Mastering Rhodamine 123 Quenching Mode: A Troubleshooting Guide for Accurate Acute ΔΨm Measurement
This article provides a comprehensive guide for researchers and drug development professionals on the effective use of Rhodamine 123 (Rhod123) in quenching mode to monitor acute changes in mitochondrial membrane...
Mastering Rhodamine 123 Quenching Mode: A Troubleshooting Guide for Accurate Acute ΔΨm Measurement
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on the effective use of Rhodamine 123 (Rhod123) in quenching mode to monitor acute changes in mitochondrial membrane potential (ΔΨm). It covers the foundational principles of ΔΨm and Rhod123 behavior, detailed methodological protocols for acute perturbation experiments, systematic troubleshooting for common pitfalls like insufficient quenching and photobleaching, and essential validation strategies using pharmacological controls and complementary assays. The guide synthesizes best practices to ensure accurate, reproducible, and interpretable data in the study of mitochondrial function in health, disease, and drug discovery contexts.
Understanding the Core Principles: Rhod123, ΔΨm, and the Quenching Mechanism
The Critical Role of Mitochondrial Membrane Potential (ΔΨm) in Cellular Energetics and Health
FAQs & Troubleshooting Guide: Rhodamine 123 in Quenching Mode
This guide addresses common challenges researchers face when using Rhodamine 123 (Rh123) in quenching mode to monitor acute changes in mitochondrial membrane potential (ΔΨm).
FAQ 1: What is the fundamental principle behind using Rh123 in quenching mode for ΔΨm measurement?
- Answer: Rh123 is a cationic, lipophilic fluorescent dye that accumulates in the mitochondrial matrix in a ΔΨm-dependent manner. In quenching mode, high dye concentrations are used, leading to its aggregation within energized mitochondria. This aggregation causes fluorescence quenching (a non-linear decrease in fluorescence intensity). Mitochondrial depolarization triggers the release of Rh123 into the cytoplasm, where it de-aggre gates and causes an increase in fluorescence, a phenomenon known as "fluorescence unquenching" [1] [2]. The kinetics of this fluorescence increase are proportional to the loss of ΔΨm [3].
FAQ 2: My Rh123 signal is unstable or shows unexpected changes. What could be the cause?
- Answer: Signal instability can arise from several sources. A primary concern is the potential for intracellular or intramitochondrial modification of the Rh123 molecule itself. Studies have shown that Rh123 can be metabolically altered inside cells, potentially by enzymes like cytochrome P450, which can change its fluorescent properties and lead to inaccurate ΔΨm readings [4]. Furthermore, significant binding of Rh123 to cellular components like phospholipids or proteins can also affect the signal and its interpretation [5] [4].
FAQ 3: Why does the dye release more slowly from some cell types, like cancer cells, after uncoupler treatment?
- Answer: Delayed release is often not solely due to a higher ΔΨm. It can be influenced by altered activity of efflux transporters, such as P-glycoprotein (MDR1) or other ATP-binding cassette (ABC) transporters, which are frequently overexpressed in cancer cells [6] [4]. Additionally, increased dye retention and transformation within tumor cells can contribute to this phenomenon, complicating the direct correlation between fluorescence and membrane potential [4].
FAQ 4: What are the critical controls for validating my Rh123 quenching experiments?
- Answer: Robust experiments require key pharmacological controls:
- FCCP/CCCP: These protonophores uncouple mitochondria and dissipate ΔΨm, providing a positive control for depolarization and the associated fluorescence unquenching [2].
- Oligomycin: This ATP synthase inhibitor hyperpolarizes mitochondria by blocking proton re-entry, which should lead to increased dye quenching [1] [2].
- Amiodarone: Can be used to investigate the role of dye modification, as it has been shown to block the transformation and export of Rh123 [4].
FAQ 5: How can I distinguish between a true loss of ΔΨm and other factors that affect fluorescence?
- Answer: To ensure accuracy, employ complementary approaches:
- Parallel Assays: Use additional ΔΨm probes with different chemical properties (e.g., TMRM, JC-1) to confirm the observed trends [2].
- Monitor Plasma Membrane Potential (Δψp): Changes in the plasma membrane potential can influence cationic dye uptake. Using a probe like DiBAC₄(3) allows you to account for this variable [2].
- Image Mitochondrial Morphology and Mass: Use Mitotracker dyes (e.g., MitoTracker Green) under de-energized conditions to control for changes in mitochondrial content that could affect total dye uptake [7] [2].
Troubleshooting Table for Common Problems
| Problem | Potential Cause | Recommended Solution |
|---|---|---|
| Weak or No Fluorescence Signal | Inadequate dye loading; efflux by MDR transporters.Low mitochondrial mass. | Optimize loading concentration and time.Consider using an MDR inhibitor (verify it doesn't affect your experiment).Confirm mitochondrial content with a ΔΨm-independent stain (e.g., MitoTracker Green). |
| High Background Cytosolic Fluorescence | Dye concentration too low for quenching mode.Mitochondria are depolarized. | Increase Rh123 concentration to achieve quenching conditions.Validate mitochondrial health with FCCP/CCCP. |
| Signal Instability or Drift During Acquisition | Photobleaching.Dye modification or export. | Reduce light exposure/integration time.Use a fresh dye stock and include amiodarone control to check for modification. |
| Lack of Response to FCCP/CCCP | Dye is trapped or modified in the matrix.Uncoupler is inactive or concentration is too low. | Include amiodarone control.Prepare fresh uncoupler stock and perform a dose-response curve. |
| Heterogeneous Signal Across Cell Population | Genuine biological heterogeneity in ΔΨm.Variation in dye loading/efflux. | Analyze sub-populations separately via flow cytometry.Check for consistency in cell health and treatment. |
Detailed Protocol: Measuring Acute ΔΨm Changes with Rh123 Unquenching
This protocol is adapted from studies investigating cadmium-induced mitochondrial depolarization and is suitable for real-time monitoring of acute ΔΨm changes in live cells [3].
Key Reagent Solutions:
- Rhodamine 123 Stock Solution: 1 mM in DMSO. Aliquot and store at -20°C, protected from light.
- FCCP/CCCP Stock Solution: 1-10 mM in DMSO or Ethanol.
- Oligomycin Stock Solution: 5-10 mg/mL in DMSO.
- Assay Buffer: Hanks' Balanced Salt Solution (HBSS) or another physiologically relevant buffer, supplemented with 10 mM HEPES (pH 7.4).
Methodology:
- Cell Preparation: Plate cells on glass-bottom dishes or coverslips suitable for live-cell imaging. Grow to 60-80% confluency.
- Dye Loading (Quenching Conditions): Load cells with a high concentration of Rh123 (e.g., 1-10 µM) in pre-warmed assay buffer or culture medium for 15-30 minutes at 37°C, 5% CO₂. The optimal concentration must be empirically determined to achieve mitochondrial quenching (punctate structures with dim fluorescence).
- Washing: Gently wash the cells 2-3 times with warm, dye-free assay buffer to remove extracellular Rh123.
- Baseline Acquisition: Place cells in fresh assay buffer on the microscope stage (maintained at 37°C). Acquire baseline fluorescence images (Ex/Em ~505/534 nm) for 2-5 minutes to establish a stable signal.
- Acute Treatment & Kinetic Recording: Add the experimental treatment (e.g., a toxicant, drug, or metabolic inhibitor) directly to the dish. Continue acquiring images at short intervals (e.g., every 10-30 seconds) for the duration required by the experiment.
- Pharmacological Controls: At the end of the experiment, add FCCP/CCCP (e.g., 1-10 µM) to fully depolarize mitochondria and record the maximum fluorescence unquenching signal.
- Data Analysis: Quantify the mean fluorescence intensity in the mitochondrial regions over time. The data is often normalized to the baseline (F/F₀) or the FCCP-induced maximum signal.
Quantitative Data from Key Studies
The following table summarizes quantitative findings on how various perturbations affect ΔΨm as measured by Rh123 fluorescence.
Table: Quantitative Effects on ΔΨm Measured via Rhodamine 123
| Experimental Model | Intervention | Effect on ΔΨm (Rh123 Fluorescence) | Key Quantitative Finding | Citation |
|---|---|---|---|---|
| AD Patient iPSC-Derived Neurons | RyR negative modulator (Ryanodex) | Prevents pathological hyperpolarization/depolarization | Prevented increased Ca²⁺ uptake and exaggerated mitochondrial membrane depolarization. | [8] |
| Isolated Rat Liver Mitochondria | ADP (+ Succinate) | Partial depolarization (State 3) | Rate of RH-123 fluorescence decay (quenching) is proportional to ΔΨm. Addition of ADP decreased quenching rate. | [1] |
| Isolated Rat Liver Mitochondria | Oligomycin (+ Succinate & ADP) | Hyperpolarization | Increased the initial rate of RH-123 quenching. | [1] |
| Human Intestinal TC7 Cells | Cadmium (Cd, 50 µM) | Dissipation (Depolarization) | Induced ΔΨm dissipation; effect was delayed but not prevented by the antioxidant mannitol. | [3] |
| HEK293 IF1-KO Cells | Genetic deletion of ATP5IF1 | Chronic Hyperpolarization | Showed higher resting ΔΨm than WT cells, as measured by TMRE/MitoTracker Green ratio. | [7] |
| HEK293 IF1-KO Cells | Culture in Galactose Medium | Depolarization (more pronounced in KO) | ΔΨm decreased in both WT and KO cells, but the effect was significantly larger in hyperpolarized IF1-KO cells. | [7] |
Signaling Pathways & Experimental Workflows
This section provides visual summaries of the core concepts and experimental workflows.
Rh123 Quenching Mechanism for ΔΨm
The diagram below illustrates the principle of Rhodamine 123 accumulation and fluorescence quenching in energized mitochondria.
Experimental Workflow for Acute ΔΨm Measurement
This flowchart outlines the key steps in a typical experiment designed to measure acute changes in ΔΨm using Rh123 in quenching mode.
The Scientist's Toolkit: Essential Research Reagents
This table details key reagents and their functions for experiments focused on ΔΨm and mitochondrial function using Rh123.
Research Reagent Solutions
| Reagent | Function/Brief Explanation | Key Considerations |
|---|---|---|
| Rhodamine 123 (Rh123) | Cationic, fluorescent ΔΨm probe. Used in quenching mode for acute changes and non-quenching mode for chronic measurements. | Subject to intracellular modification and efflux by MDR transporters. Concentration is critical for quenching vs. non-quenching mode [2] [4]. |
| FCCP / CCCP | Protonophores that dissipate the proton gradient and ΔΨm. Used as a positive control for complete mitochondrial depolarization. | Prepare fresh stock solutions in DMSO/EtOH. Final concentration typically 1-10 µM [1] [2]. |
| Oligomycin | ATP synthase inhibitor. Blocks proton flow through Complex V, leading to mitochondrial hyperpolarization. | A key control to demonstrate hyperpolarization and confirm dye responsiveness. Used at ~1-10 µg/mL [1] [2]. |
| TMRE / TMRM | Tetramethylrhodamine-based ΔΨm probes. Often used in non-quenching mode. Generally exhibit less binding to mitochondria and lower toxicity than some other dyes. | Preferred for long-term or quantitative imaging in non-quenching mode due to more reliable Nernstian distribution [2] [7]. |
| MitoTracker Green (MTG) | A cell-permeant dye that accumulates in mitochondria regardless of membrane potential. Used to normalize for mitochondrial mass and morphology. | Staining is not dependent on ΔΨm. Ideal for co-staining with potential-sensitive dyes to control for mitochondrial content [7]. |
| Ryanodex | Ryanodine receptor (RyR) negative allosteric modulator. Used to inhibit pathological ER-calcium release, preventing downstream mitochondrial Ca²⁺ overload and dysfunction. | Shown to preserve mitochondrial function in Alzheimer's disease models by normalizing ER-mitochondrial Ca²⁺ transfer [8]. |
| Amiodarone | A drug that can block the export and transformation of xenobiotics from cells. Useful as a control to investigate intracellular modification of Rh123. | Helps determine if changes in Rh123 fluorescence are due to true ΔΨm shifts or probe metabolism/export [4]. |
Why Rhodamine 123? Properties of a Cationic Lipophilic Dye for ΔΨm Measurement
Fundamental Principles: How Rhodamine 123 Functions as a ΔΨm Probe
Rhodamine 123 (R123) is a lipophilic monovalent cationic dye that serves as a robust fluorescent indicator for mitochondrial membrane potential (ΔΨm). Its functionality is based on the Nernst equation, governing its distribution across the mitochondrial inner membrane [9] [10]. In living cells, the dye accumulates within the mitochondrial matrix in response to the negative internal potential generated by the electron transport chain [11] [12]. This potential-dependent accumulation is the cornerstone of its use as a potentiometric probe.
Upon accumulation in energized mitochondria, R123 exhibits two key spectral changes: a red shift in its fluorescence emission spectrum and significant concentration-dependent fluorescence quenching [13] [14]. The quenching phenomenon is particularly critical for its use in "quench mode," where the accumulated dye becomes self-quenched, leading to a decrease in overall fluorescence intensity that correlates with increased ΔΨm [9]. The dye can achieve remarkable accumulation ratios, with concentration gradients (in-to-out) approaching 4000:1 in highly energized mitochondria [14].
Table 1: Key Spectral and Accumulation Properties of Rhodamine 123
| Property | Description | Experimental Significance |
|---|---|---|
| Chemical Nature | Lipophilic monovalent cation [9] | Permeates phospholipid bilayers and accumulates in response to ΔΨm |
| Excitation/Emission | ~505 nm / ~560 nm [15] | Compatible with standard FITC filter sets |
| Spectral Shift on Energization | Red shift in absorption and fluorescence [13] | Provides basis for ratiometric measurements in isolated mitochondria |
| Fluorescence Change on Accumulation | Quenching (decreased intensity) [13] [14] | Enables "quench mode" detection of ΔΨm increases |
| Typical Accumulation Ratio | Up to ~4000:1 (in-to-out) [14] | High sensitivity to changes in membrane potential |
Comparative Advantages and Limitations of R123
Advantages in Experimental Applications
R123 offers several compelling advantages that explain its persistent popularity in mitochondrial research. The dye demonstrates high specificity for mitochondrial labeling in response to energization, with staining that is effectively prevented by uncouplers that collapse ΔΨm [9]. It also possesses high quantum yield (0.90), providing excellent signal-to-noise ratio in fluorescence measurements [15]. From a practical standpoint, R123 is readily available and relatively cost-effective compared to some newer-generation dyes [9] [10]. When used at appropriate concentrations (typically low nanomolar range for cellular experiments), it exhibits minimal suppression of mitochondrial respiration, making it suitable for monitoring physiological processes without significantly perturbing the system under study [13].
Critical Limitations and Cytotoxic Effects
Despite its advantages, researchers must be aware of significant limitations. R123 exhibits concentration-dependent inhibition of mitochondrial function, particularly affecting ADP-stimulated (State 3) respiration with a reported Ki of 12 μM in isolated rat-liver mitochondria [14]. The dye also suppresses ATPase activity in inverted inner membrane vesicles and partially purified F1-ATPase [14]. At higher concentrations (above approximately 10 μM), R123 can induce rapid swelling in energized mitochondria [14]. Furthermore, the relationship between fluorescence intensity and membrane potential is non-linear and highly sensitive to experimental conditions, including total dye concentration and mitochondrial density [9]. This necessitates careful calibration for quantitative interpretations.
Table 2: Comparison of Rhodamine 123 with Other Common ΔΨm Probes
| Probe | Binding Characteristics | Metabolic Inhibition | Best Use Cases |
|---|---|---|---|
| Rhodamine 123 | Binds to inner and outer aspects of inner membrane; temperature-dependent [13] | Suppresses State 3 respiration (Ki = 12 μM); inhibits ATPase [13] [14] | Qualitative assessment of ΔΨm changes; flow cytometry |
| TMRM | Lower membrane binding compared to TMRE and R123 [13] | Minimal suppression of respiration at low concentrations [13] | Quantitative potential measurements; kinetic studies |
| TMRE | Highest degree of membrane binding [13] | Greatest suppression of mitochondrial respiration [13] | Tissue slice imaging; when high accumulation is needed |
| JC-1 | Forms J-aggregates at high membrane potentials | Potential-dependent spectral shift | Distinguishing high vs. low ΔΨm; flow cytometry |
Troubleshooting Guide: Frequently Asked Questions
Q1: Why does my R123 fluorescence signal become unreliable or inconsistent during kinetic measurements of acute ΔΨm changes?
This common issue typically stems from violation of the fundamental principles of the R123 quenching assay [11]. The problem often occurs in glucose-stimulated or oligomycin-inhibited β-cells, where the dye's behavior deviates from expected patterns. Ensure you are using the lowest effective dye concentration (typically 50-200 nM for cells) to minimize metabolic inhibition [16] [14]. Additionally, account for inner filter effects—the attenuation of fluorescence due to absorption of incident light by the dye itself—which become significant at higher concentrations and can distort measurements [9].
Q2: How does self-quenching affect my R123 measurements and what concentration range is optimal?
R123 fluorescence exhibits a well-characterized non-linear relationship with concentration due to self-quenching [9]. The fluorescence intensity peaks at specific concentrations (approximately 11-20 μM in aqueous solution, depending on light path) then decreases toward zero at higher concentrations [9]. For practical experiments, use low nanomolar concentrations (50-200 nM) for cellular work to avoid quenching artifacts and minimize toxicity [16]. In isolated mitochondria, slightly higher concentrations may be used (up to low micromolar), but careful calibration is essential [13].
Q3: My R123 staining shows unexpected patterns in isolated brain mitochondria. What could explain spontaneous fluorescence fluctuations?
Approximately 70% of energized isolated brain mitochondria exhibit large-amplitude spontaneous fluctuations in ΔΨm when measured with R123 [16]. This represents an intermediate, unstable state of mitochondria that may reflect underlying dysfunction. These fluctuations are stochastic phenomena observed in individual mitochondria and are not necessarily indicative of technical problems with your staining protocol [16]. Control experiments with uncouplers (e.g., FCCP) can help distinguish true biological phenomena from artifacts.
Q4: When should I avoid using R123 and consider alternative dyes like TMRM?
Choose alternative probes when: (1) conducting quantitative measurements of absolute ΔΨm magnitude (TMRM is preferred) [11]; (2) working with intact tissues or organs where R123's spectral shifts may not occur as in isolated mitochondria [13]; (3) studying processes highly sensitive to F1F0-ATPase inhibition; or (4) when you observe significant cytotoxicity at your working concentrations.
Essential Experimental Protocols
Protocol for Isolated Mitochondria (Bulk Suspension Measurements)
This protocol adapts methodologies from multiple sources for measuring ΔΨm in isolated mitochondrial suspensions [13] [9] [17].
- Isolation Buffer Preparation: Prepare isolation buffer appropriate for your tissue type. For cardiac mitochondria: 200 mM mannitol, 50 mM sucrose, 5 mM KH₂PO₄, 5 mM MOPS, 0.1% fatty acid-free BSA, 1 mM EGTA, pH to 7.15 with KOH [9].
- Respiratory Buffer Preparation: Prepare respiration buffer: 130 mM KCl, 5 mM K₂HPO₄·3H₂O, 20 mM MOPS, 2.5 mM EGTA, 1 μM tetrasodium pyrophosphate, 0.1% BSA, pH to 7.15 with KOH [9].
- Mitochondrial Isolation: Isolate mitochondria using standard differential centrifugation protocols. Determine protein concentration using Bradford or Biuret assay [9] [16].
- Dye Preparation: Prepare a stock solution of R123 in ethanol or DMSO. Final concentration in assay typically ranges from 100 nM to 1 μM [13] [17].
- Fluorometer Setup: Set excitation to 503 nm and emission to 527 nm with appropriate slits (e.g., 2-5 nm bandpass). Use continuous stirring and maintain temperature at 25-37°C [9].
- Assay Execution: Add respiratory buffer to cuvette. Add mitochondrial suspension (0.5 mg protein/mL). Add R123 to desired final concentration. Initiate energization by adding substrates (e.g., 5 mM pyruvate/malate or 10 mM succinate with rotenone). Record baseline fluorescence.
- Validation and Calibration: At conclusion, add uncoupler (e.g., 4 μM CCCP or FCCP) to collapse ΔΨm and confirm fluorescence recovery [9] [17].
Protocol for Single-Cell Imaging in Cultured Systems
This protocol incorporates best practices for live-cell imaging with R123 [16] [11] [12].
- Dye Loading Solution: Prepare loading solution using standard extracellular buffer (e.g., Hanks' Balanced Salt Solution, pH 7.4) containing 100-200 nM R123 [16].
- Cell Preparation: Culture cells on appropriate imaging-compatible dishes (e.g., glass-bottom dishes). Ensure cells are 60-80% confluent at time of imaging.
- Loading Protocol: Incubate cells with R123 loading solution for 15-30 minutes at 37°C in the dark.
- Dye Removal and Stabilization: Replace R123-containing medium with dye-free pre-warmed buffer. Allow 10-15 minutes for dye stabilization before imaging.
- Microscope Configuration: Use epifluorescence or confocal microscope with FITC filter sets (excitation ~480 nm, emission ~535 nm). Use minimal laser power or illumination intensity to minimize phototoxicity and dye bleaching. Acquire images every 10-60 seconds depending on kinetics of interest [16].
- Controls and Validation: Include parallel samples treated with uncoupler (e.g., 1-2 μM FCCP) to confirm ΔΨm-dependent staining. Use non-quench mode (low dye concentrations) for most reliable single-cell assessments [11].
Diagram 1: Comprehensive Workflow for Rhodamine 123-Based ΔΨm Measurements. This flowchart outlines key steps from experimental planning through data interpretation, highlighting critical decision points and quality control measures.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for R123-Based ΔΨm Measurements
| Reagent/Category | Specific Examples | Function/Purpose | Critical Considerations |
|---|---|---|---|
| Potentiometric Dyes | Rhodamine 123, TMRM, TMRE | ΔΨm-dependent accumulation and fluorescence signal | R123 has higher binding and toxicity than TMRM [13] |
| Substrates | Succinate (with rotenone), Pyruvate/Malate, Glutamate | Provide reducing equivalents to electron transport chain | Different substrates drive different respiration rates |
| Inhibitors/Uncouplers | FCCP, CCCP, Oligomycin | Collapse ΔΨm (uncouplers) or inhibit ATP synthase (oligomycin) | Essential controls for validation [16] [17] |
| Isolation Reagents | Mannitol, Sucrose, BSA, EGTA, Percoll | Maintain mitochondrial integrity during isolation | BSA absorbs free fatty acids; EGTA chelates calcium |
| Buffers | MOPS, HEPES, KCl-based media | Maintain physiological pH and ionic environment | KCl-based buffers better mimic intracellular environment |
Diagram 2: Mechanism of Rhodamine 123 Accumulation and Fluorescence Response. This diagram illustrates the potential-dependent accumulation of R123 in mitochondria and the subsequent fluorescence quenching that enables ΔΨm measurement.
Fluorescence quenching is a reversible process where the intensity of light emitted by a fluorescent dye is reduced due to molecular interactions or environmental conditions. Unlike permanent photobleaching, quenching can be reversed when conditions change, making it particularly valuable for monitoring dynamic cellular processes [18].
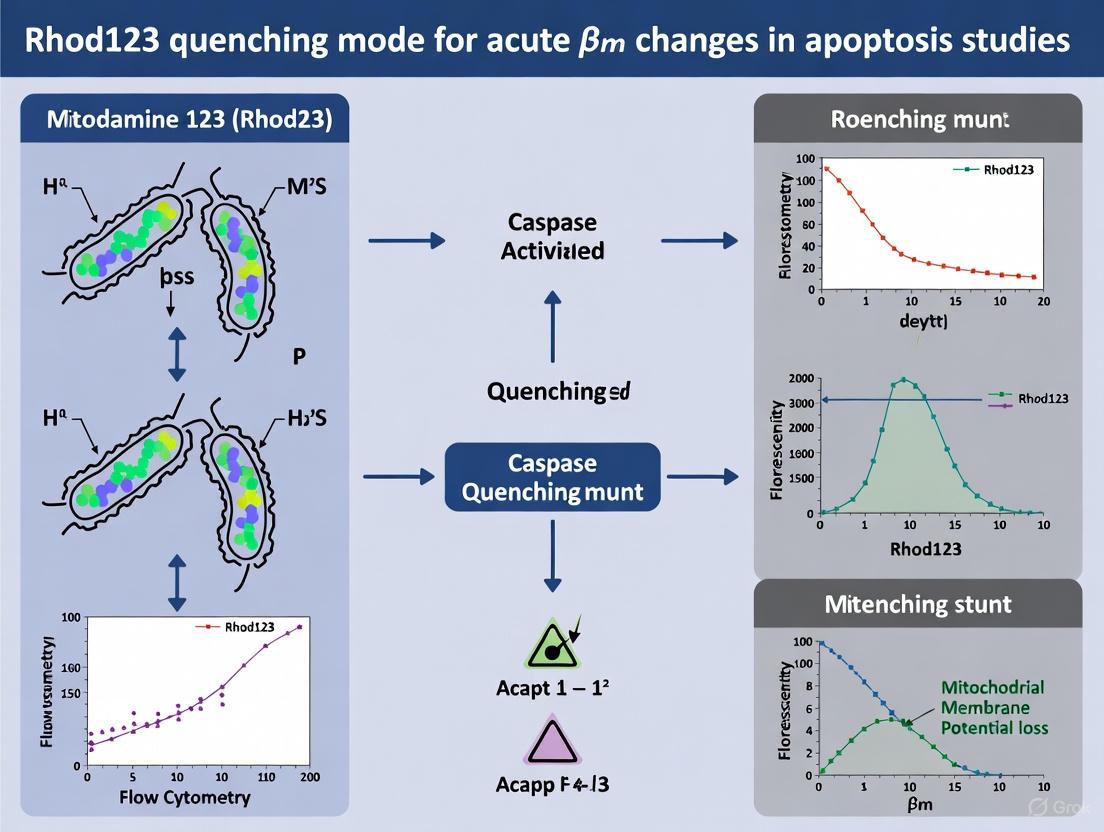
In the context of mitochondrial membrane potential (ΔΨm) measurement, quenching mode refers to an experimental setup where lipophilic cationic dyes, such as Rhodamine 123 (Rhod123), are used at high concentrations (typically ~1-10 μM). At these concentrations, the dyes accumulate in the mitochondrial matrix to such an extent that they form aggregates, leading to self-quenching—a phenomenon where fluorescence is reduced due to close molecular proximity between dye molecules [19] [18]. This operational mode is especially suited for monitoring rapid, acute changes in mitochondrial membrane potential in living cells.
Fundamental Mechanisms of Fluorescence Quenching
Physical Basis of Quenching
Fluorescence quenching in mitochondrial dyes occurs through several physical mechanisms:
- Self-Quenching: Occurs when the concentration of fluorescent molecules is too high, causing them to aggregate and interfere with each other's fluorescence through various energy transfer mechanisms [18].
- Collisional Quenching: Results from collisions between excited fluorescent molecules and quenchers, leading to energy transfer without light emission [18] [20].
- Static Quenching: Involves the formation of non-fluorescent complexes between fluorescent molecules in the ground state before excitation occurs [18] [20].
For Rhod123 specifically, the quenching mechanism primarily involves self-quenching through dye aggregation at high matrix concentrations. When mitochondria are polarized (more negative interior), more cationic dye accumulates in the matrix, increasing aggregation and thus quenching. Mitochondrial depolarization reduces dye accumulation, decreasing aggregation and causing fluorescence "unquenching" or increased fluorescence signal [19].
Table 1: Common Quenching Mechanisms in Fluorescence Spectroscopy
| Mechanism | Process | Distance Dependence | Reversibility |
|---|---|---|---|
| Self-Quenching | Dye aggregation at high concentrations | Molecular proximity | Fully reversible |
| Collisional Quenching | Energy loss through molecular collisions | Diffusion-dependent | Reversible |
| FRET | Non-radiative energy transfer between dyes | 1/R⁶ (strong distance dependence) | Reversible |
| Static Quenching | Non-fluorescent complex formation | Direct contact | Often reversible |
Rhod123 Quenching Mode: Experimental Framework
Optimal Usage Parameters for Rhod123
Rhod123 is particularly well-suited for quenching mode applications in acute ΔΨm studies due to its specific physicochemical properties. The dye is typically used at concentrations of ~1-10 μM in quenching mode, which promotes the dye aggregation necessary for the quenching/unquenching response to membrane potential changes [19].
Compared to other common ΔΨm dyes like TMRM and TMRE, Rhod123 exhibits slower permeation across membranes, which makes the quenching/unquenching changes in fluorescence easier to detect and monitor in real-time experiments [19]. This characteristic is particularly valuable for capturing transient mitochondrial membrane potential fluctuations.
Experimental Workflow for Acute ΔΨm Monitoring
The standard protocol for Rhod123 quenching mode experiments follows this sequence:
- Dye Loading: Incubate cells with 1-10 μM Rhod123 to achieve sufficient mitochondrial loading
- Washout: Remove extracellular dye to eliminate background signal
- Baseline Imaging: Record initial fluorescence under experimental conditions
- Treatment Application: Introduce experimental treatments while continuously monitoring fluorescence
- Signal Interpretation: Interpret fluorescence changes as indicators of ΔΨm changes
In this operational mode, depolarization of ΔΨm causes dye release from mitochondria, reducing aggregation and resulting in increased fluorescence (unquenching). Conversely, hyperpolarization increases dye accumulation and aggregation, leading to decreased fluorescence (further quenching) [19].
Troubleshooting Guide: Common Experimental Challenges
FAQ: Addressing Rhod123 Quenching Mode Issues
Q: My Rhod123 fluorescence signal is too weak, even in control conditions. What could be the problem? A: Several factors could cause insufficient signal:
- Inadequate dye loading: Verify Rhod123 concentration (should be 1-10 μM) and loading duration
- Dye efflux: Some cell types express multidrug resistance transporters that actively export cationic dyes; consider using transporter inhibitors like verapamil
- Excessive photobleaching: Reduce illumination intensity or exposure time, use neutral density filters
- Incorrect filter sets: Confirm your microscope has appropriate excitation/emission filters for Rhod123 (≈507/529 nm)
Q: I observe unexpected fluorescence increases when applying depolarizing agents. How should I interpret this? A: This is the expected response in quenching mode. Remember the fundamental principle: Depolarization → Dye release from matrix → Reduced aggregation → Fluorescence unquenching (increase). Validate your system using pharmacological controls:
- FCCP/CCCP (1-5 μM): Should cause rapid fluorescence increase due to complete depolarization
- Oligomycin (1-5 μg/mL): Should cause gradual fluorescence decrease due to hyperpolarization
Q: My fluorescence signal shows excessive noise or instability during time-lapse imaging. How can I improve signal quality? A: Consider these optimization strategies:
- Maintain constant dye presence: For acute treatments after dye loading, keep dye in bath during imaging
- Control environmental factors: Stabilize temperature and CO₂ levels, as pH fluctuations affect dye behavior
- Optimize imaging parameters: Increase binning, reduce sampling frequency, or use averaging to improve signal-to-noise ratio
- Validate mitochondrial specificity: Confirm signal co-localization with mitochondrial markers
Q: How can I distinguish true ΔΨm changes from artifacts caused by altered mitochondrial mass or morphology? A: Always implement complementary controls:
- Mitochondrial mass markers: Use Mitotracker dyes (under depolarizing conditions) or mitochondrial-targeted fluorescent proteins
- Morphological assessment: Evaluate mitochondrial network structure using high-resolution imaging
- Parallel validation: Confirm key findings using alternative ΔΨm dyes in non-quenching mode
Research Reagent Solutions and Experimental Controls
Essential Reagents for Rhod123 Quenching Mode Experiments
Table 2: Key Reagents for Quenching Mode Experiments
| Reagent/Category | Specific Examples | Function/Application | Working Concentration |
|---|---|---|---|
| ΔΨm Dyes (Quenching Mode) | Rhodamine 123 (Rhod123) | Monitoring acute ΔΨm changes via quenching/unquenching | 1-10 μM |
| Pharmacological Controls | FCCP/CCCP | Positive control: complete depolarization | 1-5 μM |
| Oligomycin | Positive control: hyperpolarization | 1-5 μg/mL | |
| Validation Dyes | TMRM, TMRE | Non-quenching mode validation | 1-30 nM |
| JC-1 | Ratiometric confirmation | Concentration-dependent | |
| Mitochondrial Markers | Mitotracker Deep Red | Mitochondrial mass control | 50-100 nM |
| Inhibitors | Verapamil | Blocks dye efflux transporters | 10-50 μM |
Critical Experimental Controls for Valid Interpretation
To ensure accurate interpretation of Rhod123 quenching mode results, implement these essential controls:
Pharmacological Validation
- Include FCCP/CCCP and oligomycin treatments in every experimental series
- Verify expected directional responses: FCCP→increased fluorescence; oligomycin→decreased fluorescence
Plasma Membrane Potential (ΔΨp) Controls
- Monitor ΔΨp with complementary probes like DiBAC₄(3)
- Rule out contributions from plasma potential changes to observed fluorescence signals
Specificity Controls
- Correlate fluorescence changes with functional parameters (ATP production, oxygen consumption)
- Use complementary assays to validate mitochondrial functional state
Advanced Technical Considerations
Optimizing Imaging Parameters
For reliable Rhod123 quenching mode data, specific imaging conditions should be established:
- Excitation/Emission: ≈507/529 nm (standard Rhod123 spectra)
- Acquisition Timing: For acute treatments after dye loading, dye can be washed out before imaging
- Temporal Resolution: Balance between capture speed and phototoxicity based on expected kinetics
- Environmental Control: Maintain physiological temperature and pH throughout imaging
Limitations and Alternative Approaches
While powerful, Rhod123 quenching mode has specific limitations:
- Nonlinear Response: Quenching demonstrates nonlinear concentration dependence
- Limited Temporal Window: Best suited for acute rather than chronic measurements
- Dye-Specific Artifacts: Rhod123 shows slight inhibition of electron transport chain compared to TMRM [19]
For extended temporal monitoring or more quantitative measurements, consider complementary approaches:
- Non-quenching mode with TMRM/TMRE (1-30 nM) for pre-existing ΔΨm assessment
- Ratiometric dyes like JC-1 for yes/no discrimination of polarization state
- FRET-based biosensors for specific biochemical activities [21] [22]
The quenching mode operation of Rhod123 provides researchers with a sensitive method for monitoring acute changes in mitochondrial membrane potential. The fundamental principle of high-dye concentrations leading to matrix-based aggregation and fluorescence quenching enables detection of transient mitochondrial depolarization and hyperpolarization events through unquenching and enhanced quenching responses, respectively. By implementing appropriate controls, optimization strategies, and validation protocols outlined in this guide, researchers can effectively leverage this powerful technique for investigating mitochondrial function in health and disease contexts.
Core Principles and Troubleshooting FAQs
FAQ 1: The theoretical distribution of Rhodamine 123 (Rhod123) is described by the Nernst equation. Why does the measured fluorescence in my experiment not follow the predicted linear relationship with ΔΨm?
The Nernst equation provides the fundamental thermodynamic principle for cation distribution across a membrane. However, several experimental factors cause significant deviation from the ideal Nernstian prediction in practice.
- Dye Self-Quenching: At high intramitochondrial concentrations, Rhod123 fluorescence is quenched (i.e., the fluorescence signal decreases even as more dye is accumulated). This results in a non-linear, peak-shaped relationship between dye concentration and fluorescence intensity. The peak fluorescence occurs at approximately 50 μM in aqueous solution, but the exact concentration for peak intensity in mitochondria is sensitive to experimental conditions [9].
- Dye Binding and Partitioning: Rhod123 does not remain freely dissolved in the aqueous matrix but significantly partitions into and binds to the mitochondrial membranes. This binding is temperature-dependent and means the total dye accumulation is greater than predicted by the Nernst equation for a free cation in solution [13]. The extent of binding follows the order: TMRE > Rhod123 > TMRM [13].
- Inner Filter Effect: The dye itself absorbs excitation and emission light, attenuating the detected fluorescence signal. This effect becomes more pronounced with longer light paths and higher dye concentrations, and must be corrected for using established formulas [9].
- Intracellular Probe Modification: In living cells, Rhod123 can be chemically modified, for instance, by cytochrome P450 or other enzymes, potentially converting it to a membrane-impermeable form (e.g., rhodamine 110). This can lead to the probe being trapped inside the cell or mitochondrion, causing a discrepancy between the actual membrane potential and the measured fluorescence [4].
FAQ 2: I am observing a slow fluorescence change after a rapid perturbation. Is my measurement failing to capture the true kinetics of ΔΨm?
Yes, this is a common limitation. The kinetics of the Rhod123 fluorescence signal are not instantaneous with changes in ΔΨm due to the finite time required for the dye to redistribute across the membrane.
- Limited Permeability: The rate of dye transport across the mitochondrial inner membrane limits the temporal resolution. Computational models indicate that the true characteristic response time of mitochondrial membrane potential to a change in substrate concentration can be less than 0.1 seconds. The observed transient in fluorescence intensity can be significantly slower and may not accurately reflect the rapid changes in electron transport fluxes [9].
- Recommendation: For measuring very acute changes in ΔΨm, the kinetic analysis of Rhod123 fluorescence quenching upon energization can provide a more sensitive evaluation of the membrane potential than steady-state measurements [1].
FAQ 3: Why do I see different Rhod123 fluorescence and retention between my normal and cancer cell lines?
This is a frequently observed phenomenon and is not solely due to a higher ΔΨm in cancer cells.
- Altered Efflux and Retention: Tumor cells, such as glioma cells, can exhibit a dramatically increased ability to retain Rhod123 compared to normal cells (e.g., astrocytes), even after the membrane potential is dissipated with an uncoupler like CCCP. This is likely due to reduced activity of non-specific efflux pumps (e.g., MDR proteins) or differences in the intracellular modification of the dye [4].
- Interpretation Caution: Increased fluorescence or retention in tumor cells should not be automatically interpreted as evidence of a higher ΔΨm without controlling for these alternative mechanisms [4].
The following tables consolidate key quantitative information for experimental planning and data interpretation.
Table 1: Rhodamine 123 Fluorescence and Quenching Properties
| Parameter | Value / Relationship | Experimental Context |
|---|---|---|
| Self-Quenching Peak | Fluorescence intensity peaks at ~50 μM [9]. | In aqueous solution. The peak concentration in mitochondria is condition-dependent. |
| Fluorescence-ΔΨm Relationship | Non-linear calibration curve [9]. | Sensitive to total dye and mitochondrial concentration. |
| Critical Time Constant | Mitochondrial response to substrate change < 0.1 s [9]. | True ΔΨm kinetics are faster than dye redistribution. |
Table 2: Comparative Properties of Common ΔΨm Probes
| Probe | Binding to Mitochondria (Relative Extent) | Effect on Mitochondrial Respiration | Key Characteristic |
|---|---|---|---|
| Rhodamine 123 | Intermediate (TMRE > R123 > TMRM) [13] | Suppresses respiratory control [13]. | Widely used; susceptible to self-quenching [9]. |
| TMRM | Lowest of the three [13]. | No suppression at low concentrations [13]. | Recommended for minimal interference; rationetric capability [13]. |
| TMRE | Highest of the three [13]. | Greatest suppression of respiratory control [13]. | High accumulation; greater metabolic interference. |
Essential Experimental Protocols
Protocol 1: Establishing a Calibration Curve in Isolated Mitochondria
This protocol is adapted from studies on isolated cardiac mitochondria [9].
Research Reagent Solutions:
| Reagent | Function / Explanation |
|---|---|
| Isolation Buffer | Typically contains mannitol, sucrose, EDTA, and BSA to maintain mitochondrial integrity during isolation [9]. |
| Respiration Buffer | KCl-based buffer with substrates (e.g., pyruvate, succinate) to energize mitochondria [9]. |
| Rhodamine 123 Stock | Fluorescent potentiometric probe; prepare a concentrated stock solution (e.g., 1 mM) in DMSO or water [9]. |
| ADP | Initiates State 3 respiration, causing a transient depolarization [9]. |
| CCCP (Uncoupler) | Collapses the proton gradient and ΔΨm, providing a signal for minimum fluorescence (Fmin) [9]. |
| Oligomycin | Inhibits ATP synthase; used to isolate specific proton fluxes [1]. |
Methodology:
- Mitochondrial Isolation: Isolate mitochondria from guinea pig heart (or your tissue of interest) by differential centrifugation. Determine protein concentration [9].
- Fluorescence Setup: Suspend mitochondria (e.g., 0.5 mg protein/mL) in respiration buffer with substrates (e.g., 10 mM pyruvate) in a stirred cuvette. Use a spectrofluorometer with excitation at 503 nm and emission at 527 nm [9].
- Titration and Recording: Add a low, non-quenching concentration of Rhod123 (e.g., 50 nM). Record the baseline fluorescence (Finitial).
- Induce Depolarization: Add successive small volumes of uncoupler (e.g., CCCP to 4 μM) to progressively collapse ΔΨm. After each addition, record the stable fluorescence value until a minimum value (Fmin) is reached.
- Data Analysis: The fluorescence at each point can be related to the corresponding ΔΨm. Note that the relationship is non-linear and must be modeled, accounting for self-quenching and binding, to convert fluorescence traces into accurate ΔΨm transients [9].
Protocol 2: Kinetic Analysis of Proton Flux During ATP Synthesis
This protocol uses the kinetics of Rhod123 fluorescence quenching to evaluate proton flow through F0 [1].
Methodology:
- Energization: Add a substrate (e.g., succinate) to isolated mitochondria to initiate respiration and hyperpolarize the membrane. This leads to dye uptake and fluorescence quenching.
- ADP Challenge: Add ADP to initiate State 3 respiration. Protons flow back through ATP synthase (F0) to drive ATP synthesis, causing a partial depolarization and a decrease in the rate of fluorescence quenching.
- Inhibition: Repeat the experiment in the presence of oligomycin (an ATP synthase inhibitor) or DCCD. These inhibitors block proton flow through F0, resulting in a significantly increased initial rate of fluorescence quenching upon succinate/ADP addition due to the unopposed action of the respiratory chain.
- Interpretation: The difference in the quenching kinetics in the absence and presence of oligomycin provides a quantitative measure of the proton flux through the F0 channel during ATP synthesis [1].
Signaling Pathways and Experimental Workflows
Kinetic Analysis of Proton Flux via F₀
Troubleshooting Rhod123 and ΔΨm Discrepancies
Rhodamine 123 (Rh123) is a cationic, fluorescent dye widely used for monitoring acute changes in mitochondrial membrane potential (ΔΨm). Its particular strength lies in quenching mode applications, where it enables researchers to track rapid kinetic changes in mitochondrial function in response to experimental treatments. In quenching mode, Rh123 accumulates in mitochondria at high concentrations, leading to dye aggregation and consequent fluorescence quenching. When mitochondria depolarize, dye releases into the cytoplasm causing dequenching and increased fluorescence signal - providing a sensitive readout of ΔΨm changes. This makes Rh123 particularly valuable for studying acute mitochondrial membrane dynamics in fields ranging from neurobiology to cancer research and toxicology.
Technical Specifications and Comparative Analysis
Quantitative Comparison of Mitochondrial Membrane Potential Probes
Table 1: Technical specifications and recommended usage of common ΔΨm probes
| Probe | Spectra (Ex/Em) | Primary Use Case | Recommended Concentration | Equilibration Rate | Key Advantages | Principal Limitations |
|---|---|---|---|---|---|---|
| Rhodamine 123 | 507/529 nm | Acute kinetic studies (quenching mode) | 1-10 μM (quenching) | Slow | Superior for tracking rapid ΔΨm changes; well-established protocol | Slow membrane permeation requires longer loading times |
| TMRM / TMRE | 549/575 nm | Chronic studies & pre-existing ΔΨm (non-quenching) | 1-30 nM (non-quenching); >50-100 nM (quenching) | Fast | Low mitochondrial binding & minimal ETC inhibition | Less suited for quenching studies than Rh123 |
| JC-1 | 514/529 nm (monomer); 585/590 nm (J-aggregate) | Apoptosis studies ("yes/no" polarization assessment) | 2-10 μM | Slow (aggregate form) | Ratiometric measurement (color shift) | Sensitive to factors beyond ΔΨm; photosensitive |
| DiOC₆(3) | 484/501 nm | Flow cytometry | <1 nM | Fast | Effective for population studies | Requires very low concentrations to accurately monitor ΔΨm |
Kinetic Parameters of Rhodamine Transport
Table 2: Experimentally determined kinetic parameters for Rh123 and related dyes
| Parameter | Rh123 | Rhodamine 6G | Tetramethylrosamine | Tetramethylrhodamine methyl ester |
|---|---|---|---|---|
| Passive Permeability Rate Constant (k) | Determined experimentally for each cell type | Similar to Rh123 | Similar to Rh123 | Similar to Rh123 |
| Outward Pumping Constant (kₐ) | ~10-fold lower than anthracyclines | Similar to Rh123 | Similar to Rh123 | Similar to Rh123 |
| Glutathione Dependence | Required for MRP1-mediated transport | Required for MRP1-mediated transport | Required for MRP1-mediated transport | Required for MRP1-mediated transport |
| Efflux Transporters | P-gp and MRP1 substrate | P-gp and MRP1 substrate | P-gp and MRP1 substrate | P-gp and MRP1 substrate |
Experimental Protocol: Rh123 Quenching Mode for Acute ΔΨm Changes
Workflow for Acute Kinetic Studies
Detailed Methodology
Step 1: Cell Preparation and Dye Loading
- Grow cells on appropriate imaging-compatible dishes (e.g., glass-bottom culture dishes)
- Prepare Rh123 stock solution (typically 1-10 mM in DMSO or ethanol)
- Load cells with 1-10 μM Rh123 in culture medium for 15-30 minutes at 37°C [19]
- For dissipating membrane potential effects, use HEPES/K⁺ buffer with valinomycin (10 nM) and FCCP (1 μM) [23]
Step 2: Dye Washout and Equilibrium
- Remove Rh123-containing medium
- Wash cells 2-3 times with dye-free buffer (e.g., HEPES-buffered saline)
- Incubate for additional 10-15 minutes to allow complete washout of extracellular dye
- Critical note: In quenching mode, dye should NOT remain in bath during imaging [19]
Step 3: Fluorescence Measurement and Experimental Treatment
- Set up fluorescence detection with excitation at 507 nm and emission at 529 nm
- Acquire baseline fluorescence for 2-5 minutes to establish stability
- Apply experimental treatment while maintaining continuous fluorescence recording
- For real-time monitoring, use flow cytometry or fluorescence microscopy with time-lapse capability [23]
Step 4: Controls and Validation
- Include positive controls with mitochondrial uncouplers (FCCP/CCCP, 1-10 μM)
- Use hyperpolarization controls with oligomycin (1-5 μM) to inhibit ATP synthase
- Validate specificity with pharmacological inhibitors where appropriate
Troubleshooting Guide: Common Experimental Challenges
Frequently Encountered Issues and Solutions
Table 3: Troubleshooting common problems in Rh123 quenching experiments
| Problem | Potential Causes | Solution Approaches | Preventive Measures |
|---|---|---|---|
| No fluorescence change after treatment | Insufficient dye loading; excessive extracellular dye; incorrect mode implementation | Verify dye concentration; ensure complete washout; confirm quenching mode with FCCP control | Validate protocol with positive controls in each experiment |
| Excessive background fluorescence | Incomplete washout of extracellular dye; non-specific binding | Increase wash steps; use serum-free media during loading; try lower dye concentrations | Include no-dye controls to assess background; optimize wash protocol |
| Rapid photobleaching | Excessive light exposure; high dye concentration | Reduce illumination intensity; use neutral density filters; increase camera binning | Implement minimal exposure protocols; use antifade reagents if compatible |
| Heterogeneous response between cells | Cell cycle variations; mitochondrial heterogeneity; uneven dye loading | Increase sample size; use synchronized cultures; ensure uniform dye application | Pre-screen cells for consistent morphology and growth characteristics |
| Non-specific dye modifications | Cellular metabolism of Rh123; cytochrome P450 activity | Include amiodarone to block efflux and transformation; shorten experiment duration [4] | Use fresh dye solutions; characterize dye stability in your system |
Advanced Technical Considerations
Intracellular Dye Modifications
Recent research indicates that Rh123 can undergo significant intracellular modifications over time, potentially affecting fluorescence properties. These modifications appear more pronounced in tumor cells and can be partially prevented by amiodarone, possibly through inhibition of cytochrome P450-mediated transformations or blockade of xenobiotic efflux [4]. For acute kinetic studies (typically <2 hours), this is less concerning but becomes important in prolonged experiments.
Membrane Potential vs. Proton Gradient
A critical conceptual consideration is that Rh123 measures ΔΨm (charge gradient) but does not directly report on the mitochondrial proton gradient (ΔpHm). Under certain conditions, these parameters can change in opposite directions - for example, during calcium dumping into the cytoplasm, ΔΨm may increase while ΔpHm decreases [19]. Complementary approaches using pH-sensitive dyes may be necessary for comprehensive assessment of mitochondrial bioenergetics.
Multi-drug Resistance Transporters
Rh123 is a substrate for both P-glycoprotein (P-gp) and multidrug resistance-associated protein 1 (MRP1) [23] [24]. In cells expressing high levels of these efflux transporters, dye retention may be reduced, potentially confounding results. This can be addressed by using transporter inhibitors or selecting cell lines with minimal expression of these proteins.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key reagents and their functions in Rh123-based ΔΨm studies
| Reagent / Material | Function / Application | Example Usage / Concentration |
|---|---|---|
| Rhodamine 123 | Cationic fluorescent ΔΨm probe | 1-10 μM in quenching mode; stock solutions in DMSO or ethanol |
| FCCP / CCCP | Protonophore uncouplers (positive control) | 1-10 μM to fully depolarize mitochondria |
| Oligomycin | ATP synthase inhibitor (hyperpolarization control) | 1-5 μM to induce maximal ΔΨm |
| Valinomycin | K⁺ ionophore (membrane potential control) | 10 nM with high K⁺ buffer to dissipate ΔΨ [23] |
| HEPES/K⁺ buffer | Membrane potential dissipation | Equimolar K⁺ substitution for Na⁺ with valinomycin/FCCP [23] |
| Amiodarone | Inhibitor of dye modification/efflux | 10-50 μM to reduce intracellular Rh123 transformation [4] |
| L-buthionine sulphoximine (BSO) | Glutathione depletor | 25 μM for 24h to assess glutathione dependence [23] |
FAQs: Addressing Researcher Questions
Q1: Why is Rh123 particularly suited for acute kinetic studies compared to TMRM or JC-1?
Rh123's slower equilibration kinetics make it ideal for quenching mode applications where researchers need to track rapid changes in ΔΨm. Unlike fast-equilibrating probes like TMRM, Rh123's slower membrane permeation means that quenching/unquenching changes are more easily detected and tracked over time [19]. Additionally, in quenching mode, depolarization events cause a transient increase in fluorescence (dequenching) that provides a sensitive, easily detectable signal change superior to the simple intensity decreases seen with non-quenching probes.
Q2: My Rh123 fluorescence shows an unexpected increase - does this always indicate mitochondrial depolarization?
In standard non-quenching mode, increased Rh123 fluorescence typically indicates mitochondrial hyperpolarization. However, in quenching mode (with proper dye loading and washout), increased fluorescence indicates mitochondrial depolarization as dye redistributes from mitochondria to cytoplasm, causing dequenching. Always verify you are correctly implementing quenching mode by including FCCP/CCCP controls which should produce a rapid fluorescence increase.
Q3: How long does it take for Rh123 to reach proper equilibrium for quenching experiments?
Typical loading requires 15-30 minutes at 37°C, followed by a 10-15 minute washout period to remove extracellular dye. The exact time should be determined empirically for each cell type by monitoring fluorescence stabilization. Slow equilibration is actually beneficial for acute kinetic studies as it makes the quenching/unquenching transitions more resolvable [19].
Q4: Can Rh123 be used in multi-color experiments with other fluorescent probes?
Yes, but careful spectral separation is required. Rh123 (Ex/Em: 507/529 nm) can be combined with red-emitting probes like MitoTracker Red CMXRos (Ex/Em: 579/599 nm) with appropriate filter sets. Always verify minimal spectral bleed-through by conducting single-label controls and using sequential image acquisition when possible.
Q5: What are the key limitations of Rh123 that researchers should consider?
Key limitations include: (1) susceptibility to efflux by multi-drug resistance transporters [23], (2) potential for intracellular metabolic modification over time [4], (3) measurement of ΔΨm only, not ΔpHm [19], and (4) concentration-dependent aggregation behavior that requires careful optimization. These limitations can be managed through appropriate controls and experimental design.
Executing Acute ΔΨm Experiments: A Step-by-Step Rhod123 Quenching Protocol
Frequently Asked Questions (FAQs)
Q1: What is the key difference between "quenching" and "non-quenching/redistribution" modes when using Rhodamine 123? The key difference lies in the dye concentration and the resulting fluorescence response. In quenching mode, a high dye concentration (typically ~1–10 µM) is used, leading to dye aggregation and consequent quenching of fluorescence within the mitochondria. A depolarization (loss of ΔΨm) causes dye release and a transient increase in fluorescence (unquenching). In non-quenching/redistribution mode, a low dye concentration is used to prevent aggregation, and a depolarization results in a decrease in fluorescence as the dye redistributes out of the mitochondria [25] [19].
Q2: My Rhodamine 123 signal is too low for detection. What could be the cause? Low signal can result from several factors:
- Photobleaching: Prolonged exposure to excitation light can degrade the fluorescent probe. Minimize light exposure and use neutral density filters to reduce intensity [26].
- Incorrect Dye Concentration: The concentration may be too low. Ensure you are using a sufficiently high concentration for quenching mode (e.g., 1-10 µM) [19].
- Instrument Settings: Suboptimal microscope settings, such as using an objective with a low Numerical Aperture (NA), can significantly reduce detected light. Use the highest NA objective available [26].
- Loss of Mitochondrial Potential: The cells under investigation may have a constitutively low or dissipated ΔΨm, preventing dye accumulation [25].
Q3: Upon adding Rhodamine 123, I observe an immediate, high signal that rapidly fades. Is this normal? A very high initial signal that fades quickly can indicate detector saturation. This occurs when the photomultiplier tube (PMT) is overwhelmed by the fluorescence intensity and cannot count photons linearly, leading to distorted spectra. Check that your signal intensity is below the detector's saturation threshold (often around 1.5×10⁶ counts per second for standard PMTs) and reduce the excitation light intensity or dye concentration if necessary [27].
Q4: Why is it critical to include controls like FCCP and oligomycin in my experiments? Controls are essential for validating that your fluorescence changes are due to specific changes in ΔΨm and not other artifacts.
- FCCP (A protonophore): Acts as a depolarization control. It uncouples mitochondria, collapsing ΔΨm, and should cause a large fluorescence increase in quenching mode [25] [19].
- Oligomycin (An ATP synthase inhibitor): Acts as a hyperpolarization control. By inhibiting proton flow back into the matrix, it can cause a slight hyperpolarization, which should result in a slight decrease in fluorescence in quenching mode [25] [19]. The expected response to these pharmacological agents confirms the system is functioning correctly.
Troubleshooting Guide
The following table outlines common problems, their potential causes, and solutions when performing dynamic measurements of ΔΨm using Rhodamine 123 in quenching mode.
Table 1: Troubleshooting Guide for Rhodamine 123 Quenching Mode Assays
| Problem | Potential Cause | Recommended Solution |
|---|---|---|
| No fluorescence change after an acute perturbation | Dye concentration too low for quenching mode | Increase Rhodamine 123 concentration to within the 1-10 µM range [19]. |
| Cells are not viable or mitochondria are fundamentally impaired | Assess cell viability using a viability stain (e.g., propidium iodide) [28]. Validate protocol with a positive control (e.g., FCCP) [25]. | |
| Unexpected fluorescence peaks or spectral distortion | Second-order transmission from monochromator; inner filter effect | Enable automatic filter wheels on monochromators. For inner filter effect, reduce the dye or sample concentration [27]. |
| Raman peak from solvent/buffer | Vary the excitation wavelength; a Raman peak will shift, while a true fluorescence peak will not [27]. | |
| High background fluorescence | Incomplete washing of non-specific dye | Ensure adequate washing steps with dye-free buffer after loading Rhodamine 123 and before imaging [28]. |
| Non-mitochondrial binding of dye or autofluorescence | Include a "no-dye" control to account for cellular autofluorescence. Confirm mitochondrial localization with co-staining. | |
| Signal is lost too quickly during time-lapse imaging | Photobleaching of the dye | Reduce illumination intensity and exposure time. Use a more photostable dye for very long experiments, or ensure Rhodamine 123 is protected from light during preparation and use [25] [26]. |
| Poor mitochondrial staining | Active export of dye by multidrug resistance proteins | Consider co-loading with an inhibitor like verapamil or cyclosporin H [25]. |
| Cell handling issues affecting viability | Ensure proper cell culture conditions and gentle handling to maintain viability [25]. |
Experimental Protocols
This protocol is designed for monitoring temporal changes in ΔΨm in response to acute perturbations, such as drug additions.
Research Reagent Solutions Table 2: Essential Materials and Reagents
| Item | Function/Description |
|---|---|
| Rhodamine 123 | Lipophilic cationic dye that accumulates in active mitochondria in a membrane potential-dependent manner [29]. |
| Dimethyl Sulfoxide (DMSO) | Solvent for preparing Rhodamine 123 stock and working solutions [25]. |
| FCCP | Protonophore used as a positive control for complete mitochondrial depolarization [25] [19]. |
| Oligomycin | ATP synthase inhibitor used as a control for hyperpolarization [25] [19]. |
Methodology:
- Reagent Preparation:
Cell Preparation:
- Culture cells on glass-bottom dishes or coverslips suitable for live-cell imaging.
- On the day of imaging, replace the culture medium with the Rhodamine 123 working solution.
Dye Loading and Wash:
- Incubate cells for 15-30 minutes at 37°C in the dark to allow for dye accumulation.
- Remove the dye-containing solution and wash the cells twice with a pre-warmed, dye-free imaging buffer to remove non-specific background fluorescence [28].
Image Acquisition (Dynamic Measurement):
- Place the sample on the microscope stage maintained at 37°C and 5% CO₂ (if required).
- Use an excitation wavelength of ~480 nm and collect emission at ~530 nm [28] [29].
- Acquire baseline images for 1-5 minutes.
- Without stopping acquisition, add the compound of interest (or vehicle control) and continue acquiring images to monitor the fluorescence changes over time.
Data Interpretation:
This protocol is useful for a high-throughput, population-level analysis of mitochondrial function and cell viability.
Methodology:
- Cell Preparation:
- Prepare a single-cell suspension. For sperm or cultured cells, wash with phosphate-buffered saline (PBS) and centrifuge.
- Adjust the cell concentration to approximately 5 × 10⁶ cells/mL in PBS [28].
Staining:
- Add Rhodamine 123 to the cell suspension to a final concentration of 5 µg/mL. Incubate for 5-10 minutes at 37°C in the dark.
- Wash the cells with dye-free PBS by centrifugation to remove unbound dye.
- Resuspend the cell pellet and add Propidium Iodide (PI) to a final concentration of 5 µg/mL. Incubate for an additional 5 minutes at 37°C in the dark [28].
Flow Cytometry Analysis:
- Analyze the cells immediately using a flow cytometer.
- Set the excitation to 480 nm (or a nearby blue laser).
- Detect Rhodamine 123 fluorescence (green) in the FL1 channel (e.g., 530/30 nm filter).
- Detect PI fluorescence (red) in the FL2 or FL3 channel (e.g., >670 nm filter) [28].
- Collect data for at least 10,000 events per sample.
Data Interpretation and Gating:
- Viable cells with high ΔΨm: Rh123+/PI- (High green, low red fluorescence).
- Dead/Dying cells: Rh123-/PI+ (Low green, high red fluorescence).
- Cells with low ΔΨm but intact membranes: Rh123-/PI- (Low green, low red fluorescence). This population is of particular interest in pathologies like asthenospermia [28].
Experimental Workflow and Signaling Visualization
The following diagram illustrates the logical workflow for a typical experiment designed to measure acute ΔΨm changes using Rhodamine 123 in quenching mode, highlighting key decision points.
The diagram below summarizes the relationship between mitochondrial state, dye distribution, and the resulting fluorescent signal in quenching mode, which is the core principle underlying this methodology.
FAQ: Why is using a high concentration of Rhodamine 123 critical for quenching mode experiments?
In quenching mode, a high concentration of Rhodamine 123 (Rhod123) is used to ensure the dye accumulates in the mitochondrial matrix to a point where it forms aggregates, leading to fluorescence self-quenching [2]. This phenomenon is fundamental for the technique's sensitivity. When mitochondria depolarize, Rhod123 is released from the matrix into the cytosol, where the dilution causes the fluorescence to de-quench and increase. Conversely, when mitochondria hyperpolarize, more dye is accumulated, leading to further quenching and a decrease in fluorescence [2]. This inverse relationship makes the quenching mode highly sensitive for detecting acute changes in membrane potential (ΔΨm).
FAQ: What are the consequences of using an incorrect Rhod123 concentration?
Using an incorrect concentration can lead to unreliable data and erroneous conclusions.
- Too Low a Concentration: The dye will not reach the critical concentration needed for self-quenching in the mitochondria. The fluorescence signal will be proportional to the amount of dye, behaving as in non-quenching mode. This results in a poor signal-to-noise ratio and low sensitivity to changes in ΔΨm [9] [2].
- Too High a Concentration: Excessive dye can be toxic to mitochondria, potentially suppressing respiratory control and altering the very ΔΨm you are trying to measure [13] [9]. Furthermore, extreme concentrations can lead to near-total quenching, leaving no dynamic range to detect further hyperpolarization.
Quantitative Guide to Rhod123 Concentration and Fluorescence
The relationship between Rhod123 concentration and its fluorescence intensity is not linear. The table below summarizes key data on Rhod123 behavior from experimental studies.
Table 1: Rhod123 Fluorescence Properties and Concentration Guidelines
| Parameter | Description | Experimental Context | Source |
|---|---|---|---|
| Fluorescence Peak | Intensity peaks at ~11-20 μM and then decreases due to self-quenching and inner filter effects. | Measured in aqueous solution using cuvettes with different light paths. | [9] |
| Inner Filter Effect | Attenuation of fluorescence signal due to the dye itself absorbing excitation and emission light. More significant with longer light paths. | Corrected using a formula that accounts for absorption at excitation and emission wavelengths. | [9] |
| Respiratory Suppression | Rhod123 can suppress mitochondrial respiration. Inhibition is less than TMRE but greater than TMRM. | Observed in isolated rat heart mitochondria; effect is concentration-dependent. | [13] |
| Quenching Mode | Requires a high dye concentration so that accumulated dye in the matrix is quenched. Used for monitoring rapid, robust ΔΨm changes. | A standard methodology for acute real-time monitoring of membrane potential. | [2] |
Experimental Protocol: A Step-by-Step Guide to Titrating Rhod123
The optimal high concentration for your specific experimental setup (e.g., cell type, instrumentation) must be determined empirically. The following protocol, based on established methods, will help you find this critical value [9] [30].
Principle: Directly measure the fluorescence intensity of Rhod123 at different concentrations in your experimental buffer system to identify the point where self-quenching begins to dominate, which will be your starting point for a working concentration in quenching mode.
Materials:
- Rhodamine 123 stock solution (e.g., 1 mM in DMSO)
- Experimental buffer (e.g., respiration buffer: 130 mM KCl, 5 mM K₂HPO₄, 20 mM MOPS, 2.5 mM EGTA, pH 7.15) [9]
- Fluorescence spectrophotometer or plate reader with magnetic stirring (if applicable)
- Cuvette or multi-well plate
Procedure:
- Prepare Dilutions: Create a series of Rhod123 working solutions in your experimental buffer, covering a range from low nanomolar (e.g., 50 nM) up to 50-100 μM. Ensure the concentration of DMSO from the stock is consistent and low enough to not affect the system (typically <0.1%).
- Measure Fluorescence: Load each concentration into your detection system (cuvette or well). For cuvette-based systems, note the light path, as this affects the inner filter effect [9].
- Excitation Wavelength: 503 nm
- Emission Wavelength: 527 nm
- Plot and Analyze: Graph the measured fluorescence intensity against the Rhod123 concentration. You should observe an initial near-linear increase, followed by a plateau, and then a clear decrease in intensity as concentration increases further.
- Determine Optimal Range: The concentration at which the fluorescence intensity peaks (e.g., ~20 μM for a 10 mm path length) is the point where self-quenching begins to outweigh the signal from additional dye. For quenching mode experiments, your working concentration should be at or slightly above this peak concentration [9] [2]. A common starting point is in the 10-20 μM range.
Troubleshooting Common Issues
- Problem: Poor Signal-to-Noise Ratio or Lack of Quenching
- Solution: This is likely due to insufficient dye concentration. Repeat the titration, ensuring you test high enough concentrations and confirm that your detection system is capable of observing the quenching effect.
- Problem: High Background Fluorescence or Non-Specific Staining
- Problem: Altered Mitochondrial Function or Cytotoxicity
- Solution: This indicates the dye concentration is too high. Titrate downwards to find a concentration that provides a strong quenching signal without inhibiting respiration, as TMRM has been shown to do at low concentrations [13]. Always include controls with mitochondrial uncouplers (e.g., FCCP, CCCP) and inhibitors (e.g., oligomycin) to verify that the dye is responding to genuine changes in ΔΨm [32] [2] [33].
The Scientist's Toolkit: Essential Reagents and Controls
Table 2: Key Reagents for Rhod123 Quenching Mode Experiments
| Item | Function / Purpose | Example |
|---|---|---|
| Rhodamine 123 | Lipophilic cationic fluorescent dye used as the primary ΔΨm probe. | Rhodamine-123 (Molecular Probes/Invitrogen) [9] [30] |
| Pharmacologic Uncoupler | Positive control for mitochondrial depolarization; collapses the proton gradient. | FCCP or CCCP (e.g., 4 μM) [9] [2] [33] |
| ATP Synthase Inhibitor | Positive control for mitochondrial hyperpolarization; inhibits ΔΨm consumption. | Oligomycin [32] [2] [33] |
| Ionophore / H+ Ionophore | Serves as an uncoupler to dissipate the proton motive force. | Carbonyl cyanide 3-chlorophenylhydrazone (CCCP) [9] |
| Resistance-Modifying Agent | Inhibits dye efflux by ABC transporters (e.g., P-gp) in multidrug-resistant cells. | SDZ PSC 833 [34] |
| MitoTracker Dyes | Validate mitochondrial mass and localization independently of membrane potential. | MitoTracker Deep Red [35] |
Visualizing the Rhod123 Quenching Workflow and Mechanism
The following diagram illustrates the logical workflow for determining the correct high concentration of Rhod123 and the principle of how it reports ΔΨm in quenching mode.
Diagram 1: Rhod123 titration workflow and quenching mechanism.
Research Reagent Solutions
The following table details key reagents essential for experiments investigating acute changes in mitochondrial membrane potential (ΔΨm) using Rhodamine 123 (Rhod123) in quenching mode.
| Reagent/Material | Function/Description | Key Handling & Storage Notes |
|---|---|---|
| Rhodamine 123 | Cell-permeant, cationic, green-fluorescent dye that accumulates in active mitochondria in a ΔΨm-dependent manner. [25] [36] [37] | Prepare stock solution in dry DMSO; aliquot and store at -20°C, protected from light. [25] [37] |
| Dimethyl Sulfoxide (DMSO) | Common solvent for preparing concentrated stock solutions of Rhod123 and other fluorescent dyes. [25] [37] | Hygroscopic; can carry toxins through the skin. Handle with care in a fume hood. [25] [38] |
| FCCP (Carbonyl cyanide-p-trifluoromethoxyphenylhydrazone) | Protonophore used as a positive control to collapse ΔΨm fully, validating the dye's response. [25] [32] | Toxic. Prepare concentrated stock in DMSO or ethanol. Handle with care. [25] |
| Oligomycin | ATP synthase inhibitor used to hyperpolarize mitochondria transiently by halting proton consumption. [25] [32] | Toxic. Prepare concentrated stock in DMSO or ethanol. Handle with care. [25] |
| Cell Culture Medium (Serum-free) | Buffer for preparing Rhod123 working solutions for cell staining. [37] | Must be serum-free for staining, as serum can contain components that quench fluorescence or affect dye uptake. |
Core Protocol: Utilizing Rhodamine 123 in Quenching Mode for Acute ΔΨm Changes
This protocol details the methodology for using Rhodamine 123 in quenching mode to monitor rapid, acute changes in mitochondrial membrane potential in living mammalian cells. [25]
Preparation of Reagents
Rhodamine 123 Stock Solution (1 mM):
- Dissolve 1 mg of Rhodamine 123 in 525 μL of dry DMSO to obtain a 5 mM solution. [37] Alternatively, for a 1 mM stock, dissolve 1 mg in 2.63 mL of DMSO. [25]
- Vortex thoroughly to ensure complete dissolution.
- Aliquot the stock solution into small, single-use volumes to avoid repeated freeze-thaw cycles.
- Storage: Store aliquots at -20°C, protected from light. Under these conditions, the solution is stable for up to one year. [25]
Rhodamine 123 Working Solution (1-20 μM):
- Thaw an aliquot of the 1 mM stock solution.
- Dilute the stock in serum-free cell culture medium or PBS to obtain a final working concentration between 1 and 20 μM. [37]
- Critical: The working concentration must be optimized for each cell type and instrumentation to ensure operation in quenching mode (typically leading to a higher loading concentration). [25]
- Prepare this working solution fresh immediately before use and keep it shielded from light.
Cell Staining and Live-Cell Imaging
Cell Preparation: Seed and culture cells (e.g., Primary Human Skin Fibroblasts) on sterile, glass-bottom dishes or coverslips suitable for live-cell microscopy. [25]
Dye Loading:
- Aspirate the culture medium from the cells.
- Add the pre-warmed Rhod123 working solution (1-20 μM) to completely cover the cells.
- Incubate at room temperature or 37°C for 30-60 minutes. [37]
- Critical: Perform this incubation in the dark to prevent dye photobleaching.
Washing: After incubation, carefully remove the dye-containing solution and wash the cells twice with fresh, pre-warmed serum-free medium or PBS to remove any non-specific background fluorescence. Each wash should last approximately 5 minutes. [37]
Acute Perturbation & Imaging:
- Maintain the cells in an appropriate imaging buffer.
- Acquire a stable baseline fluorescence signal for 1-2 minutes using a fluorescence microscope with the appropriate filters (Excitation: ~507 nm, Emission: ~529 nm). [36] [37]
- Inducing Acute ΔΨm Change: Add the pharmacological agent of interest (e.g., 1-2 μM FCCP to depolarize, or 1-5 μM Oligomycin to induce transient hyperpolarization) directly to the cell medium while imaging continues. [25]
- Record the temporal fluorescence changes in real-time. In quenching mode, a decrease in fluorescence intensity indicates mitochondrial depolarization, while an increase indicates hyperpolarization. [25]
Troubleshooting Guide for Rhodamine 123 Quenching Mode Assays
| Problem | Possible Cause | Recommended Solution |
|---|---|---|
| Poor mitochondrial staining / High background | 1. Multidrug resistance proteins exporting the dye. [25] 2. Incorrect dye concentration. [25] 3. Cell viability issues. | 1. Co-load with inhibitors like verapamil or cyclosporin H. [25] 2. Optimize the dye loading concentration for your specific cell type. [25] 3. Ensure proper cell handling and check viability. [25] |
| No fluorescence change upon FCCP addition | 1. Dye not operating in quenching mode. [25] 2. Inadequate FCCP concentration or activity.3. Mitochondrial dysfunction. | 1. Increase the Rhod123 loading concentration to achieve a quenched state. [25] 2. Titrate FCCP to establish a concentration that fully collapses ΔΨm. 3. Validate mitochondrial function and health using independent assays. |
| Excessive photobleaching | 1. Prolonged or high-intensity light exposure during imaging. | 1. Reduce the intensity of the excitation light or the frequency of image acquisition. 2. Include an oxygen-scavenging system in the imaging media. 3. Use a more photostable dye for long-term experiments. |
| High cytosolic background fluorescence | 1. Dye concentration is too low for quenching mode. [25] 2. Insufficient washing after loading. | 1. Systematically increase the Rhod123 working concentration until a quenched signal is achieved. [25] 2. Ensure thorough but gentle washing steps are performed after the loading incubation. [37] |
| Unexpected hyperpolarization | 1. Contamination with respiratory chain inhibitors. | 1. Use dedicated, clean glassware and solutions for mitochondrial assays to avoid cross-contamination. |
Frequently Asked Questions (FAQs)
Q1: What is the fundamental difference between quenching and non-quenching modes for Rhodamine 123? The mode is determined by the concentration of the dye loaded into the mitochondria. In non-quenching mode (lower dye concentration), the fluorescence signal is proportional to the mitochondrial accumulation of the dye, which reflects ΔΨm. In quenching mode (higher dye concentration), the dye becomes so concentrated in the mitochondria that its fluorescence self-quenches. A depolarization then causes the dye to redistribute out of the mitochondria, leading to a de-quenching and an increase in fluorescence. For acute changes, quenching mode is often used, where a depolarization is directly observed as a decrease in fluorescence intensity due to the release of the quenched dye into the cytosol. [25]
Q2: Why is DMSO the preferred solvent for stock solutions, and what are the safety considerations? DMSO is highly effective at dissolving a wide range of organic compounds, including Rhodamine 123. However, it is also a potent carrier that can rapidly transport dissolved chemicals through the skin and into the bloodstream. Therefore, always handle DMSO and DMSO-based stock solutions with gloves and other appropriate personal protective equipment in a fume hood. Prudent practice dictates assuming that any mixture will be more toxic than its most toxic component. [38]
Q3: How do I determine the correct Rhodamine 123 working concentration for my specific cell type? The optimal concentration must be determined empirically. Prepare a range of working concentrations (e.g., 1, 5, 10, 20 μM) and load your cells following the standard protocol. The correct concentration for quenching mode is one that yields a bright mitochondrial signal that then shows a strong, rapid decrease in fluorescence upon the addition of a known depolarizing agent like FCCP. If the signal increases with FCCP, the concentration is too low. [25] [37]
Q4: My positive control (FCCP) works, but my experimental treatment shows no effect. What should I check? A functioning positive control validates your entire assay system. If your treatment shows no effect, it is likely a biological reality rather than a technical failure. Ensure your treatment conditions (concentration, duration) are pharmacologically relevant. Confirm that the treatment itself does not directly interfere with the fluorescence of the dye.
Workflow and Signaling Pathways
Cell Preparation, Loading Conditions, and Incubation Parameters
The mitochondrial membrane potential (Δψm) is a key indicator of cellular health, reflecting the mitochondria's capacity to generate ATP via oxidative phosphorylation. It represents the electrical gradient across the inner mitochondrial membrane, typically accounting for 150-180 mV of the total proton electrochemical gradient potential [19]. Fluorescent dyes like Rhodamine 123 (Rhod123) are vital tools for monitoring acute changes in Δψm, as they accumulate within the mitochondrial matrix in a Nernstian fashion relative to the potential [19]. In quenching mode, Rhod123 is particularly suited for resolving rapid, acute changes in Δψm, making it invaluable for research in cell death, metabolic studies, and drug development.
Key Dyes for Measuring Δψm
Table: Common Fluorescent Probes for Assessing Mitochondrial Membrane Potential
| Probe | Best Application | Key Usage Considerations | Typical Concentration |
|---|---|---|---|
| Rhod123 | Fast-resolving acute studies (quenching mode) [19] | Slowly permeant; depolarization causes fluorescence unquenching. Slight ETC inhibition [19]. | ~1–10 μM (quenching mode) [19] |
| TMRM, TMRE | Slow-resolving acute studies or measuring pre-existing Δψm (non-quenching mode) [19] | Lowest mitochondrial binding and ETC inhibition. Fast equilibration [19]. | ~1–30 nM (non-quenching); >50-100 nM (quenching) [19] |
| JC-1 | "Yes/No" discrimination of polarization state (e.g., apoptosis) [19] | Ratiometric (monomer/aggregate); very sensitive to dye concentration and load time [19]. | Concentration-dependent |
| DiOC6(3) | Flow cytometry [19] | Requires very low concentrations (<1 nM) to monitor Δψm specifically and prevent toxicity [19]. | <1 nM [19] |
Diagram: Workflow and Signal Interpretation for Rhod123 in Quenching Mode.
Frequently Asked Questions (FAQs)
1. Why is my Rhod123 signal too weak or dim? This is commonly due to insufficient dye loading or excessive washout. Ensure you are using the recommended concentration for quenching mode (1-10 μM) and validate your loading incubation time. Also, confirm that your fluorescence detector settings (e.g., laser power, gain) are optimized for Rhod123. Check the dye stock solution for degradation by testing on cells with a known, stable Δψm.
2. I am observing an increase in fluorescence, but my positive control (FCCP) does not cause a sharp change. What is wrong? This suggests a potential issue with your control reagents or experimental conditions. First, verify the concentration and freshness of your FCCP stock. Ensure the dye is properly washed out before measurement, as its presence in the bath can alter the equilibrium. This phenomenon can also occur if the mitochondrial pool is heterogeneous, with some mitochondria depolarizing while others hyperpolarize.
3. After treatment, the fluorescence signal decreases. Does this always indicate hyperpolarization? Not necessarily. A decrease in fluorescence can indicate true hyperpolarization (increased dye uptake and quenching), but it can also result from dye leakage, cell death, or photobleaching. It is critical to run parallel assays for cell viability and to use specific inhibitors like oligomycin to confirm hyperpolarization. Always correlate fluorescence changes with other cell health markers [19].
4. How can I be sure my signal is specific to Δψm and not other factors? Δψm is not the only factor affecting cationic dye distribution. Changes in cytosolic ionic charges, particularly Ca²⁺, can also influence dye uptake and lead to misinterpretation [19]. Always include a full set of controls, including FCCP (depolarization) and oligomycin (can induce hyperpolarization), to validate your readings. For definitive proof, consider complementary assays, such as using a pH-sensitive dye to rule out contributions from the mitochondrial pH gradient (ΔpHm) [19].
Troubleshooting Guide
Table: Common Issues and Solutions when using Rhod123 in Quenching Mode
| Problem | Potential Causes | Recommended Solutions |
|---|---|---|
| No signal change upon treatment | • Incorrect dye concentration• Faulty control reagents (e.g., degraded FCCP)• Instrument detection failure | • Confirm dye stock concentration and prepare fresh.• Validate FCCP and oligomycin stocks on a control cell line.• Check instrument filters and laser functionality. |
| High background fluorescence | • Incomplete dye washout• Cell death leading to non-specific dye binding• Contaminated media or reagents | • Optimize wash protocol (number of washes, volume).• Assess cell viability before and during experiment; use viability dye.• Use filtered media and ensure sterile technique. |
| Uninterpretable or noisy data | • Cell clumping or low viability• Excessive light exposure (photobleaching)• Non-uniform cell plating | • Start with a high-viability, single-cell suspension.• Reduce exposure time and use neutral density filters.• Ensure even cell distribution when plating. |
| Signal change opposite to expected | • Contribution from non-protonic charges (e.g., Ca²⁺ overload) [19]• Misinterpretation of quenching/unquenching | • Measure cytosolic/mitochondrial Ca²⁺ levels in parallel.• Re-review the principles of quenching mode: depolarization = unquenching (signal increase). |
| Poor reproducibility between experiments | • Donor-to-donor variation (primary cells)• Slight variations in dye loading or wash timing• Fluctuations in incubation conditions (temp, CO₂) | • Use more donors or a well-characterized cell line for setup.• Standardize and meticulously time all protocol steps.• Regularly calibrate incubators and use pre-warmed media. |
Experimental Protocols
Protocol 1: Standardized Rhod123 Loading for Acute Δψm Measurements in Adherent Cells
This protocol is optimized for detecting acute changes in Δψm using Rhod123 in quenching mode [19].
Materials:
- Rhodamine 123 (Rhod123)
- Appropriate cell culture medium (without serum or Phenol Red to reduce background)
- Assay buffer (e.g., Hanks' Balanced Salt Solution, HBSS)
- Control compounds: FCCP (1-10 mM stock in DMSO) and Oligomycin (5-10 mg/mL stock in DMSO)
- Cell cultureware (e.g., 96-well black-walled, clear-bottom plates)
- Fluorescence plate reader or live-cell imaging system with FITC/GFP filter sets.
Method:
- Cell Preparation: Plate cells in a 96-well plate at an optimal density 24-48 hours before the experiment to ensure 70-90% confluency at the time of assay.
- Dye Loading:
- Prepare a 10X Rhod123 working solution in pre-warmed (37°C) assay buffer or serum-free medium to achieve a final working concentration of 1-10 μM [19].
- Remove culture medium from cells and gently add the dye-containing solution.
- Incubate for 15-30 minutes at 37°C, 5% CO₂.
- Dye Washout:
- Carefully remove the Rhod123 loading solution.
- Gently wash the cells 2-3 times with a large volume (e.g., 200 μL for a 96-well) of pre-warmed, dye-free assay buffer.
- Acute Measurement:
- Add a final volume of assay buffer or treatment solution to the wells.
- Place the plate in the pre-equilibrated (37°C) reader or imager.
- Establish a stable baseline by recording fluorescence (Ex/Em ~505/534 nm) for 1-2 minutes.
- Pause the reading, add treatment compounds (e.g., FCCP to 1-10 μM final concentration) directly to the wells, mix gently, and immediately resume recording for the desired duration (typically 10-30 minutes).
Protocol 2: Validating Δψm Specificity Using Pharmacological Controls
This protocol outlines the use of inhibitors to confirm that observed fluorescence changes are specifically due to alterations in Δψm [19] [39].
Method:
- Control Groups: For any experiment, include the following control groups in parallel:
- Vehicle Control: Cells treated with the vehicle (e.g., DMSO) used for compound dissolution.
- Full Depolarization Control: Cells treated with an uncoupler like FCCP (1-10 μM) which collapses the proton gradient, fully depolarizing mitochondria.
- Hyperpolarization Control (Optional): Cells treated with the ATP synthase inhibitor oligomycin (1-5 μg/mL), which can induce hyperpolarization by inhibiting proton reflux into the matrix.
- Expected Results:
- FCCP Treatment: A rapid and sustained increase in Rhod123 fluorescence (unquenching due to depolarization).
- Oligomycin Treatment: A rapid decrease in Rhod123 fluorescence (increased quenching due to hyperpolarization).
- Interpretation: A valid experiment requires a robust response in these control groups. The magnitude and direction of the signal in test conditions can then be reliably interpreted relative to these controls.
The Scientist's Toolkit: Essential Reagent Solutions
Table: Key Reagents for Rhod123-based Δψm Research
| Reagent / Material | Function / Role | Technical Considerations |
|---|---|---|
| Rhodamine 123 (Rhod123) | Cationic fluorescent dye that accumulates in active mitochondria in a Δψm-dependent manner. | Use at 1-10 μM for quenching mode. Less mitochondrial binding than TMRE. Check for lot-to-lot variability [19]. |
| FCCP (Carbonyl cyanide-p-trifluoromethoxyphenylhydrazone) | Proton ionophore; uncouples oxidative phosphorylation by dissipating the proton gradient. Positive control for full depolarization [19] [39]. | Typically used at 1-10 μM final concentration. Prepare fresh stock in DMSO and protect from light. |
| Oligomycin | Inhibitor of the F1/F0 ATP synthase (Complex V). Can induce hyperpolarization by preventing proton re-entry [19]. | Used at 1-5 μg/mL final concentration. Stock solution stable in DMSO at -20°C. |
| Cell Culture Medium (without Phenol Red) | Provides physiological environment during imaging without autofluorescence. | Serum can quench fluorescence; use serum-free or low-serum buffer during readings. |
| HBSS or other Physiological Buffer | A balanced salt solution for maintaining pH and osmolarity during dye loading and acute measurements. | Supplement with HEPES (10-20 mM) if working outside a CO₂ incubator to maintain pH [40]. |
| Cyclosporin A (CsA) | Specific inhibitor of cyclophilin D, used to inhibit the Mitochondrial Permeability Transition Pore (MPT) [39]. | Crucial control for determining if Δψm loss is due to MPT (CsA-sensitive). Use at 0.2-2 μM [39]. |
Diagram: Key Mitochondrial Targets and Mechanisms of Common Reagents. ETC complexes pump protons out, creating Δψm. Rhod123 influx is driven by this potential. FCCP dissipates the gradient, oligomycin blocks proton reflux, and CsA inhibits pore opening.
Technical Support Center
Troubleshooting Guides
Guide 1: Troubleshooting Rhodamine 123 Quenching Mode for Acute ΔΨm Changes
Problem: Inconsistent or Absent Fluorescence Quenching Response When adding an uncoupler like FCCP, you expect a rapid increase in fluorescence due to de-quenching. If this response is absent, weak, or inconsistent, consider the following solutions:
- Confirm Dye Concentration for Quenching Mode: Ensure the intracellular concentration of Rhodamine 123 is sufficiently high to induce self-quenching within the mitochondrial matrix. Follow established protocols precisely. In non-quenching or redistribution mode, the fluorescence intensity will drop upon uncoupler addition, whereas in quenching mode, it will rise [25] [9].
- Verify Uncoupler Efficacy and Stock Solution: Prepare fresh stock solutions of uncouplers (e.g., FCCP, CCCP) in dry DMSO and shield from light. A final concentration of 4-10 µM is typically sufficient to collapse ΔΨm. Include a positive control with a known uncoupler to confirm system functionality [41] [25].
- Check for Probe Efflux or Modification: Be aware that some cells, particularly tumor lines, possess multidrug resistance proteins that can actively export Rhodamine 123. This can lead to reduced intracellular dye concentration and delayed fluorescence responses. If suspected, co-incubate with an efflux inhibitor like verapamil or cyclosporin H. Also, note that intracellular esterases can modify rhodamine dyes, potentially trapping them and altering fluorescence properties [41].
- Optimize Microscope and Detection Settings: Use a heat filter to protect the sample. Ensure your filter cubes are appropriate for Rhodamine 123 (Excitation ~503 nm, Emission ~527 nm). For fast kinetic measurements, verify that your acquisition settings (e.g., sampling rate, exposure time) are capable of capturing the rapid fluorescence change [42] [9].
Problem: High Background or Non-Specific Staining
- Distinguish Specific from Non-Specific Signal: Include an unstained control to assess the level of cellular autofluorescence. Use a control sample treated with an uncoupler (ΔΨm collapsed) to identify non-specific dye binding [43].
- Quench Tissue Autofluorescence: If working with tissues, use commercial autofluorescence quenchers, such as TrueBlack, to suppress background signal, particularly in blue wavelengths [43].
- Ensure Thorough Washing: After dye loading, wash cells adequately with dye-free buffer to remove excess, unincorporated Rhodamine 123 that contributes to background signal [42].
Guide 2: General Fluorescence Microscopy Troubleshooting for Live-Cell Imaging
Problem: Rapid Photobleaching
- Use Antifade Reagents: Add an anti-fading mounting medium to your samples if possible [43].
- Minimize Light Exposure: Reduce light intensity where possible and use a shutter to block the excitation light when not acquiring images [42].
- Choose Photostable Dyes: Rhodamine-based dyes are generally more photostable. For other targets, select dyes known for high photostability [43].
Problem: Low Signal-to-Noise Ratio
- Optimize Optical Path: Use high-quality, high-numerical aperture objective lenses. For high-resolution work, consider oil-immersion objectives to minimize light loss. Ensure all optical components are clean and free of dust [42].
- Confirm Antibody and Dye Compatibility: In immunofluorescence, validate primary antibodies for your application and titrate for optimal concentration. Ensure secondary antibodies are compatible with your blocking serum and are highly cross-adsorbed to minimize cross-reactivity [43].
Frequently Asked Questions (FAQs)
Q1: What is the fundamental difference between using Rhodamine 123 in "quenching" versus "non-quenching/redistribution" mode, and how does this affect my experimental design? The mode depends on the intramitochondrial concentration of the dye [25] [9].
- Quenching Mode: High matrix dye concentration leads to self-quenching, causing fluorescence to be low at high ΔΨm. When ΔΨm drops, dye redistributes out of the matrix, concentration decreases, and fluorescence increases due to de-quenching. This mode offers a large fluorescence change but can itself affect ΔΨm.
- Non-Quenching/Redistribution Mode: Low matrix dye concentration avoids self-quenching. Fluorescence is high at high ΔΨm and decreases as dye leaves the mitochondria upon depolarization. This mode is less perturbing to the system but yields a smaller signal change. Your choice should be based on whether you need a large signal dynamic range (quenching) or minimal system perturbation (redistribution) for your acute measurements.
Q2: My positive control (FCCP) is not giving the expected fluorescence change. Where should I start troubleshooting? Begin by systematically checking your experimental workflow:
- Dye Stock: Is the Rhodamine 123 stock solution fresh and prepared correctly?
- Uncoupler Stock: Is the FCCP/CCCP stock solution fresh and properly diluted?
- Cell Health: Are your cells healthy? Compromised cell viability can prevent proper dye loading and mitochondrial function.
- Dye Loading: Was the dye loading time and concentration sufficient?
- Microscope Settings: Are the excitation and emission settings correct for Rhodamine 123? Is the background fluorescence excessively high?
Q3: I observe different Rhodamine 123 retention and release kinetics between normal and tumor cell lines. Why does this happen? This is a documented phenomenon. Tumor cells can exhibit increased retention of cationic dyes like Rhodamine 123, not solely due to a higher ΔΨm, but also because of differences in dye metabolism and efflux. Studies have shown that modifications of the Rhodamine 123 molecule by cellular enzymes and differences in the activity of efflux pumps can lead to delayed release of the dye in glioma cells compared to normal astrocytes, which could be misinterpreted as a sustained higher membrane potential [41].
Q4: For multi-color experiments, what precautions should I take when using Rhodamine 123 with other fluorescent probes?
- Check for Spectral Cross-Talk: Perform control experiments with each dye alone and image in all detection channels to check for bleed-through.
- Select Spectrally Separated Dyes: Choose fluorescent probes with well-separated excitation and emission spectra.
- Optimize Imaging Settings: Use sequential scanning on confocal microscopes to avoid cross-excitation and adjust emission filter cut-offs to minimize cross-talk [43].
Table 1: Key Experimental Parameters for Rhodamine 123-based ΔΨm Assays
| Parameter | Typical Range or Value | Function/Impact | Key Reference |
|---|---|---|---|
| Rhodamine 123 Working Concentration | 50 nM - 30 µM* | *Concentration-dependent; critical for establishing quenching vs. non-quenching mode. Lower for redistribution, higher for quenching. | [25] [9] |
| Uncoupler Concentration (FCCP/CCCP) | 4 - 10 µM | Collapses proton gradient, fully dissipating ΔΨm; used for positive control and calibration. | [41] [9] |
| Dye Loading Time | 15 - 30 minutes | Allows for equilibration of the dye across membranes according to ΔΨm. | [25] |
| Peak Fluorescence (in solution) | ~11-20 µM (path-length dependent) | Indicates concentration where self-quenching begins; essential for interpreting matrix dye concentration. | [9] |
| Inhibitors for Efflux (Verapamil) | 10 - 50 µM | Blocks multidrug resistance pumps to improve dye retention in problematic cell lines. | [41] [25] |
*Concentration is highly dependent on cell type, dye loading protocol, and desired operational mode (quenching vs. non-quenching).
Table 2: Troubleshooting Common Issues with Rhodamine 123 Fluorescence
| Observed Problem | Potential Causes | Recommended Solutions |
|---|---|---|
| No staining / Low signal | Incorrect dye concentration; probe efflux; dead cells; wrong filter settings. | Titrate dye; use efflux inhibitors; check cell viability; confirm microscope settings. |
| High background | Incomplete washing; autofluorescence; non-specific binding. | Increase wash steps; use autofluorescence quencher; include unstained control. |
| Unexpected dye retention after uncoupler | Dye modification by cellular esterases; active efflux mechanisms. | Use amiodarone to block modification [41]; employ efflux inhibitors. |
| Rapid signal loss (photobleaching) | Excessive light exposure; lack of antifade reagent. | Reduce light intensity/duration; use antifade mounting medium. |
Experimental Protocols
Application: Semi-quantitative comparison of ΔΨm between different cell populations or treatments.
Reagents:
- TMRM (Tetramethylrhodamine methyl ester) or Rhodamine 123
- Appropriate cell culture medium (e.g., M199 for fibroblasts)
- Dry DMSO
- Uncoupler (e.g., FCCP, 4 µM)
- Imaging buffer (e.g., Hanks' Balanced Salt Solution)
Methodology:
- Dye Preparation: Prepare a 1 mM stock of TMRM/Rhodamine 123 in dry DMSO. Aliquot and store at -20°C, protected from light. Create a working solution (e.g., 30 nM for TMRM) in buffer or medium.
- Cell Preparation: Seed cells on glass-bottom dishes (e.g., FluoroDishes) and culture until desired confluency is reached.
- Dye Loading: Incubate cells with the working solution of TMRM/Rhodamine 123 for 15-30 minutes at 37°C in a humidified incubator.
- Washing: Replace the dye-containing solution with fresh, pre-warmed imaging buffer to remove excess extracellular dye.
- Image Acquisition: Place the dish on a pre-warmed microscope stage. Use an inverted epifluorescence or confocal microscope. For TMRM/Rhodamine 123, use excitation at ~540 nm and emission detection at ~570-600 nm. Acquire images with minimal laser power to avoid phototoxicity and quenching.
- Background Correction: For each image, subtract the background fluorescence from a cell-free region.
- Stimulation & Data Analysis: Acquire a baseline. Add your stimulus (e.g., drug, metabolic inhibitor) or uncoupler (for maximum response) and continue time-lapse imaging. Analyze mean fluorescence intensity per cell or within mitochondrial regions over time.
Application: Monitoring rapid, transient changes in ΔΨm in response to acute perturbations.
Reagents:
- As in Protocol 1.
Methodology:
- High Dye Loading: Incubate cells with a higher concentration of Rhodamine 123 (e.g., 1-5 µM, requires optimization) for 20-40 minutes to achieve a matrix concentration that induces self-quenching.
- Thorough Washing: Wash cells thoroughly with dye-free buffer to ensure a low extracellular dye concentration, which promotes dye efflux upon depolarization.
- Kinetic Imaging: Set up the microscope for fast kinetic measurements. The key is to capture the rapid fluorescence increase (de-quenching) that occurs upon a drop in ΔΨm.
- Apply Acute Stimulus: After a stable baseline is recorded, rapidly add the stimulating agent (e.g., ADP to stimulate ATP synthesis, a toxin, or an uncoupler).
- Data Interpretation: In this mode, an increase in fluorescence corresponds to a decrease in ΔΨm. The kinetics of the fluorescence rise can be correlated with the kinetics of depolarization.
Signaling Pathways and Experimental Workflows
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for Rhodamine 123 ΔΨm Experiments
| Item | Function/Application | Key Considerations |
|---|---|---|
| Rhodamine 123 / TMRM | Cationic, fluorescent potentiometric probe for measuring ΔΨm. | Select based on required sensitivity and instrumentation. TMRM is often preferred for its brighter fluorescence and better retention [25]. |
| Tetramethylrodamine (TMRM/TMRE) | Alternative cationic potentiometric probe. | |
| Uncouplers (FCCP, CCCP) | Positive control; dissipates ΔΨm by shuttling protons across the IMM. | Prepare fresh stocks in DMSO; highly toxic. |
| ATP Synthase Inhibitor (Oligomycin) | Inhibits ATP synthase, causing ΔΨm to increase under certain conditions. | Useful for testing coupling between ETC and ATP synthesis. |
| Inhibitors of Efflux (Verapamil, Cyclosporin H) | Block multidrug resistance pumps to enhance dye retention in cells. | Use when dye loading is inefficient despite correct protocol [41] [25]. |
| Dry DMSO | Solvent for preparing stock solutions of dyes and inhibitors. | Use high-quality, dry DMSO to maintain reagent stability. |
| Glass-Bottom Culture Dishes | Optimal optical clarity for high-resolution live-cell imaging. | Ensure compatibility with microscope objectives (e.g., thickness). |
| Antifade Mounting Medium | Reduces photobleaching during prolonged imaging sessions. | Critical for fixed-cell imaging; some are compatible with live cells [43] [42]. |
Solving Common Problems: A Systematic Guide to Rhod123 Quenching Pitfalls
FAQ: What does "quenching mode" mean for Rhodamine 123, and why is my signal too weak?
In quenching mode, Rhodamine 123 (Rhod123) is used at high concentrations (typically 1–10 μM). At these concentrations, the dye accumulates heavily in the mitochondrial matrix, leading to dye aggregation and consequent self-quenching of its fluorescence [19]. When mitochondria depolarize (ΔΨm decreases), dye is released from the matrix into the cytoplasm, the aggregation is reduced, and fluorescence increases (unquenching). Therefore, in a properly functioning quenching experiment, a depolarization event is marked by a transient increase in fluorescence intensity [19] [44].
A weak or inconsistent signal often means this robust quenching/unquenching cycle is not being achieved, which can be due to incorrect dye concentration, improper loading, or other interfering factors.
Troubleshooting Guide: Common Causes and Solutions
The table below summarizes the primary causes of insufficient quenching signals and the recommended corrective actions.
| Potential Cause | Diagnostic Checks | Recommended Solution |
|---|---|---|
| Incorrect Dye Concentration | Verify stock concentration and final working concentration. | Use Rhod123 in the 1–10 μM range for quenching mode [19]. |
| Dye Not at Equilibrium | Check if fluorescence signal is stable before applying treatment. | Allow sufficient time for dye loading and equilibration; follow with a washout step [19]. |
| Interference from Plasma Membrane Potential (ΔΨp) | Use a dedicated probe like DiBAC4(3) to monitor ΔΨp. | Ensure changes in Rhod123 signal are not correlated with shifts in ΔΨp [19] [2]. |
| Inadequate Instrument Sensitivity | Confirm detection settings can capture small intensity changes. | Optimize microscope gain and PMT voltages; ensure a strong initial (quenched) baseline signal. |
| Loss of Mitochondrial Health | Validate with pharmacological controls (see Section 4). | Isolate mitochondria to confirm dye response in a simplified system. |
Experimental Protocol: Validating Rhod123 Quenching Mode for Acute ΔΨm Changes
This detailed protocol is designed for real-time monitoring of acute changes in ΔΨm in live cells.
Step 1: Dye Loading and Initialization
- Prepare a stock solution: Dissolve Rhod123 in DMSO to create a concentrated stock (e.g., 1 mM).
- Create working solution: Dilute the stock in your cell culture medium to a final concentration of 2–5 μM [19].
- Load the dye: Incubate cells with the working solution for 15–20 minutes at 37°C. The optimal loading time may vary by cell type and should be determined empirically.
- Washout: Replace the dye-containing medium with fresh, pre-warmed medium. This step is critical to remove extracellular dye and establish a stable baseline for imaging in quenching mode [19].
Step 2: Baseline Acquisition and Treatment
- Acquire baseline signal: Begin imaging and record the fluorescence for a few minutes to establish a stable, quenched baseline.
- Apply treatment: Introduce the experimental treatment (e.g., a drug or stressor) while continuing to record the fluorescence signal.
Step 3: Data Interpretation and Validation
- A valid depolarization (loss of ΔΨm) is indicated by a rapid increase in fluorescence (unquenching) [19] [44].
- A valid hyperpolarization (increase in ΔΨm) is indicated by a decrease in fluorescence (further quenching) [44].
Rhod123 Quenching and Unquenching Cycle
Essential Controls for Valid Interpretation
To confirm that your Rhod123 signal accurately reflects changes in ΔΨm, these pharmacological controls are mandatory [19] [2].
| Pharmacological Agent | Final Working Concentration | Expected Effect in Quenching Mode | Purpose |
|---|---|---|---|
| FCCP/CCCP | 1–4 µM | Rapid and large increase in fluorescence (unquenching) due to complete depolarization. | Positive control for depolarization; validates dye responsiveness. |
| Oligomycin | 1–10 µM | Decrease in fluorescence (increased quenching) due to hyperpolarization. | Positive control for hyperpolarization; confirms the link between ETC and ΔΨm. |
The Scientist's Toolkit: Key Research Reagents
| Reagent / Material | Function in Experiment | Key Considerations |
|---|---|---|
| Rhodamine 123 | Cationic fluorescent dye that accumulates in mitochondria in a ΔΨm-dependent manner. | Use high-purity grade. For quenching, use 1–10 µM. Aliquot and protect from light [19]. |
| FCCP / CCCP | Protonophore uncoupler; collapses the proton gradient and ΔΨm. | Prepare fresh stock in DMSO or ethanol. A final concentration of 1–4 µM is typically sufficient [9] [2]. |
| Oligomycin | ATP synthase inhibitor; causes hyperpolarization by blocking proton flow through Complex V. | Use to validate hyperpolarization. Typical working concentration is 1–10 µg/mL [19] [2]. |
| DiBAC₄(3) | Anionic dye for monitoring plasma membrane potential (ΔΨp). | Use to rule out confounding effects of changes in ΔΨp on cationic dye uptake [19] [2]. |
| MitoTracker Green / Deep Red | ΔΨm-insensitive mitochondrial dye. | Use to control for changes in mitochondrial mass, morphology, or localization [2] [44]. |
Troubleshooting Workflow Logic
What are the primary causes of high background fluorescence in flow cytometry experiments?
High background fluorescence and non-specific binding are frequently caused by issues related to antibody usage, sample composition, and staining protocol. The table below summarizes the primary causes and their respective solutions.
| Primary Cause | Specific Mechanism | Recommended Solution |
|---|---|---|
| Excess Antibody [45] [46] | High antibody concentrations promote binding to low-affinity, off-target sites. | Titrate antibodies to determine the optimal concentration [45] [47]. |
| Fc Receptor Binding [45] [46] | Fc regions of antibodies bind to Fc receptors on immune cells (e.g., neutrophils, macrophages). | Use an Fc receptor blocking reagent prior to antibody staining [45] [47]. |
| Presence of Dead Cells [45] [46] | Damaged cell membranes expose DNA, making cells "sticky" and prone to non-specific binding. | Include a viability dye (e.g., PI, 7-AAD) to identify and gate out dead cells [45] [46]. |
| Insufficient Blocking or Protein [45] [48] | Lack of protein in buffers allows antibodies to stick non-specifically to cells and surfaces. | Add protein (e.g., BSA or FBS) to staining and wash buffers [45] [46]. |
| Inadequate Washing [48] [46] | Unbound antibodies remain trapped in the sample, increasing background. | Increase washing steps and duration; consider adding a low concentration of detergent like Tween to wash buffers [46] [49]. |
| Autofluorescence [46] [47] | Some cell types (e.g., neutrophils) naturally fluoresce. | Use fluorophores that emit in the red channel (e.g., APC); use bright fluorophores to overcome autofluorescence [46] [47]. |
How can I troubleshoot high background specifically when using Rhodamine 123 (R123) in quenching mode to measure acute ΔΨm changes?
When using Rhodamine 123 in quenching mode to monitor acute changes in mitochondrial membrane potential (ΔΨm), standard flow cytometry troubleshooting principles apply, but with specific considerations for this dye and mode. The high dye concentration used in quenching mode can exacerbate several issues.
A. Causes and Solutions Specific to R123 Quenching Mode
| Problem Area | Specific Considerations for R123 Quenching Mode | Troubleshooting Action |
|---|---|---|
| Dye Concentration | The high concentration required for quenching is a common source of non-specific binding and high background [2]. | Precisely validate the minimum dye concentration needed to achieve a quenched state in your specific cell type. |
| Cell Health and Viability | Non-viable cells accumulate R123 non-specifically and show altered fluorescence patterns, such as a patchy or uniformly strong signal [50]. | rigorously exclude dead cells during analysis using a viability dye compatible with R123's emission spectrum. |
| Instrument Settings | The high fluorescent signal from a quenched dye can saturate detectors, obscuring true changes. | Optimize PMT voltages using a control sample where mitochondria have been depolarized (e.g., with FCCP). |
| Pharmacologic Controls | Without proper controls, it is impossible to confirm that signal changes are due to ΔΨm and not non-specific effects. | Always include controls with FCCP/CCCP (depolarizing agent) and oligomycin (hyperpolarizing agent) to confirm the directionality of the dye's response [2]. |
B. Experimental Protocol: Optimizing R123 Staining to Minimize Background
This protocol outlines key steps to minimize non-specific binding when using R123 in quenching mode.
1. Sample Preparation (Critical for Cell Health)
- Cell Handling: Use freshly isolated cells whenever possible. Avoid harsh vortexing or high-speed centrifugation to prevent cell lysis and the creation of debris [46] [47].
- Viability Staining: Resuspend your cell pellet in a buffer containing a viability dye (e.g., 1 µg/mL Propidium Iodide) and incubate for 5-10 minutes on ice before proceeding with R123 staining [45] [46].
2. Fc Receptor Blocking (For Immune Cells)
- If working with immune cells, incubate the cell sample with an Fc receptor blocking reagent for 10-15 minutes on ice before adding R123 [45] [47].
3. R123 Staining in Quenching Mode
- Dye Preparation: Prepare a stock solution of R123 in DMSO and dilute it in your cell culture medium or staining buffer (containing protein like BSA) to the desired high concentration for quenching. The exact concentration must be determined empirically but often ranges from 0.1 to 10 µg/mL [50] [2].
- Staining: Add the R123 working solution to the cell pellet, mix gently, and incubate for 15-30 minutes at 37°C in a water bath or incubator protected from light.
- Include Controls: In parallel, prepare and stain control samples:
- Viable Cell Control: Healthy, untreated cells.
- Depolarized Control: Cells pre-treated with 10-50 µM FCCP for 5-10 minutes prior to and during R123 staining [2].
4. Washing and Acquisition
- Washing: After incubation, pellet the cells by centrifugation. Carefully aspirate the supernatant and wash the cells twice with a large volume (e.g., 2 mL) of ice-cold PBS or staining buffer containing 0.5-1% BSA. This is critical for removing unbound dye [46].
- Resuspension: Resuspend the final cell pellet in ice-cold buffer containing a fixative like 1% paraformaldehyde if the cells are not to be acquired immediately. Note that extended fixation can increase autofluorescence [46] [47].
- Acquisition: Run samples on the flow cytometer immediately. Use the depolarized (FCCP) control to help set the voltage on the FL1 (FITC/Green) channel to ensure the signal is on-scale.
C. Workflow Diagram: Troubleshooting High Background in R123 Experiments
The following diagram outlines a logical, step-by-step workflow for diagnosing and resolving high background fluorescence.
The Scientist's Toolkit: Essential Reagents for Mitigating Non-Specific Binding
This table details key reagents used to prevent high background and non-specific binding in flow cytometry and ΔΨm experiments.
| Research Reagent | Function/Brief Explanation | Key Considerations |
|---|---|---|
| Fc Blocking Reagent | Recombinant protein that binds to Fc receptors on cells, preventing non-specific antibody binding [45]. | Essential for staining immune cells. Some antibody vendors include it in their staining buffer. |
| Viability Dyes (e.g., PI, 7-AAD) | DNA-binding dyes that are excluded from live cells with intact membranes. Used to identify and gate out dead cells [45] [46]. | Choose a dye with an emission spectrum that does not overlap significantly with your primary fluorophores. |
| Bovine Serum Albumin (BSA) | A protein added to staining and wash buffers (typically at 0.5-5%) to saturate non-specific binding sites on cells and plastic [45]. | A simple and effective way to reduce background staining across all experiments. |
| Fetal Bovine Serum (FBS) | Can be used as an alternative blocking agent to BSA, as it provides a mix of proteins [45]. | May be more physiologically relevant but composition can vary between lots. |
| Pharmacologic Controls (FCCP/CCCP) | Protonophores that dissipate the proton gradient across the mitochondrial membrane, collapsing ΔΨm. Used as a control to confirm ΔΨm-dependent dye binding [2]. | A crucial control for verifying that changes in R123 fluorescence are due to changes in ΔΨm and not other factors. |
| Pharmacologic Controls (Oligomycin) | An ATP synthase inhibitor that causes hyperpolarization of mitochondria by preventing proton re-entry, used as a control to confirm ΔΨm-dependent dye binding [2]. | Useful for demonstrating an increase in ΔΨm. |
Frequently Asked Questions (FAQs)
FAQ 1: What is the primary function of Rhodamine 123 (R123) in mitochondrial research? R123 is a cationic, fluorescent dye widely used as a sensitive probe for measuring the mitochondrial membrane potential (ΔΨm). Its accumulation in the mitochondrial matrix is driven by the highly negative inner membrane potential, and the fluorescence intensity can be quenched upon depolarization (a decrease in ΔΨm), allowing for the monitoring of acute changes in mitochondrial function [6] [5].
FAQ 2: My R123 fluorescence signal is low or absent. What could be the cause? A reduced cellular R123 staining can be caused by two main factors that must be distinguished:
- True Mitochondrial Depolarization: A genuine decrease in ΔΨm reduces the driving force for R123 accumulation [6].
- Efflux by Transport Proteins: R123 is a substrate for multidrug-resistance (MDR) transporters like P-glycoprotein (P-gp). Overexpression of these efflux pumps in certain cell types (e.g., stem cells, cancer cells) can actively export the dye, leading to low signal regardless of the actual ΔΨm [6]. It is critical to quantify Δψm in parallel experiments using alternative methods when this is suspected [6].
FAQ 3: Can R123 itself affect mitochondrial function? Yes. R123 can be used experimentally to dissipate the membrane potential. Incubation with R123 (e.g., 60 ng/μL) prior to import assays has been shown to eliminate the ΔΨm-dependent movement of proteins across the inner mitochondrial membrane [6]. Researchers should use the minimum effective dye concentration and limit incubation times to avoid unintended artifacts.
FAQ 4: How does the molecular structure of R123 influence its behavior? R123 has a cationic charge that facilitates its accumulation in the negatively charged mitochondrial matrix. Its interaction with lipid bilayers is weaker than that of structurally similar dyes like Rhodamine B (RhB), resulting in a lipid/water partition coefficient more than two orders of magnitude lower than RhB [5]. This lower affinity for membranes can influence its local concentration and accessibility to proteins like P-gp [5].
Troubleshooting Guide: Low or Unexpected R123 Signal
| Problem Phenotype | Potential Root Cause | Recommended Troubleshooting Action |
|---|---|---|
| Consistently low fluorescence across all experimental conditions. | High background activity of efflux transporters (e.g., P-gp, BCRP) [6]. | Inhibit efflux pumps with specific inhibitors (e.g., verapamil for P-gp) and re-measure. Use a cell line with known low transporter activity. |
| Signal decreases as expected with depolarizing agents, but baseline is low. | Combination of active efflux and functional ΔΨm sensing. | Confirm ΔΨm with an alternative, non-substrate dye like TMRE. Optimize dye loading concentration and time. |
| Unexpected signal increase or lack of quenching. | Saturation of the fluorescence signal; dye concentration too high. | Perform a dye titration experiment to establish the linear range of detection for your specific cell model. |
| Unspecific cellular staining; high background noise. | Non-specific binding or precipitation of the dye. | Ensure proper washing steps after dye loading. Check dye solubility and prepare fresh working solutions. |
Experimental Protocols for Key Assays
Protocol 1: Validating R123 Quenching for Acute ΔΨm Changes
Objective: To establish a robust protocol for using R123 fluorescence quenching to monitor acute drops in mitochondrial membrane potential.
Materials:
- Cell culture or isolated mitochondria
- Rhodamine 123 stock solution (e.g., 1 mM in DMSO)
- Assay buffer (e.g., KCl-based respiration buffer)
- Positive control depolarizing agent (e.g., FCCP, 1-10 μM)
- Fluorescence plate reader or fluorometer (Ex/Em: ~488 nm / ~500-530 nm) [6]
Methodology:
- Dye Loading: Incubate cells or mitochondria with a pre-determined optimal concentration of R123 (typically 0.1-10 μM) for 15-30 minutes at 37°C [6].
- Washing: Gently wash the cells/mitochondria twice with assay buffer to remove excess, unincorporated dye.
- Baseline Measurement: Transfer the suspension to a fluorescence-compatible plate or cuvette. Record the baseline fluorescence for 2-5 minutes.
- Induced Depolarization (Quenching): Add a known mitochondrial uncoupler like FCCP (1-10 μM final concentration). The subsequent dissipation of ΔΨm will cause R123 to be released from the mitochondria into the buffer, leading to a decrease in fluorescence signal (quenching).
- Data Analysis: The percentage of fluorescence quenching is calculated as
(1 - (F_after_FCCP / F_baseline)) * 100%. This value represents the acute change in ΔΨm.
Protocol 2: Controlling for P-gp Mediated Efflux
Objective: To determine if multidrug resistance transporters are interfering with R123 accumulation.
Materials:
- All materials from Protocol 1.
- P-gp inhibitor (e.g., Verapamil, 50-100 μM).
Methodology:
- Split the cell sample into two groups: "Test" and "Control."
- Pre-treat the "Test" group with the P-gp inhibitor for 20-30 minutes before R123 loading.
- Load both groups with R123 as in Protocol 1.
- Measure and compare the fluorescence intensity of both groups.
- Interpretation: A significantly higher fluorescence signal in the inhibitor-treated "Test" group compared to the "Control" group indicates that P-gp efflux was likely limiting R123 accumulation [6].
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function / Explanation |
|---|---|
| Rhodamine 123 | The core fluorescent potentiometric dye used to track changes in ΔΨm via its quenching behavior [6] [5]. |
| FCCP (Carbonyl cyanide-p-trifluoromethoxyphenylhydrazone) | A standard protonophore uncoupler used as a positive control to completely dissipate ΔΨm, validating the R123 quenching response [51] [32]. |
| Verapamil | A calcium channel blocker that also acts as a potent inhibitor of P-glycoprotein (P-gp). Used to confirm or rule out dye efflux as a confounding factor [6]. |
| Oligomycin | An ATP synthase inhibitor. Used to hyperpolarize mitochondria by preventing proton reflux, which can be used to test R123 response to the opposite ΔΨm change [32]. |
| MitoTracker Red (e.g., CMXRos) | A cell-permeant dye that covalently binds to thiol groups in mitochondria, useful for staining morphology regardless of ΔΨm, serving as a counter-stain [51]. |
Signaling Pathways and Experimental Workflows
Diagram 1: R123 Quenching in Mitochondrial Depolarization
Diagram 2: Troubleshooting R123 Signal Confounders
Signal Instability from Photobleaching and Environmental Factors
Troubleshooting Guides
Q1: Why does my Rhodamine-123 (Rhod123) signal rapidly fade during time-lapse imaging of mitochondrial membrane potential (ΔΨm)?
A: Rapid signal fading is primarily caused by photobleaching, where the fluorophore permanently loses its ability to fluoresce due to photon-induced chemical damage [52]. This occurs when the excited Rhod123 molecule interacts with molecular oxygen, generating reactive oxygen species (ROS) that degrade the dye [53]. This process is exacerbated by:
- High-intensity illumination: Common in super-resolution or prolonged time-lapse imaging [52].
- Prolonged light exposure: The rate of photobleaching is directly related to the total light dose the sample receives [53].
- Lack of oxygen-scavenging systems: The presence of molecular oxygen in the buffer accelerates photobleaching [54].
Q2: How can I distinguish between a genuine drop in ΔΨm and signal loss from photobleaching?
A: Distinguishing these is critical for accurate interpretation. The following table compares the key characteristics:
| Feature | Genuine ΔΨm Drop | Photobleaching |
|---|---|---|
| Primary Cause | Physiological or toxicological insult to mitochondria [55] | Photodamage from light exposure during imaging [52] |
| Morphology Change | May occur later in the process (e.g., during apoptosis) [55] | Often precedes signal loss; mitochondria transform from tubular to spherical, with cristae reduction [52] |
| Re-dyeing the Sample | Signal will be recovered if ΔΨm is restored | Signal loss is permanent and will not recover with fresh dye [52] |
| Use of Ratio Dyes | A change in the emission ratio (e.g., JC-1) confirms ΔΨm change [55] | Photobleaching affects both emission wavelengths, complicating ratio interpretation |
Q3: What specific morphological changes indicate that phototoxicity is affecting my experiment?
A: Phototoxicity, often linked to photobleaching, induces specific structural alterations in mitochondria that can be observed via microscopy [52]:
- Shape Change: A clear transition from elongated, tubular structures to spherical, swollen organelles [52].
- Cristae Loss: The internal cristae structure of the mitochondria deteriorates, which can be visualized with super-resolution techniques [52].
- Context: These changes are a general stress response and have also been linked to aging, apoptosis, and chemical insults, but when observed during illumination, they strongly point to phototoxicity [52].
Q4: What are the best practices to minimize Rhod123 photobleaching and phototoxicity?
A: Implement a multi-faceted approach to protect your samples:
- Optimize Imaging Parameters: Use the lowest light intensity and shortest exposure time possible to acquire a usable signal [52].
- Use Advanced Dyes: Consider next-generation dyes like "Gentle Rhodamines," which are engineered with triplet-state quenchers (e.g., cyclooctatetraene, COT) to reduce ROS generation and phototoxicity, allowing for longer time-lapse imaging [53].
- Include Protective Agents: Use photostabilizing imaging buffers that contain antioxidant systems to scavenge ROS [52].
- Validate Findings: Correlate fluorescence measurements with other parameters, such as mitochondrial morphology, to confirm that observed signal changes are not artifacts [52].
Experimental Protocols for Validation
Protocol 1: Assessing Phototoxicity via Mitochondrial Morphology
This protocol uses live-cell imaging to visualize light-induced damage.
- Cell Staining: Seed cells in a glass-bottom dish and stain with Rhod123 (e.g., 100-500 nM) in culture medium for 20-30 minutes at 37°C [55].
- Image Acquisition: Acquire a baseline image of the tubular mitochondrial network using low-light settings.
- Induction of Stress: Subject the stained cells to your standard experimental imaging regimen (light intensity and duration).
- Post-Stress Imaging: After the illumination period, re-image the mitochondria using the same low-light settings as in step 2.
- Analysis: Quantify the percentage of cells displaying spherical mitochondria versus those retaining a tubular network. A high percentage of spherical mitochondria indicates significant phototoxicity [52].
Protocol 2: Distinguishing Photobleaching from True ΔΨm Loss
This method uses a re-staining step to confirm the cause of signal loss.
- Initial Staining and Imaging: Stain cells with Rhod123 and perform your time-lapse experiment until significant signal fading is observed.
- Wash and Re-stain: Gently wash the cells with fresh, dye-free medium to remove the bleached Rhod123. Re-incubate the cells with a fresh batch of Rhod123 using the original staining protocol [55].
- Final Imaging: Image the cells again using the same acquisition settings.
- Interpretation:
- If the fluorescence signal returns to baseline levels, the initial signal loss was likely due to genuine ΔΨm depolarization that has since recovered, and the new dye has accumulated normally.
- If the fluorescence signal remains low, the initial signal loss was likely due to photobleaching of the dye, and the mitochondria may be too damaged to re-accumulate the fresh dye properly [52].
Research Reagent Solutions
The following table lists key reagents used in troubleshooting Rhod123 signal instability.
| Reagent / Material | Function / Explanation |
|---|---|
| Gentle Rhodamine GR555-mito | A COT-conjugated TMRM derivative that shows a 5-fold reduction in phototoxicity, ideal for long-term ΔΨm imaging [53]. |
| MitoTracker Green (MTG) | A common mitochondrial structure dye; reported to have lower phototoxicity compared to NAO in side-by-side tests [52]. |
| Tetramethylrhodamine, Ethyl Ester (TMRE) | A voltage-sensitive dye similar to Rhod123; often used as a more photostable alternative for ΔΨm measurement [52] [55]. |
| JC-1 | A ratiometric, voltage-sensitive dye that shifts emission from green (~520 nm) to red (~590 nm) as ΔΨm increases. The ratio is less susceptible to artifacts from dye concentration and photobleaching [35] [56] [55]. |
| 10-N-Nonyl Acridine Orange (NAO) | A cardiolipin-binding structure dye for cristae imaging; known to cause significant phototoxicity and membrane potential loss under illumination and should be used with caution [52]. |
| Oxygen Scavenging System (OSS) | A chemical system (e.g., glucose oxidase + catalase) added to imaging buffer to reduce local oxygen, thereby slowing photobleaching and phototoxicity [54]. |
| Photostabilizing Buffer | Commercial buffers designed to extend fluorophore longevity by reducing ROS and free radicals generated during illumination [52]. |
Visualization of Photobleaching Mechanisms and Workflows
Photobleaching and Phototoxicity Pathways
This diagram illustrates the molecular mechanisms leading to photobleaching and phototoxicity of Rhodamine dyes.
Experimental Workflow for Diagnosis
This workflow provides a step-by-step guide to diagnose the root cause of Rhod123 signal instability.
Troubleshooting Guides
Guide 1: Addressing Inconsistent Fluorescence Signals in Quenching Mode
Problem: Unexpectedly high or low Rhodamine 123 (Rho-123) fluorescence intensity during acute ΔΨm measurements, making data interpretation difficult.
Explanation: In quenching mode, fluorescence intensity is inversely related to mitochondrial membrane potential (ΔΨm) because dye accumulation in the mitochondrial matrix leads to self-quenching [9]. Higher ΔΨm drives more Rho-123 into mitochondria, causing more quenching and lower fluorescence signal. Several factors can disrupt this relationship.
Solution: Follow this systematic troubleshooting workflow:
Detailed Troubleshooting Steps:
Verify Dye Concentration and Working Solution:
- Issue: Rho-123 fluorescence exhibits concentration-dependent self-quenching, peaking around 50 μM and decreasing at higher concentrations [9].
- Action: Confirm stock solution concentration and prepare fresh working solutions in 1% (v/v) methanol in HBSS for optimal fluorescence (λex = 505 nm, λem = 525 nm) [57]. Ensure final assay concentration is appropriate for your experimental setup.
Check for Efflux Transporter Activity:
- Issue: Rho-123 is a known substrate for multidrug efflux transporters like P-glycoprotein (P-gp) [58] [59] and uptake transporters like Organic Cation Transporters (OCTs) [60]. Transporter activity can significantly alter intracellular dye availability independent of ΔΨm.
- Action: Use specific transporter inhibitors as controls (e.g., Tariquidar for P-gp [59]) to assess their contribution. Be aware that some compounds, like Cannabidiol (CBD), may also inhibit P-gp-mediated Rho-123 efflux [59].
Confirm Mitochondrial Specificity and Dye Localization:
- Issue: Fluorescence changes may not reflect ΔΨm if the dye is sequestered in other cellular compartments or metabolized.
- Action: Use imaging to confirm mitochondrial localization. Note that Rho-123 can be metabolized to Rho-110, which has different spectral properties [57]. In tumor cells, increased dye retention and metabolism may occur [41].
Validate with Pharmacological Controls:
- Issue: Without proper controls, fluorescence changes cannot be confidently attributed to ΔΨm.
- Action: Include controls with mitochondrial uncouplers (e.g., FCCP/CCCP) to dissipate ΔΨm and confirm an increase in fluorescence, and inhibitors (e.g., oligomycin) to induce hyperpolarization and confirm decreased fluorescence [2] [9].
Guide 2: Managing Cell-Type Specific Variability in Rho-123 Loading
Problem: Significant differences in Rho-123 loading and retention between different cell types, particularly between normal and tumor cells.
Explanation: Tumor cells often exhibit altered mitochondrial physiology, increased expression of drug efflux transporters, and different metabolic activities that affect Rho-123 handling independent of ΔΨm [41].
Solution:
Pre-characterize Cell Lines:
Optimize Loading Protocols by Cell Type:
- For cells with high efflux activity, consider pre-incubation with transporter inhibitors or using modified loading conditions (e.g., milder temperatures).
- Establish cell-type specific loading concentrations and times empirically.
Account for Non-ΔΨm Dependent Retention:
Guide 3: Mitigating Efflux Transporter Interference in Rho-123 Assays
Problem: Efflux transporters actively remove Rho-123 from cells, reducing signal intensity and masking true ΔΨm changes.
Explanation: P-glycoprotein (P-gp/ABCB1), multidrug resistance-associated proteins (MRPs), and other ATP-binding cassette (ABC) transporters recognize Rho-123 as a substrate and pump it out of cells [58] [61] [59]. This creates competing pathways that oppose mitochondrial dye accumulation.
Solution: Follow this decision pathway to identify and mitigate transporter interference:
Key Experimental Modifications:
Use Specific Transporter Inhibitors:
Include Appropriate Controls:
- Always include inhibitor-only controls to assess their direct effects on ΔΨm
- Compare fluorescence patterns with and without inhibitors
Consider Alternative Dyes:
- For systems with high efflux activity, consider dyes less susceptible to transporter interference (e.g., TMRM, TMRE) [2]
Frequently Asked Questions (FAQs)
Q1: Why does my Rhodamine 123 fluorescence sometimes decrease instead of increase when mitochondria depolarize?
This paradoxical response typically indicates significant efflux transporter activity. When mitochondria depolarize, Rho-123 is released into the cytosol where it becomes accessible to efflux transporters like P-gp, which rapidly pump it out of the cell. This net efflux can overwhelm the expected fluorescence increase from de-quenching [59]. Solution: Repeat experiments with specific transporter inhibitors (e.g., Tariquidar for P-gp) to confirm this mechanism.
Q2: How do I determine whether fluorescence changes are truly due to ΔΨm changes and not other factors?
Always implement a comprehensive control strategy [2]:
- Pharmacological controls: Use FCCP/CCCP (uncouplers) to collapse ΔΨm and oligomycin (ATP synthase inhibitor) to induce hyperpolarization
- Transporter controls: Include specific transporter inhibitors to assess their contribution
- Technical controls: Verify dye concentration linearity and account for inner filter effects
- Parallel assays: Correlate with other mitochondrial parameters when possible (e.g., oxygen consumption)
Q3: Are there specific cell types where Rhodamine 123 is particularly problematic for ΔΨm measurements?
Yes, several cell types present challenges:
- Cancer cells: Often overexpress P-gp and other efflux transporters [41] [6]
- Primary hepatocytes and renal cells: High expression of OCTs that transport Rho-123 [60]
- Multidrug-resistant cell lines: Selected for high efflux transporter expression
- Stem cells: Often exhibit strong dye efflux capability through ABC transporters like ABCG2 [6]
Q4: What are the critical differences between quenching and non-quenching modes for Rho-123?
Table: Comparison of Rhodamine 123 Quenching vs. Non-Quenching Modes
| Parameter | Quenching Mode | Non-Quenching Mode |
|---|---|---|
| Dye Concentration | Higher (micromolar range) | Lower (nanomolar range) |
| Signal Relationship to ΔΨm | Inverse (increased potential = decreased fluorescence) | Direct (increased potential = increased fluorescence) |
| Primary Application | Acute ΔΨm changes (Scenario 2) [2] | Chronic treatments & slower acute changes (Scenario 1) [2] |
| Key Advantage | Monitors rapid, robust changes | Linear relationship to potential |
| Main Limitation | Nonlinear response | Lower signal intensity |
| Self-Quenching | Pronounced at matrix concentrations [9] | Minimized |
| Best for Transport Studies | Less suitable | More suitable |
Q5: Can hypoxia affect my Rhodamine 123 measurements independently of ΔΨm?
Yes. Hypoxia can induce P-gp overexpression through HIF-1α signaling, potentially increasing Rho-123 efflux independent of actual ΔΨm changes [59]. This is particularly relevant in studies of cancer cells, ischemic models, or any experiments involving oxygen limitation.
Research Reagent Solutions
Table: Essential Reagents for Troubleshooting Rhodamine 123 Experiments
| Reagent/Category | Specific Examples | Function/Application | Key Considerations |
|---|---|---|---|
| Transporter Inhibitors | Tariquidar (P-gp), MK-571 (MRP), Verapamil (OCT) | Identify/block specific efflux pathways | Use most specific inhibitors available; Tariquidar preferred for P-gp due to high specificity [59] |
| Metabolic Modulators | FCCP/CCCP (uncouplers), Oligomycin (ATP synthase inhibitor) | Control and validate ΔΨm changes | Essential for confirming dye response direction [2] [9] |
| Alternative Dyes | TMRM, TMRE, JC-1 | Cross-verification and specialized applications | TMRM/TMRE have different transporter susceptibilities; JC-1 provides ratio-metric measurement [2] |
| Cell Line Controls | P-gp overexpressing lines, Parental counterparts | Validate transporter effects | Critical for establishing assay specificity [61] |
| Solvents & Buffers | 1% methanol in HBSS [57] | Optimal dye solvent | Maintain consistent solvent composition across experiments |
Experimental Protocols
Protocol 1: Validating Specificity of Rho-123 ΔΨm Measurements
Purpose: To confirm that observed fluorescence changes primarily reflect ΔΨm rather than transporter activity or other confounding factors.
Materials:
- Rho-123 stock solution (prepared in 1% methanol in HBSS)
- Transporter inhibitors: Tariquidar (1 μM), Verapamil (10-50 μM)
- Mitochondrial modulators: FCCP (2-5 μM), Oligomycin (1-5 μg/mL)
- Appropriate cell culture reagents and fluorescence detection system
Procedure:
- Cell Preparation: Plate cells in appropriate multi-well plates or culture dishes 24-48 hours before experiment
- Inhibitor Pre-treatment: Pre-incubate cells with relevant transporter inhibitors for 30-60 minutes
- Dye Loading: Load cells with Rho-123 at optimized concentration for 15-30 minutes
- Baseline Recording: Record baseline fluorescence for 5-10 minutes
- Pharmacological Challenges:
- Apply FCCP to collapse ΔΨm - expect fluorescence increase in quenching mode
- Apply Oligomycin to induce hyperpolarization - expect fluorescence decrease
- Data Interpretation:
- Compare response magnitude with and without transporter inhibitors
- Significant differences indicate transporter interference
Expected Outcomes: In the absence of significant transporter interference, FCCP should cause rapid fluorescence increase, while oligomycin should cause decrease. Altered responses with inhibitors suggest transporter contribution.
Protocol 2: Quantitative Assessment of P-gp-Mediated Rho-123 Efflux
Purpose: To specifically quantify P-glycoprotein contribution to Rho-123 efflux in your experimental system.
Materials:
- Rho-123 stock solution
- Tariquidar (specific P-gp inhibitor)
- Control and test cell lines
Procedure:
- Cell Loading: Load parallel samples of cells with identical Rho-123 concentrations
- Inhibitor Application: Add Tariquidar (final concentration 1 μM) to test samples
- Efflux Phase: Monitor fluorescence decrease over time as dye effluxes
- Data Analysis:
- Compare efflux rates between treated and untreated samples
- Calculate percentage inhibition of efflux by Tariquidar
- Use mathematical modeling if quantitative values needed [9]
Interpretation: Significant reduction in efflux rate with Tariquidar indicates substantial P-gp contribution. If no effect observed, consider other transporters (MRPs, OCTs) or passive diffusion as primary efflux mechanisms.
Ensuring Data Integrity: Controls, Correlative Assays, and Advanced Techniques
FAQ: The Role of Pharmacological Controls in Rhodamine-123 Experiments
Why are FCCP and oligomycin considered essential pharmacological controls for Rhodamine-123 (Rhod123) experiments?
FCCP and oligomycin are indispensable because they produce predictable, opposite effects on the mitochondrial membrane potential (ΔΨm), allowing you to verify that your Rhod123 signal is truly reporting changes in ΔΨm and not an artifact.
- FCCP/CCCP acts as a protonophore, making the mitochondrial inner membrane permeable to protons. This dissipates the proton gradient, collapsing ΔΨm and causing Rhod123 to be released from the mitochondria. In quenching mode, this release leads to a marked increase in fluorescence (unquenching) [19] [2] [1].
- Oligomycin inhibits the F1F0 ATP synthase (Complex V). By blocking the primary pathway for proton re-entry into the matrix, it causes ΔΨm to increase (hyperpolarization). In quenching mode, this leads to greater Rhod123 uptake and aggregation, resulting in a decrease in fluorescence (further quenching) [32] [1].
The table below summarizes the expected responses:
Table 1: Expected Fluorescence Responses with Pharmacological Controls in Rhod123 Quenching Mode
| Pharmacological Control | Mechanism of Action | Effect on ΔΨm | Effect on Rhod123 Fluorescence (Quenching Mode) |
|---|---|---|---|
| FCCP / CCCP | Protonophore, dissipates proton gradient | Collapse (Depolarization) | ↑ Increase (Unquenching) |
| Oligomycin | Inhibits ATP synthase (Complex V) | Increase (Hyperpolarization) | ↓ Decrease (Further Quenching) |
What should I do if my controls do not show the expected response?
If the controls fail, it indicates a problem with the experimental setup, the health of the mitochondria, or the interpretation of the signal.
- Verify mitochondrial health: Ensure your isolated mitochondria or cells are functionally intact. Check the Respiratory Control Index (RCI) for isolated preparations [9].
- Confirm reagent integrity and concentration: Prepare fresh stocks of FCCP and oligomycin. Test different concentrations to ensure efficacy (e.g., a common final concentration for FCCP is 1-4 µM [9]).
- Check the dye concentration and loading: Rhod123 self-quenching peaks around 50 µM [9]. Using the correct concentration for quenching mode (typically 1-10 µM [19]) is critical. Ensure dye loading has reached equilibrium.
- Rule out off-target effects: Be aware that changes in the plasma membrane potential (ΔΨp) can influence cationic dye uptake. Use a probe like DiBAC4(3) to monitor ΔΨp if this is a concern [19] [2].
FAQ: Optimizing the Rhodamine-123 Quenching Assay
My Rhod123 fluorescence signal is weak. What could be the cause?
A weak signal can stem from several factors:
- Insufficient dye loading: Ensure an adequate incubation time and concentration. Verify that multidrug resistance transporters are not actively exporting the dye; co-loading with an inhibitor like verapamil may be necessary [25] [2].
- Excessive inner filter effect: This occurs when high dye concentrations absorb the excitation and emission light, attenuating the signal. This is highly dependent on the cuvette's light path [9]. Use a cuvette with a shorter path length or dilute the sample.
- Low mitochondrial mass or unhealthy mitochondria: Confirm the protein concentration in your assays and assess mitochondrial health via a respiratory control experiment [9].
I observe a high background signal. How can I reduce it?
High background is often due to incomplete washout of non-mitochondrial dye in a post-load/wash protocol [19]. Ensure you perform thorough washing steps with a dye-free buffer after the loading period. For experiments where the dye is maintained in the bath, high background may indicate that the dye concentration is too high for the non-quenching mode.
Experimental Protocol: Validating Acute ΔΨm Changes with Pharmacological Controls
This protocol is designed for monitoring acute changes in ΔΨm in a suspension of isolated mitochondria using Rhod123 in quenching mode.
Materials and Reagents
- Isolation or respiration buffer (e.g., 130 mM KCl, 5 mM K₂HPO₄, 20 mM MOPS, 2.5 mM EGTA, pH 7.15 [9])
- Rhodamine-123 stock solution (e.g., 1 mM in DMSO)
- Substrate (e.g., 10 mM sodium pyruvate or succinate)
- ADP (e.g., 200-400 µM for state-3 respiration)
- Pharmacological controls: Oligomycin (e.g., 1-5 µg/mL), FCCP (e.g., 1-4 µM)
- Spectrofluorometer with magnetic stirrer and temperature control
Procedure
- Prepare mitochondrial suspension: Suspend isolated mitochondria (e.g., 0.5 mg protein/mL) in respiration buffer in a cuvette [9].
- Load dye: Add Rhod123 to a final quenching-mode concentration (e.g., 1-10 µM [19]). Allow the signal to stabilize.
- Energize mitochondria: Add a substrate (e.g., pyruvate/succinate). Observe a rapid decrease in fluorescence as mitochondria energize and take up the dye (quenching).
- Acute perturbation test: Once the signal is stable, add your experimental compound and record the fluorescence transient.
- Apply pharmacological controls:
- Oligomycin control: To the energized state, add oligomycin. Expect to see a decrease in fluorescence due to hyperpolarization [1].
- FCCP control: At the end of the experiment, add FCCP. Expect to see a rapid increase in fluorescence to the level observed before energization, confirming complete depolarization [9] [19].
Diagram: Experimental Workflow for Validating Acute ΔΨm Changes
The Scientist's Toolkit: Essential Reagents for Rhod123 ΔΨm Research
Table 2: Key Research Reagents for Mitochondrial Membrane Potential Studies
| Reagent | Function | Key Considerations |
|---|---|---|
| Rhodamine-123 | Cationic, fluorescent ΔΨm probe. Accumulates in mitochondria, quenches at high concentrations. | Ideal for kinetic studies of acute changes [19]. Use 1-10 µM for quenching mode [19]. Subject to self-quenching and inner filter effects [9]. |
| FCCP / CCCP | Proton ionophore; uncoupler used as a depolarization control. | Dissipates ΔΨm by equalizing proton gradient. Final concentration typically 1-4 µM [9]. |
| Oligomycin | ATP synthase inhibitor; used as a hyperpolarization control. | Blocks proton flow through Complex V, increasing ΔΨm. Final concentration typically 1-5 µg/mL. |
| ADP | Substrate for ATP synthase; induces State 3 respiration. | Used to test coupled respiration. A pulse of ADP causes a transient depolarization (unquenching) that recovers [9] [1]. |
| Substrates (e.g., Pyruvate, Succinate) | Provide electrons to the Electron Transport Chain (ETC) to energize mitochondria. | Essential for generating ΔΨm. Choice of substrate (complex I vs. II) can influence results. |
Advanced Troubleshooting: Beyond Basic Controls
My FCCP addition only causes a partial unquenching. What does this mean?
Partial unquenching suggests that your mitochondria were not fully polarized to begin with, or that a subpopulation has lost membrane integrity.
- Investigate mitochondrial coupling: Test the respiratory control ratio (RCR) to ensure your mitochondria are healthy and well-coupled.
- Check for non-specific dye binding: Rhod123 can partition into mitochondrial membranes, which may contribute to a non-ΔΨm-sensitive fluorescence signal [9].
- Confirm FCCP potency and solubility: Ensure your FCCP stock is fresh and properly dissolved in ethanol or DMSO.
How can I be sure that a change in fluorescence is due to ΔΨm and not other factors like mitochondrial morphology or mass?
This is a critical consideration for accurate interpretation.
- Image mitochondrial morphology: Use complementary dyes like MitoTracker (which are less dependent on ΔΨm in fixed cells) to visualize and quantify mitochondrial mass and network structure [35] [2].
- Perform numerical analysis: Calculate the coefficient of variance of pixel intensities within mitochondrial regions; higher variance often indicates a healthy, polarized network, while a homogenized signal can indicate depolarization [2].
- Use complementary assays: Correlate your Rhod123 findings with measurements of oxygen consumption rate (OCR) or cellular ATP levels to build a cohesive bioenergetic profile [32] [2].
Diagram: Interpreting Rhod123 Fluorescence in the Context of Mitochondrial Function
FAQs: Resolving Key Experimental Challenges
Q1: My Rhodamine 123 fluorescence quenching data does not correlate with oxygen consumption rate (OCR) measurements. What could be the cause?
Several factors could cause this discrepancy:
- Dye Self-Quenching: At high intramitochondrial concentrations, R123 exhibits self-quenching, where fluorescence decreases without a change in membrane potential (ΔΨm). This can be misinterpreted as hyperpolarization. The fluorescence intensity peaks at concentrations near 50 μM and then decreases [9].
- Inner Filter Effects: The absorption of incident or emitted light by the R123 itself can artificially reduce the detected fluorescence signal. This trivial quenching can be identified by checking for progressive shifts in emission spectra and mitigated by using cuvettes with shorter path lengths [62] [9].
- Improper Calibration: The relationship between R123 fluorescence intensity and ΔΨm is non-linear and sensitive to experimental conditions like total dye and mitochondrial concentrations. A quantitative model that accounts for dye transport and self-quenching is often necessary for accurate interpretation [9].
Q2: How can I validate that my observed Rhodamine 123 quenching is truly reporting on ΔΨm and not an artifact?
A robust validation requires multiple approaches:
- Pharmacological Controls: Use inhibitors and uncouplers to manipulate ΔΨm. The addition of oligomycin (inhibits ATP synthase) should increase the quenching rate, while the uncoupler FCCP should rapidly collapse ΔΨm and cause fluorescence recovery. The absence of these expected responses indicates an artifact [17].
- Lifetime Measurements: Fluorescence lifetime measurements are a key diagnostic. Dynamic quenching (true response to ΔΨm) shortens the fluorescence lifetime, while static quenching or inner filter effects do not. A discrepancy between intensity-based and lifetime-based measurements indicates a non-ΔΨm-related quenching mechanism [62].
- Cross-Validation with Independent Assays: Correlate R123 quenching kinetics with direct measurements of mitochondrial function, such as OCR using a Seahorse XF Analyzer or a Clark electrode. A concerted response to substrates and inhibitors across these platforms validates the findings [63] [64].
Q3: When performing combined OCR and fluorescence assays, what are the critical parameters to ensure data quality?
- Cell/Organism Number and Size: The number of cells or organisms used per assay is critical. Using too few may result in a signal too low to detect, while overloading can lead to hypoxia and inaccurate measurements. Consistency in biological material size and number is essential for reproducibility [63].
- Elimination of Food Source: When working with whole organisms like C. elegans, the presence of bacteria from their food source can significantly consume oxygen and confound the host's OCR measurement. It is crucial to wash the animals thoroughly before the assay [63].
- Sequential Inhibitor Injection: The mitochondrial stress test relies on sequential drug injections. The concentrations and order of compounds (e.g., oligomycin, FCCP, rotenone/antimycin A) must be optimized for the specific cell type or model organism to reliably reveal parameters like ATP-linked respiration and maximal respiratory capacity [64].
Troubleshooting Guide: Common Problems and Solutions
Table 1: Troubleshooting R123 Quenching and Correlation Experiments
| Problem | Potential Cause | Solution |
|---|---|---|
| Low signal-to-noise ratio in R123 traces | • Dye concentration too low.• Photobleaching. | • Titrate R123 concentration (start with 50 nM) [9].• Reduce exposure time/intensity; use a more photostable dye. |
| No recovery of fluorescence after FCCP addition | • Insufficient FCCP concentration.• Non-specific dye binding or toxic effects. | • Titrate FCCP to determine optimal concentration for your system.• Include a positive control with a known uncoupler; ensure mitochondrial viability. |
| Discrepancy between ΔΨm and OCR data | • Dye self-quenching or inner filter effects.• Different time scales of response. | • Measure fluorescence lifetime to confirm dynamic quenching [62].• Model the dye kinetics; the metabolic response to ADP occurs on a sub-second scale [9]. |
| High variability in OCR measurements | • Inconsistent sample loading (cell number).• Contamination from food bacteria. | • Normalize OCR to protein content or cell number.• Implement rigorous washing protocols for whole organisms [63]. |
| Lack of response to oligomycin in Seahorse assay | • Faulty drug preparation or concentration.• Compromised mitochondrial membrane integrity. | • Confirm drug stocks are fresh and properly dissolved.• Check mitochondrial isolation procedure or cell health; validate with a positive control. |
Experimental Protocols for Key Methodologies
Protocol 1: Evaluating Acute ΔΨm Changes Using R123 Quenching Kinetics
This protocol uses the kinetics of R123 fluorescence quenching to assess proton flux through the F0 channel of ATP synthase during ATP synthesis [17].
Research Reagent Solutions: Table 2: Essential Reagents for R123 Quenching Assays
| Reagent | Function | Working Concentration |
|---|---|---|
| Rhodamine 123 (R123) | Fluorescent potentiometric probe that accumulates in energized mitochondria. | 50 nM - 1 μM [17] [9] |
| Oligomycin | Inhibits F0 subunit of ATP synthase, blocking proton flow back into the matrix. | 1 - 5 μg/mL [17] |
| FCCP | Proton ionophore uncoupler; collapses ΔΨm. Positive control for fluorescence recovery. | 0.2 - 4 μM [17] [9] |
| DCCD (Dicyclohexylcarbodiimide) | Inhibits proton flow through the F0 channel. Similar effect to oligomycin. | Subsaturating concentrations [17] |
| Succinate | Complex II substrate to energize mitochondria. | 10 mM [17] |
| ADP | Substrate for ATP synthesis; induces State 3 respiration and proton flux through F0. | Determine empirically [17] |
Methodology:
- Isolate Mitochondria: Isolate mitochondria (e.g., from rat liver or guinea pig heart) using differential centrifugation in an appropriate isolation buffer. Maintain on ice and use within 8 hours, confirming a high Respiratory Control Index (RCI > 8) [9].
- Set Up Fluorescence Measurement: Suspend mitochondria (e.g., 0.5 mg protein/mL) in respiration buffer (e.g., 130 mM KCl, 5 mM K₂HPO₄, 20 mM MOPS, pH 7.15) in a fluorometer cuvette with continuous stirring [9].
- Add R123: Add R123 to the suspension and set fluorometer excitation/emission wavelengths to 503/527 nm [9].
- Energize Mitochondria: Add succinate (10 mM) to initiate electron transport and build ΔΨm, observed as a rapid quenching of R123 fluorescence [17].
- Initiate ATP Synthesis & Proton Flux: Add a pulse of ADP. This induces State 3 respiration, allowing protons to flow back into the matrix through F0 to drive ATP synthesis, resulting in a partial recovery (de-quenching) of fluorescence as ΔΨm transiently decreases [17].
- Apply Inhibitors: In a parallel experiment, pre-incubate with oligomycin (or DCCD) before adding ADP. This blocks the F0 proton channel, preventing the ADP-induced fluorescence de-quenching and resulting in a greater net quenching rate [17].
- Data Analysis: Compare the initial rates and extent of fluorescence quenching in the absence and presence of oligomycin. The difference reflects the magnitude of proton flux through F0 during ATP synthesis.
Protocol 2: Profiling Mitochondrial Function via Oxygen Consumption Rate (OCR)
This protocol uses a Seahorse XF Analyzer to measure OCR in living cells, providing a direct readout of mitochondrial function that can be correlated with fluorescence data [64].
Methodology:
- Cell Culture: Seed cells into a Seahorse XF24 cell culture microplate and culture for 24-48 hours to reach an appropriate density (e.g., 20,000-50,000 cells/well, optimized for each type).
- Prepare Assay Medium: On the day of the assay, replace growth medium with unbuffered assay medium (e.g., DMEM, pH 7.4). Incubate the cell plate in a non-CO₂ incubator for 45-60 minutes to allow temperature and pH equilibration.
- Load Compounds: Load mitochondrial inhibitors into the injection ports of the Seahorse sensor cartridge:
- Port A: Oligomycin (e.g., 1-5 μM).
- Port B: FCCP (e.g., 0.5-2 μM; titrate for optimal concentration).
- Port C: Rotenone (e.g., 0.5-1 μM) and Antimycin A (e.g., 0.5-1 μM).
- Run Assay: Calibrate the sensor cartridge and begin the assay. The instrument will measure the basal OCR, then sequentially inject the compounds, measuring the OCR after each injection.
- Data Analysis: Calculate key bioenergetic parameters from the OCR profile [64]:
- Basal Respiration: OCR before any injections.
- ATP-linked Respiration: The drop in OCR after oligomycin injection.
- Maximal Respiration: The OCR after FCCP injection.
- Proton Leak: The OCR remaining after oligomycin (non-ATP-linked).
- Non-mitochondrial Respiration: The OCR remaining after rotenone/antimycin A.
Visualizing the Experimental Workflow and Data Interpretation
The following diagram illustrates the logical workflow for integrating R123 quenching assays with OCR measurements to obtain a comprehensive view of mitochondrial function.
Diagram 1: Integrated Bioenergetics Assessment Workflow
This diagram outlines the relationship between membrane potential, the mechanisms affecting R123 fluorescence, and the resulting experimental readout.
Diagram 2: R123 Signal Interpretation Logic
FAQ: What is the fundamental difference between quenching and non-quenching mode?
The core difference lies in the concentration of the dye used and how the resulting fluorescence signal corresponds to changes in mitochondrial membrane potential (ΔΨm).
Quenching Mode: Uses high dye concentrations (typically 1–10 µM). At these concentrations, the dye accumulates densely in the mitochondrial matrix, leading to self-quenching (a reduction in fluorescence intensity). A depolarization (decrease in ΔΨm) causes the dye to be released from the mitochondria into the cytosol, resulting in de-quenching and an increase in fluorescence. A hyperpolarization (increase in ΔΨm) causes more dye to be taken up, leading to more quenching and a decrease in fluorescence [19] [65].
Non-Quenching Mode: Uses low dye concentrations (e.g., 10–30 nM). The dye accumulation is low enough to avoid self-quenching. In this mode, an increase in ΔΨm causes more dye to accumulate in the mitochondria, leading to an increase in fluorescence. A decrease in ΔΨm causes dye release and a decrease in fluorescence [19] [32].
The table below summarizes the key operational differences.
| Feature | Quenching Mode | Non-Quenching Mode |
|---|---|---|
| Dye Concentration | High (∼1–10 µM) [19] | Low (∼10–30 nM) [19] |
| Signal Response to Depolarization | Fluorescence Increases (de-quenching) [19] [65] | Fluorescence Decreases [19] [32] |
| Signal Response to Hyperpolarization | Fluorescence Decreases (increased quenching) [19] | Fluorescence Increases [19] [32] |
| Best For | Acute, dynamic changes in ΔΨm [19] | Measuring steady-state ΔΨm; chronic studies [19] |
| Key Consideration | Signal is non-linear and highly sensitive to experimental conditions like dye and mitochondrial concentration [9] | Requires dye to remain in bath during imaging if treatment precedes loading [19] |
The relationship between dye concentration and fluorescence intensity is non-linear. The following diagram illustrates the fundamental mechanisms of each mode.
Experimental Design & Protocol Guidance
FAQ: How do I decide which mode to use for my specific experiment?
Your choice should be driven by the biological question and the temporal resolution you require. Here is a practical guide.
FAQ: What is a standard protocol for using Rhodamine-123 in quenching mode?
The following table outlines a typical workflow for monitoring acute ΔΨm changes in isolated mitochondria or cell cultures using quenching mode [9] [19] [30].
| Step | Procedure | Key Parameters & Tips |
|---|---|---|
| 1. Dye Loading | Incubate cells or mitochondrial suspension with 1–10 µM Rhodamine-123 [19]. | Optimize concentration and time for your cell type. Perform at 37°C for cells, room temperature for isolated mitochondria [30]. |
| 2. Washing | After loading, wash cells/suspension to remove all extracellular dye. | Critical step. Any residual dye in bath will confound the de-quenching signal upon depolarization [19]. |
| 3. Baseline Recording | Place sample in fluorometer or imager. Record fluorescence (Ex/Em: ~503/527 nm) for a stable baseline [9]. | Use appropriate controls (e.g., baseline, CCCP/FCCP for full depolarization) [9] [19]. |
| 4. Apply Treatment | Introduce the experimental stimulus (e.g., ADP, drug, toxin). | Ensure rapid and homogenous mixing for consistent kinetics [9]. |
| 5. Data Interpretation | Monitor fluorescence kinetics. Increase = Depolarization. Decrease = Hyperpolarization. | Remember the signal is inverse. Use model-based analysis for quantitative ΔΨm transients where possible [9]. |
Troubleshooting Common Issues
FAQ: I am not seeing a clear signal in quenching mode. What could be wrong?
| Problem | Potential Cause | Solution |
|---|---|---|
| Weak or No Signal | Incomplete washing after dye loading [19]. | Increase number and volume of washes. Confirm no background fluorescence in buffer. |
| Dye concentration is too low for quenching [9]. | Titrate dye concentration (1, 2, 5, 10 µM) to find the optimal range for your system. | |
| Inner filter effect in dense suspensions [9]. | Use a cuvette with a shorter excitation path length (e.g., 2 mm instead of 10 mm) to mitigate inner filtering [9]. | |
| Inconsistent Results Between Experiments | Variations in total dye and mitochondrial/cell concentration [9]. | Precisely standardize protein/cell count and dye concentration across preparations. |
| Dye-induced toxicity inhibiting respiration [13]. | Use the lowest possible dye concentration that still gives a robust quenching signal. TMRM may be less inhibitory [13]. | |
| Signal Goes in the Opposite Direction Than Expected | Misinterpretation of the inverse signal in quenching mode. | Remember: Fluorescence ↑ = Depolarization; Fluorescence ↓ = Hyperpolarization. Validate with control compounds (FCCP/CCCP should cause a large increase) [19]. |
| Dye concentration is in an intermediate, non-linear range. | Perform a concentration titration to ensure you are firmly in the quenching regime [9]. |
FAQ: My fluorescence signal is unstable. How can I improve the quality of my recordings?
- Minimize Photobleaching: Rhodamine dyes are light-sensitive. Reduce laser power or exposure time during live-cell imaging, and use an anti-fade mounting medium if working with fixed samples [30].
- Account for Non-Protonic Charges: ΔΨm dyes measure charge gradient, not specifically the proton gradient. Changes in cytosolic Ca²⁺ or K⁺ can significantly affect ΔΨm without a change in proton motive force. Use parallel assays (e.g., with a pH-sensitive dye like SNARF-1) to dissect these effects [19].
- Validate with Pharmacological Controls: Always include controls to verify your signal is specific to ΔΨm.
- FCCP/CCCP (2-4 µM): Protonophores that collapse the proton gradient, causing full depolarization (in quenching mode: large fluorescence increase) [9] [19].
- Oligomycin (1-5 µg/mL): ATP synthase inhibitor. In coupled mitochondria, this hyperpolarizes ΔΨm by halting proton consumption (in quenching mode: fluorescence decrease) [19].
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Tool | Function | Considerations for Use |
|---|---|---|
| Rhodamine-123 | Cationic fluorescent dye used to track dynamic changes in ΔΨm [9] [19]. | Preferred for acute studies in quenching mode due to slower permeation, making fluorescence changes easier to resolve [19]. |
| TMRE / TMRM | Alternative cationic dyes for ΔΨm measurement [19] [13]. | Preferred for non-quenching mode and chronic studies due to lower mitochondrial binding and less inhibition of the electron transport chain (ETC) [19] [13]. |
| FCCP / CCCP | Proton ionophores; positive controls that completely collapse ΔΨm [9] [19]. | Used to validate the dye's response. Final concentration typically 2-4 µM [9]. |
| Oligomycin | Inhibitor of ATP synthase (Complex V) [19] [32]. | Causes hyperpolarization in coupled mitochondria by preventing ΔΨm consumption. Used at 1-5 µg/mL [19]. |
| BSA (Fatty Acid Free) | Component of isolation and respiration buffers [9]. | Binds free fatty acids and other contaminants, helping to maintain mitochondrial integrity and function during experiments [9]. |
A technical support guide for troubleshooting mitochondrial membrane potential measurements.
Why Dye Selection Matters for Δψm Assessment
The mitochondrial membrane potential (Δψm) is a key indicator of cellular health, reflecting the capacity of mitochondria to generate ATP through oxidative phosphorylation. Its dissipation is a central event in apoptosis and bioenergetic dysfunction. Fluorescent cationic dyes are the primary tools for monitoring Δψm, yet each probe has distinct strengths and limitations. Relying on a single dye can lead to misinterpretation, whereas a strategic combination of TMRM and JC-1 provides a more robust, validated assessment of mitochondrial status, strengthening the conclusions of your research [19] [66].
Comparative Dye Profiles: TMRM vs. JC-1
The table below summarizes the core characteristics and recommended applications for TMRM and JC-1 to guide your experimental design.
| Probe | Primary Strength | Mechanism of Action | Typical Working Concentration | Best Suited For |
|---|---|---|---|---|
| TMRM / TMRE | Detecting subtle, acute changes in Δψm [19] | Nernstian distribution; fluorescence intensity correlates with Δψm [19] [66]. Can be used in non-quenching or quenching modes. | 1–30 nM (non-quenching); >50–100 nM (quenching) [19] | Slow-resolving acute studies; measuring pre-existing Δψm; long-term imaging with low phototoxicity [19]. |
| JC-1 | Discriminating "Yes" or "No" polarization states [19] | Δψm-dependent formation of J-aggregates. Emits at different wavelengths as a monomer (green, ~529 nm) vs. aggregate (red, ~590 nm) [66]. | Manufacturer's protocol; sensitive to concentration | Apoptosis studies; flow cytometry; endpoint assays where a ratiometric (red/green) measure is beneficial [19]. |
The following workflow outlines a decision path for integrating these dyes into your experimental strategy, particularly when troubleshooting Rhod123-based findings:
Detailed Experimental Protocols
Protocol 1: Using TMRM for Acute Δψm Measurements
TMRM (Tetramethylrhodamine Methyl Ester) is ideal for tracking dynamic changes in membrane potential over time with minimal artifacts.
- Dye Preparation: Dissolve TMRM in DMSO to create a 5 mM stock solution. Dilute this stock in serum-free culture medium or PBS to prepare a working solution between 1–20 μM. For non-quenching mode, use the lowest possible concentration that yields a detectable signal, typically resulting in a final concentration of 1–30 nM in the well [19] [67].
- Cell Staining (Adherent Cells):
- Culture cells on sterile coverslips.
- Aspirate the culture medium and add enough TMRM working solution to cover the cells.
- Incubate at room temperature or 37°C for 30–60 minutes.
- Wash the cells twice with fresh medium or PBS to remove excess, non-specific dye.
- For non-quenching mode, the dye can be maintained in the bath during imaging to allow for re-equilibration during live-cell experiments [19].
- Data Acquisition & Analysis: Image using a fluorescence microscope with TRITC/Rhodamine filters (Ex/Em ~550/576 nm). In non-quenching mode, a decrease in fluorescence intensity indicates mitochondrial depolarization, while an increase indicates hyperpolarization.
Protocol 2: Using JC-1 for Ratiometric Assessment
JC-1 provides an internal ratio that is largely independent of mitochondrial mass, dye concentration, and cell size, making it excellent for endpoint assays.
- Dye Preparation: Prepare JC-1 according to the manufacturer's instructions. It is critical to optimize the loading concentration and time for your specific cell type, as the formation of J-aggregates is highly sensitive to these parameters [19].
- Cell Staining:
- Load cells with the optimized JC-1 working solution.
- Incubate for 30–45 minutes at 37°C.
- Wash cells thoroughly to remove excess dye. Note that JC-1 should ideally remain in the bath during imaging to prevent fluorescence changes from probe redistribution [19].
- Data Acquisition & Analysis: Image using both FITC (Ex/Em ~510/530 nm) and TRITC (Ex/Em ~560/590 nm) filter sets to detect monomer (green) and J-aggregate (red) forms, respectively. Calculate the red-to-green fluorescence intensity ratio. A high ratio indicates polarized mitochondria, while a low ratio indicates depolarization.
Troubleshooting FAQs
FAQ 1: My TMRM signal is too low or absent for detection. What should I check?
Low TMRM signal is a common issue. Follow this checklist to identify the source of the problem:
- Confirm Dye Viability and Concentration: Ensure your stock solution is fresh and properly stored. Perform a concentration titration (e.g., 1, 10, 20 nM) to find the optimal signal-to-noise ratio for your cells.
- Verify Mitochondrial Depolarization: Treat cells with a mitochondrial uncoupler like FCCP (e.g., 1–10 µM) as a control. A significant drop in fluorescence confirms that the dye is responding to Δψm [19].
- Check Instrument Settings: Use visual inspection to ensure the excitation beam is correctly aligned with the sample. Confirm you are using the correct excitation/emission settings (Ex ~550 nm, Em ~576 nm) [27] [43]. For weak signals, increase the detector integration time or PMT voltage.
- Rule out Photobleaching: Include an antifade agent in your mounting medium and minimize light exposure during sample preparation and imaging [43].
FAQ 2: My JC-1 ratio is low, but I see strong green fluorescence. What does this mean, and how can I confirm my results?
A low red/green ratio with strong green signal is a classic signature of mitochondrial depolarization, as JC-1 remains in its monomeric form. To confirm this result and rule out technical artifacts:
- Validate with TMRM: Perform the same experimental treatment on a separate sample and stain with TMRM. A correlated decrease in TMRM fluorescence confirms depolarization.
- Check JC-1 Loading Conditions: A low ratio can also result from using a dye concentration that is too low or an incubation time that is too short for J-aggregate formation. Re-optimize your loading protocol [19].
- Use a Positive Control: Include a control with a known apoptosis inducer (e.g., staurosporine) to verify that the JC-1 assay correctly reports depolarization in your system.
FAQ 3: I am observing high background and non-specific staining with my dyes. How can I reduce this?
High background can obscure genuine signals and lead to incorrect conclusions.
- Optimize Washing: Increase the number and volume of washes after dye loading. Ensure the wash buffer is rocking freely to remove all unbound dye.
- Titrate Antibody Concentrations: If performing immunostaining alongside Δψm measurement, high antibody concentrations can cause non-specific binding. Perform a titration to find the optimal concentration [43].
- Account for Autofluorescence: Include an unstained control to determine your sample's intrinsic autofluorescence level. Cellular autofluorescence is often high in blue/green wavelengths, so using red-shifted dyes like TMRM can help. Specific autofluorescence quenchers are also available [43].
- Avoid the Inner-Filter Effect: For any fluorescence measurement, ensure your sample absorbance is below 0.1 to prevent the re-absorption of emitted light, which distorts spectra and quenches signal [68].
Research Reagent Solutions
A carefully selected toolkit is essential for reliable Δψm measurement. The table below lists key reagents and their functions.
| Reagent / Tool | Function in Δψm Research |
|---|---|
| TMRM / TMRE | Lipophilic cationic dye for tracking acute, dynamic changes in Δψm via fluorescence intensity. Preferred for low phototoxicity and minimal binding to mitochondria [19] [66]. |
| JC-1 | Cationic dye for ratiometric (red/green) assessment of mitochondrial health, ideal for confirming large-scale depolarization events like apoptosis [19] [66]. |
| FCCP | Protonophore uncoupler that dissipates the proton gradient and collapses Δψm. Serves as a critical positive control for depolarization [19]. |
| Oligomycin | ATP synthase inhibitor that causes Δψm to increase (hyperpolarization) by blocking proton flow back into the matrix. A key control for validating dye response [19]. |
| Cyclosporin A (CsA) | Inhibitor of Cyclophilin D, used to confirm the involvement of the Mitochondrial Permeability Transition Pore (MPTP) in depolarization events [39]. |
| SNARF-1 | Ratiometric pH-sensitive dye. Used to measure mitochondrial pH (ΔpHm), demonstrating that Δψm and ΔpHm can change independently [19]. |
Technical Support Center: Troubleshooting Rhod123 Quenching Mode for Acute ΔΨm Changes
Frequently Asked Questions (FAQs)
Q1: My Rhod123 fluorescence signal is not quenching upon the addition of an uncoupler (e.g., FCCP). What could be wrong? A: This indicates the assay is not capturing the acute depolarization of ΔΨm. Potential causes and solutions are detailed in the troubleshooting guide below.
Q2: Why is the baseline fluorescence so high and variable between replicates? A: High baseline often indicates incomplete dye loading, non-quenching conditions (low dye concentration), or excessive background fluorescence from dead cells or debris. Ensure you are using a sufficiently high Rhod123 concentration (typically 1-5 µM) and that you wash cells thoroughly post-loading to remove extracellular dye.
Q3: After a treatment, I see a drop in Rhod123 fluorescence. Can I directly interpret this as mitochondrial depolarization? A: Not necessarily. A drop in fluorescence can indicate either depolarization (de-quenching) or a loss of mitochondrial mass/number. You must confirm results with complementary assays, such as measuring mitochondrial mass (e.g., with Citrate Synthase activity) or cell viability. Rhod123 fluorescence is a component of the health picture, not the sole readout.
Q4: My positive control (FCCP) works, but my experimental compound shows no effect. Does this mean it doesn't impact mitochondria? A: Not necessarily. The compound may be affecting other aspects of mitochondrial physiology (e.g., ATP production, ROS generation, Ca²⁺ buffering) without causing an acute, large-scale depolarization. It is crucial to integrate other assays to build a complete profile of mitochondrial function.
Troubleshooting Guide
| Symptom | Possible Cause | Solution |
|---|---|---|
| No quenching with uncoupler | 1. Rhod123 concentration too low.2. Cells are already fully depolarized.3. Uncoupler is inactive or wrong concentration. | 1. Increase Rhod123 to 2-5 µM.2. Check cell health and pre-treat with an oxidizable substrate (e.g., pyruvate).3. Prepare fresh FCCP stock and titrate (typically 1-5 µM final). |
| High, variable baseline fluorescence | 1. Incomplete washing.2. Excessive cell death.3. Plate reader optics/calibration. | 1. Increase wash steps (2-3x with buffer).2. Check viability; reduce stress during handling.3. Clean plate bottom, calibrate instrument. |
| Fluorescence decreases over entire experiment | 1. Photobleaching.2. Gradual loss of dye from mitochondria. | 1. Reduce excitation light intensity/intensity.2. Include a time-matched vehicle control to distinguish specific effects from drift. |
| Poor signal-to-noise ratio | 1. Low cell seeding density.2. High background from media/components. | 1. Optimize cell number for your plate format.2. Use a clear, phenol-red-free assay buffer. |
Experimental Protocol: Acute ΔΨm Change Assay Using Rhod123 Quenching
Principle: In healthy, polarized mitochondria, Rhod123 accumulates and self-quenches. Depolarization causes the dye to release into the cytoplasm, de-quench, and increase fluorescence.
Materials:
- Cells cultured in a 96-well black-walled, clear-bottom plate.
- Rhod123 stock solution (1 mM in DMSO).
- Assay Buffer (e.g., Krebs-Ringer Bicarbonate Buffer).
- Positive Control: Carbonyl cyanide-p-trifluoromethoxyphenylhydrazone (FCCP, 50 µM stock in DMSO).
- Plate reader capable of fluorescence top/bottom reading (Ex/Em: 505/534 nm).
Procedure:
- Cell Preparation: Seed cells 24-48 hours prior to achieve 70-90% confluency at assay time.
- Dye Loading:
- Prepare a 2 µM Rhod123 solution in pre-warmed assay buffer from the 1 mM stock.
- Aspirate culture media from wells and add 100 µL of the 2 µM Rhod123 solution.
- Incubate for 15-30 minutes at 37°C in the dark.
- Washing:
- Carefully aspirate the dye-containing buffer.
- Gently add 100 µL of fresh, pre-warmed assay buffer to each well. Repeat this wash step twice.
- After the final wash, add 100 µL of fresh assay buffer.
- Fluorescence Measurement:
- Place the plate in a pre-warmed (37°C) plate reader.
- Monitor baseline fluorescence for 5-10 minutes (read every 1-2 minutes).
- Pause the reading. Automatically add 10-20 µL of your experimental compound or vehicle control (e.g., 0.1% DMSO) to the wells. Gently mix if possible.
- Resume reading for 20-30 minutes.
- At the endpoint, add 10 µL of 50 µM FCCP (5 µM final concentration) to all wells to achieve full depolarization and record the maximum de-quenching signal.
Data Analysis:
- Normalize fluorescence values (F) to the average baseline fluorescence before compound addition (F₀).
- Calculate the percentage of maximal depolarization using the FCCP-induced fluorescence (F_max).
% Depolarization = [(F - F₀) / (F_max - F₀)] * 100
Table 1: Typical Fluorescence Responses in a Rhod123 Quenching Assay
| Condition | Expected Fluorescence Response | Interpretation of Mitochondrial Status |
|---|---|---|
| Baseline (Polarized) | Low, Stable | Healthy, high ΔΨm (dye is quenched) |
| Acute Depolarization (e.g., FCCP) | Rapid Increase | Collapse of ΔΨm (dye de-quenches) |
| Hyperpolarization (e.g., Oligomycin) | Slight Decrease | Increased ΔΨm (increased quenching) |
| Cell Death / Permeabilization | Slow, Gradual Increase | Non-specific; loss of dye sequestration |
Table 2: Key Reagent Concentrations for Rhod123 Quenching Assay
| Reagent | Typical Stock Concentration | Final Working Concentration | Function |
|---|---|---|---|
| Rhod123 | 1 mM in DMSO | 1 - 5 µM | Potentiometric fluorescent dye |
| FCCP (Uncoupler) | 50 µM in DMSO | 1 - 5 µM | Positive control; collapses ΔΨm |
| Oligomycin | 10 mM in DMSO | 1 - 10 µM | ATP synthase inhibitor; can induce hyperpolarization |
| Assay Buffer | N/A | N/A | Provides ionic and metabolic environment |
The Scientist's Toolkit
| Item | Function |
|---|---|
| Rhodamine 123 (Rhod123) | Cationic, fluorescent dye that accumulates in active mitochondria in a ΔΨm-dependent manner. Exhibits fluorescence quenching at high matrix concentrations. |
| Carbonyl cyanide-p-trifluoromethoxyphenylhydrazone (FCCP) | Protonophore uncoupler that dissipates the proton gradient across the inner mitochondrial membrane, serving as a reliable positive control for complete depolarization. |
| Oligomycin | ATP synthase inhibitor. Can be used to induce a state of mitochondrial hyperpolarization by preventing proton re-entry through the synthase. |
| Black-walled, clear-bottom microplate | Minimizes cross-talk and background fluorescence while allowing for microscopic confirmation of cell health. |
| Phenol-red-free Assay Buffer | Eliminates background fluorescence from pH indicators commonly found in cell culture media. |
Visualizations
Rhod123 Quenching Under High ΔΨm
Rhod123 De-quenching Under Low ΔΨm
Rhod123 Quenching Assay Workflow
Conclusion
Successfully employing Rhodamine 123 in quenching mode for acute ΔΨm measurements demands a rigorous blend of theoretical understanding and practical optimization. By mastering the quenching mechanism, adhering to robust protocols, systematically troubleshooting artifacts, and employing essential validation controls, researchers can transform Rhod123 from a simple fluorescent dye into a powerful, reliable tool. This disciplined approach is paramount for generating high-quality data that can accurately inform our understanding of mitochondrial biology in fundamental research and advance the development of therapeutics targeting mitochondrial dysfunction in diseases ranging from neurodegeneration to cancer.
References
- 1. evaluation of proton flux through F(0) during ATP synthesis [https://pubmed.ncbi.nlm.nih.gov/14507434/]
- 2. Mitochondrial membrane potential probes and the proton ... [https://www.bohrium.com/paper-details/mitochondrial-membrane-potential-probes-and-the-proton-gradient-a-practical-usage-guide/811679850480795648-4675]
- 3. studies using rhodamine-123 fluorescence unquenching [https://pubmed.ncbi.nlm.nih.gov/14600273/]
- 4. Is the Mitochondrial Membrane Potential (∆Ψ) Correctly ... [https://www.mdpi.com/1422-0067/23/1/482]
- 5. Interactions between Rhodamine Dyes and Model ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8876248/]
- 6. Rhodamine 123 - an overview [https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/rhodamine-123]
- 7. Mitochondrial membrane hyperpolarization modulates ... [https://www.nature.com/articles/s41467-025-59427-5]
- 8. Mitochondrial dysfunction mediated by ER-calcium ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12305934/]
- 9. Mitochondrial Inner Membrane Electrophysiology Assessed ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3508792/]
- 10. Picture perfect: Imaging mitochondrial membrane potential ... [https://www.sciencedirect.com/science/article/abs/pii/S0014483520305765]
- 11. Measurement of the Absolute Magnitude and Time Courses of ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0159199]
- 12. Fluorescence microscopy imaging of mitochondrial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10326048/]
- 13. Measurement of mitochondrial membrane potential using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1302536/]
- 14. Rhodamine 123 as a probe of transmembrane potential in ... [https://www.sciencedirect.com/science/article/abs/pii/000527288690112X]
- 15. Rhodamine 123 [https://en.wikipedia.org/wiki/Rhodamine_123]
- 16. Spontaneous Changes in Mitochondrial Membrane ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1303613/]
- 17. evaluation of proton flux through F0 during ATP synthesis [https://www.sciencedirect.com/science/article/pii/S0005272803001105]
- 18. What is fluorescence quenching? [https://axispharm.com/what-is-fluorescence-quenching/]
- 19. Mitochondrial membrane potential probes and the proton ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3115691/]
- 20. Quenching (fluorescence) [https://en.wikipedia.org/wiki/Quenching_(fluorescence)]
- 21. Methodology and Applications of Förster Resonance ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5800528/]
- 22. Basics of FRET Microscopy [https://www.microscopyu.com/applications/fret/basics-of-fret-microscopy]
- 23. Kinetic Analysis of Rhodamines Efflux Mediated by the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1303372/]
- 24. Rhodamine 123 - an overview [https://www.sciencedirect.com/topics/chemistry/rhodamine-123]
- 25. Visualization of mitochondrial membrane potential in ... [https://www.sciencedirect.com/science/article/abs/pii/S0091679X19301207]
- 26. Fluorescence Microscopy Errors [https://evidentscientific.com/en/microscope-resource/knowledge-hub/photomicrography/fluorescenceerrors]
- 27. Troubleshooting Measurements of Fluorescence Spectra [https://www.edinst.com/blog/fluorescence-spectra-measurements/]
- 28. Evaluation of sperm mitochondrial function using rh123/PI ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3596687/]
- 29. LumiTracker® Mito Rhodamine 123 | CAS#:62669-70-9 [https://www.lumiprobe.com/p/rhodamine-123-mitochondrial-dye?region_id=2]
- 30. Rhodamine Staining Tips for Reliable Cell Imaging [https://probes.bocsci.com/resources/rhodamine-staining-protocols-tips-for-high-quality-and-reproducible-results.html]
- 31. sciencedirect.com/topics/medicine-and-dentistry/rhodamine-123 [https://www.sciencedirect.com/topics/medicine-and-dentistry/rhodamine-123]
- 32. Measuring mitochondrial membrane potential | The EMBO ... [https://link.springer.com/article/10.1038/s44318-025-00632-9]
- 33. Comprehensive Guide to Mitochondrial Function Assays [https://www.elabscience.com/resources/cell-metabolism-technical-topics/1954?srsltid=AfmBOorMntOHsjoGHkFgyAO__-Ozkq-Cfqn3hFWdN0fBorYazAXFOYhc]
- 34. SDZ PSC 833 and SDZ 280-446 are the most active of various... [https://pubmed.ncbi.nlm.nih.gov/1421439/]
- 35. Methods to Evaluate Changes in Mitochondrial Structure and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10177605/]
- 36. Rhodamine 123, 25 mg 25 mg | Buy Online | Invitrogen™ [https://www.thermofisher.com/order/catalog/product/R302]
- 37. Rhodamine 123 (RH-123) | Fluorescent Dye [https://www.medchemexpress.com/Rhodamine_123.html?srsltid=AfmBOoobHCD45CA3Dqh0yvIaysuB_vXNmsNoZLvH394ojp9NbJABW5Ae]
- 38. Working with Chemicals - Prudent Practices in the Laboratory [https://www.ncbi.nlm.nih.gov/books/NBK55872/]
- 39. Inhibition of the Mitochondrial Permeability Transition for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3364550/]
- 40. Best Practices for Preparing and Maintaining Cell Culture ... [https://gmpplastic.com/blogs/useful-articles-on-lab-supplies-faq-section/best-practices-for-preparing-and-maintaining-a-cell-culture-environment?srsltid=AfmBOorfG8tCVltZQxkFrtrH2cS1gaeMW_iOVUtouuzB5rzFu-uDfVoY]
- 41. Is the Mitochondrial Membrane Potential (∆Ψ) Correctly ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8745654/]
- 42. Cellular Fluorescence Microscopy Troubleshooting & Best ... [https://www.aatbio.com/resources/application-notes/cellular-fluorescence-microscopy-troubleshooting-best-practices]
- 43. Troubleshooting Tips for Fluorescence Staining [https://biotium.com/tech-tips-protocols/troubleshooting-tips-for-fluorescence-staining/]
- 44. Monitoring and modeling of lymphocytic leukemia cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7536222/]
- 45. What causes non-specific antibody binding and how can it ... [https://www.cytometry.org/web/q_view.php?id=153&filter=Analysis%20Techniques]
- 46. Troubleshooting Flow Cytometry Issues [https://www.bosterbio.com/protocol-and-troubleshooting/flow-cytometry-troubleshooting?srsltid=AfmBOoqcVURe-EuipofOCeUDwRPBmvy7k-qCACJn7QMysIesaUu4iWz_]
- 47. Flow Cytometry Troubleshooting Guide [https://fluorofinder.com/flow-cytometry-troubleshooting/]
- 48. Why the immune fluorescence result has high background? [https://www.sinobiological.com/category/if-icc-troubleshooting-high-background]
- 49. Flow cytometry troubleshooting [https://www.abcam.com/en-us/technical-resources/applications/flow-cytometry/troubleshooting]
- 50. Interaction of rhodamine 123 with living cells studied by flow ... [https://pubmed.ncbi.nlm.nih.gov/7059978/]
- 51. Effects of ROS Pathway Inhibitors and NADH and FADH2 ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10528392/]
- 52. Photobleaching and phototoxicity of mitochondria in live cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11500826/]
- 53. Gentle Rhodamines for Live-Cell Fluorescence Microscopy [https://pmc.ncbi.nlm.nih.gov/articles/PMC11503488/]
- 54. Investigating the photophysical properties of rhodamines ... [https://pubs.rsc.org/en/content/articlelanding/2024/ra/d4ra06577h]
- 55. Mitochondrial Membrane Potential - an overview [https://www.sciencedirect.com/topics/medicine-and-dentistry/mitochondrial-membrane-potential]
- 56. Methods to Evaluate Changes in Mitochondrial Structure ... [https://www.mdpi.com/2072-6694/15/9/2564]
- 57. Characterization of Rhodamine-123 as a Tracer Dye for Use ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0033253]
- 58. Active transport of rhodamine 123 by the human multidrug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7105846/]
- 59. Cannabidiol (CBD) Inhibited Rhodamine-123 Efflux in ... [https://www.frontiersin.org/journals/behavioral-neuroscience/articles/10.3389/fnbeh.2020.00032/full]
- 60. The mitochondrial fluorescent dye rhodamine 123 is a high ... [https://pubmed.ncbi.nlm.nih.gov/22913740/]
- 61. The Role of Surfactants in the Reversal of Active Transport ... [https://www.sciencedirect.com/science/article/abs/pii/S0022354916312552]
- 62. Key Errors to Avoid in the Consideration of Fluorescence ... [https://www.spectroscopyonline.com/view/key-errors-avoid-consideration-fluorescence-quenching-data]
- 63. Guidelines for the measurement of oxygen consumption ... [https://www.sciencedirect.com/science/article/pii/S2213231725002368]
- 64. Measurement of Oxygen Consumption Rate (OCR) and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8275291/]
- 65. Monitoring and modeling of lymphocytic leukemia cell ... [https://www.nature.com/articles/s41467-020-18769-y]
- 66. Mitochondrial Membrane Potential - an overview | ScienceDirect Topics [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/mitochondrial-membrane-potential]
- 67. TMRM | Mitochondrial Membrane Potential Fluorescent Dye [https://www.medchemexpress.com/TMRM.html?srsltid=AfmBOorPiPdIogZoXLFGNLIGLFO9Khpn3IGQG3lwHOlq8AeD__ERL2nI]
- 68. Fluorescence/UV-VIS Spectrophotometer Troubleshooting ... [https://www.vernier.com/til/3918?srsltid=AfmBOoqed0mfFSN-FikfdT_GZ4hoCuMFjA4JsrffKreaXpPYKWfCsrs6]