cell-death-pyroptosis]
- 58. A comparative study of apoptosis, pyroptosis, necroptosis, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10898734/]
- 59. Optogenetic Induction of Pyroptosis, Necroptosis, and ... [https://en.bio-protocol.org/en/bpdetail?id=4762&type=0]
- 60. Role of Pyroptosis Pathway Activation by Gram-Negative ... [https://www.mdpi.com/2073-4409/13/21/1758]
- 61. Caspase assay selection guide [https://www.abcam.com/en-us/products/selection-guides/caspase-assay-selection-guide]
- 62. What is the difference between pyroptosis and apoptosis? [https://www.ptglab.com/news/blog/what-is-the-difference-between-pyroptosis-and-apoptosis/?srsltid=AfmBOoo9iaepqsD7XHZ2PybdLTSQUDCB-ZFim_ub8cZ77GtraxZGFaeZ]
- 63. Apoptosis DNA fragmentation analysis protocol [https://www.abcam.com/en-us/technical-resources/protocols/apoptosis-dna-fragmentation]
- 64. Molecular mechanisms and functions of pyroptosis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5416822/]
- 65. Small Molecule Inhibitors Selection Guide [https://resources.biomol.com/biomol-blog/small-molecule-inhibitors-selection-guide]
- 66. Targeting the inhibitors of apoptosis proteins (IAPs) to ... [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2025.1562167/full]
- 67. PANoptosis in cancer: bridging molecular mechanisms to ... [https://www.nature.com/articles/s41423-025-01329-z]
- 68. PANoptosis in neurological disorders - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC12187663/]
- 69. Cell death controlling complexes and their potential ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11113151/]
- 70. The complete guide to studying cell death [https://www.abcam.com/en-us/technical-resources/guides/cell-death-guide]
- 71. Machine Learning Screening and Validation of PANoptosis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11268777/]
- 72. Pyroptotic cell death defends against intracellular pathogens - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4400865/]
- 73. Caspase-1 induced pyroptotic cell death - PMC [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3609431/]
- 74. Caspase Activation Pathways: an Overview - NCBI - NIH [https://www.ncbi.nlm.nih.gov/books/NBK14011/]
- 75. Caspase-1-Mediated Activation of Interleukin-1β (IL- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1539628/]
- 76. Flow cytometry-based apoptosis detection - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3863590/]
- 77. Cleavage of DFNA5 by caspase-3 during apoptosis ... [https://www.nature.com/articles/ncomms14128]
- 78. PARP1 Inhibitors Induce Pyroptosis via Caspase 3- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9933147/]
- 79. Caspase-1 activation and mature interleukin-1β release ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3652645/]
- 80. Bcl-2 Blocks a Caspase-Dependent Pathway of Apoptosis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC111671/]
- 81. Protocol for Generation of Single-Gene Knockout in Hard- ... [https://pubmed.ncbi.nlm.nih.gov/40655415/]
- 82. Generating and validating CRISPR-Cas9 knock-out cell lines [https://www.abcam.com/en-us/stories/articles/crispr-cas9-knockout-cell-lines]
- 83. Protocol to rapidly screen CRISPR-Cas9 gene editing ... [https://www.sciencedirect.com/science/article/pii/S2666166725003569]
- 84. What is the difference between pyroptosis and apoptosis? [https://www.ptglab.com/news/blog/what-is-the-difference-between-pyroptosis-and-apoptosis/?srsltid=AfmBOooB2mO7fYfpQuyg7Qwi6uxSfpmJKlXvjslUlHezuXuclMKfIdvw]
- 85. Pyroptosis: molecular mechanisms and roles in disease - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12012027/]
- 86. LDH assay kit guide: Principles and applications [https://www.abcam.com/en-us/knowledge-center/cell-biology/ldh-assay-kit-guide-principles-and-application]
- 87. Is pyroptosis a brake or an accelerator in the fate of ... [https://www.nature.com/articles/s41419-025-07866-9]
- 88. Crosstalk of pyroptosis and cytokine in the tumor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11607834/]
- 89. Detection of Gasdermin Activation and Lytic Cell Death ... [https://pubmed.ncbi.nlm.nih.gov/35759200/]
- 90. Programmed cell death detection methods - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC9308588/]
- 91. control antiviral innate Caspases | immunity & Molecular... Cellular [https://www.nature.com/articles/cmi201744?error=cookies_not_supported]
- 92. Comparison of Caspase Activation and Subcellular ... [https://www.sciencedirect.com/science/article/pii/S0006497120561132]
- 93. Troubleshooting Guide: Immunohistochemistry [https://www.rndsystems.com/resources/technical/troubleshooting-guide-immunohistochemistry]
- 94. Immunohistochemistry (IHC) Troubleshooting Guide & the ... [https://www.cellsignal.com/learn-and-support/troubleshooting/ihc-troubleshooting-guide?srsltid=AfmBOoqMeAl155dfmIxLHV1rc4JPLVsw3t3jYx8N4BT71iUCPw2zvtLL]
- 95. Troubleshooting Flow Cytometry Issues [https://www.bosterbio.com/protocol-and-troubleshooting/flow-cytometry-troubleshooting?srsltid=AfmBOooo0PHePnpw0kIpJQL_b6dlv9nApEHz9A9d4fVsEQx2ZDoljKKW]
- 96. The pyroptosis mediated biomarker pattern: an emerging ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10765853/]
- 97. Clearance of apoptotic cells by phagocytes [https://www.nature.com/articles/4402184]
- 98. Current Understanding of the Mechanisms for Clearance of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4147779/]
- 99. Phagocytic clearance of apoptotic cells: role in lung disease [https://pmc.ncbi.nlm.nih.gov/articles/PMC3956128/]
- 100. Phagocytosis of apoptotic cells and the resolution ... [https://www.sciencedirect.com/science/article/pii/S0925443903001467]
- 101. Live Imaging of Phagocytosis: Tips to Get the Perfect Picture [https://bitesizebio.com/52776/live-imaging-of-phagocytosis/]
- 102. Protocol for detection of caspases using immunofluorescence [https://www.abcam.com/en-us/technical-resources/protocols/immunofluorescence-protocol-to-detect-caspases]