Measuring Caspase-8 Activation: A Comprehensive Guide to Methodologies and Applications in Extrinsic Apoptosis
This article provides a comprehensive resource for researchers and drug development professionals on the current methodologies for measuring caspase-8 activation in the extrinsic apoptotic pathway.
Measuring Caspase-8 Activation: A Comprehensive Guide to Methodologies and Applications in Extrinsic Apoptosis
Abstract
This article provides a comprehensive resource for researchers and drug development professionals on the current methodologies for measuring caspase-8 activation in the extrinsic apoptotic pathway. It covers the foundational biology of caspase-8, from its initiation at the Death-Inducing Signaling Complex (DISC) to its downstream effects, including both type I and type II apoptotic pathways. The content details established and emerging protocols, such as immunoprecipitation combined with activity assays and live-cell FRET-based biosensors, alongside troubleshooting and optimization strategies. Furthermore, it explores validation techniques, the role of caspase-8 in non-apoptotic processes like inflammation, and its implications in cancer and neurodegenerative diseases, offering a holistic view for both basic research and therapeutic development.
The Central Role of Caspase-8 in Initiating Extrinsic Apoptosis
The initiation of the extrinsic apoptosis pathway is a tightly regulated process, triggered by the assembly of the Death-Inducing Signaling Complex (DISC). This multi-protein complex serves as the central activation platform for caspase-8, the initiator caspase that propagates the cell death signal [1]. The DISC forms upon activation of death receptors such as CD95/Fas or TRAIL-R1/DR4/TRAIL-R2/DR5 by their respective ligands. The core molecular architecture of the DISC comprises the activated death receptor, the adaptor protein FADD (Fas-Associated protein with Death Domain), and procaspase-8 [1]. A critical feature of DISC assembly is the framework of defined interactions between death domains (DD) and death effector domains (DEDs). Recent research has revealed that procaspase-8 molecules form so-called DED chains or filaments via DED interactions, which serve as the essential platform for procaspase-8 dimerization and subsequent activation [2] [1].
The controlled activation of caspase-8 at the DISC is crucial for maintaining tissue homeostasis and eliminating potentially harmful cells. Dysregulation of this process is implicated in various diseases, including cancer, autoimmunity, and neurodegeneration [3]. Consequently, understanding the precise molecular mechanisms of DISC formation and caspase-8 activation provides valuable insights for therapeutic interventions targeting the apoptosis pathway.
Molecular Architecture and Stoichiometry of the DISC
The DED Chain Model
Traditional models suggested a 1:1 stoichiometry among core DISC components. However, advanced quantitative mass spectrometry analyses of the native TRAIL DISC have revealed a more complex architecture [4]. The data indicate that FADD is substoichiometric relative to TRAIL receptors or DED-only proteins. Strikingly, there is up to a 9-fold molar excess of caspase-8 compared to FADD within the complex [4]. This finding led to the proposal of an alternative DISC model in which procaspase-8 molecules interact sequentially via their DED domains to form an activating chain. Structural modeling supports this concept, suggesting that FADD can recruit multiple DED-containing proteins to the DISC, initiating the formation of a caspase-8 activation chain [4].
Table 1: Key Protein Components of the DISC
| Component | Role in DISC | Key Domains | Function in Apoptosis Initiation |
|---|---|---|---|
| Death Receptors (CD95/Fas, TRAIL-R1/2) | Initiation | Death Domain (DD) | Transmit extracellular death signals into the cell |
| FADD | Adaptor | Death Domain (DD), Death Effector Domain (DED) | Bridges death receptors to DED-containing proteins |
| Procaspase-8 | Executioner protease | Two Death Effector Domains (DED1, DED2), Catalytic domain | Forms DED filaments; activated via dimerization at DISC |
| c-FLIP proteins | Regulator | Two Death Effector Domains (DED1, DED2) | Modulate caspase-8 activation; can be pro- or anti-apoptotic |
Quantitative Analysis of the Native TRAIL DISC
The application of liquid chromatography with tandem mass spectrometry (LC-MS/MS) has provided unprecedented insights into the stoichiometry of the native TRAIL DISC. This complex was identified as a soluble structure exceeding 700 kDa, containing TRAIL receptors, FADD, and DED-only proteins [4]. The quantitative data demonstrating the substoichiometric presence of FADD fundamentally challenges previous models and highlights the crucial role of caspase-8 DED chain assembly in triggering cell death. Experimental validation through mutation of key interacting residues in procaspase-8 DED2 abrogates DED chain formation in cells and disrupts TRAIL/CD95 DISC-mediated procaspase-8 activation [4].
Caspase-8 Activation Mechanism at the DED Filaments
Molecular Events in Caspase-8 Activation
Caspase-8 activation at the DED filaments represents a critical control point in extrinsic apoptosis. The process involves several sequential molecular events. Initially, procaspase-8 molecules are recruited to the growing DED filaments at the DISC. Within these filaments, procaspase-8 molecules undergo dimerization, which triggers conformational changes that rearrange the L2 loop in the zymogen structure [1]. This L2 loop contains the active cysteine residue, and its rearrangement during dimerization facilitates the formation of the active center of procaspase-8.
Following dimerization, procaspase-8 undergoes cleavage at specific aspartic acid residues. The initial cleavage occurs at Asp374 within the L2 loop, generating p43/p41 and p12 cleavage products [1]. This is followed by further autocatalytic processing at Asp384 and Asp210/216, resulting in the formation of the active caspase-8 heterotetramer p10₂/p18₂ [1]. A unique feature observed in the structure of procaspase-8/c-FLIPL heterodimer is the "closed" conformation of the unprocessed L2′ fragment (L2′ loop), which appears to stabilize the active center of caspase-8 and thereby promotes catalytic activity [1].
Diagram 1: Sequential Process of Caspase-8 Activation at the DISC
Regulatory Control by c-FLIP Proteins
The initiation of caspase-8 activation at the DISC and DED filaments is extensively controlled by cellular FLICE-inhibitory protein (c-FLIP) isoforms [1]. Three main c-FLIP isoforms have been characterized: c-FLIPL (Long), c-FLIPS (Short), and c-FLIPR (Raji). All three isoforms possess two DED domains, while c-FLIPL additionally contains catalytically inactive caspase-like domains (p20 and p12) [1].
The short c-FLIP isoforms (c-FLIPS and c-FLIPR) generally block death receptor-induced apoptosis by inhibiting procaspase-8 activation at the DISC when expressed at high levels [1]. Recent studies suggest they achieve this by either interrupting the procaspase-8 chains at the DISC or by incorporating into DED chains and forming inactive heterodimers [1]. In contrast, c-FLIPL exhibits a dual function depending on its expression level. At moderate expression levels, c-FLIPL acts in a proapoptotic manner by forming procaspase-8/c-FLIPL heterodimers that enhance caspase-8 catalytic activity [1]. At high expression levels, it functions as an antiapoptotic factor.
Table 2: c-FLIP Isoforms and Their Functions in DISC Regulation
| c-FLIP Isoform | Structure | Expression Level | Primary Function | Effect on Apoptosis |
|---|---|---|---|---|
| c-FLIPL | 2 DEDs + p20/p12 domains | Moderate | Forms active heterodimers with procaspase-8 | Proapoptotic |
| c-FLIPL | 2 DEDs + p20/p12 domains | High | Competitively inhibits procaspase-8 activation | Antiapoptotic |
| c-FLIPS | 2 DEDs only | High | Incorporates into DED chains; forms inactive heterodimers | Antiapoptotic |
| c-FLIPR | 2 DEDs only | High | Incorporates into DED chains; forms inactive heterodimers | Antiapoptotic |
Research Applications and Pharmacological Targeting
Pharmacological Modulation of Caspase-8 Activity
The precise understanding of caspase-8 activation mechanisms has enabled the development of targeted pharmacological interventions. A pioneering approach involved the rational design of a first-in-class chemical probe targeting c-FLIPL in the heterodimer with caspase-8 [1]. This small molecule was designed to imitate the closed conformation of the caspase-8 L2′ loop, thereby increasing caspase-8 activity after initial processing of the heterodimer [1].
In accordance with in silico predictions, this small molecule enhanced caspase-8 activity at the DISC, CD95L/TRAIL-induced caspase activation, and subsequent apoptosis [1]. Computational modeling provided further evidence for the proposed effects, demonstrating that boosting caspase-8 activity by the small molecule at early time points after DISC assembly is crucial for promoting apoptosis induction. This strategic approach to targeting the caspase-8/c-FLIPL heterodimer provides new insights into molecular mechanisms of activation and offers potential therapeutic strategies for cancer treatment.
Experimental Protocol: Measuring Caspase-8 Activity at DED Filaments
The following detailed protocol describes the procedure for measuring caspase-8 activity directly at the death-inducing signaling complex (DISC) in adherent cells, enabling researchers to analyze caspase-8 activation in its native complex and assess the efficacy of pharmacological inhibitors targeting caspase-8 [2].
Cell Culture and Preparation
- Cell Lines: Human cervical cancer HeLa-CD95 cells (CD95-overexpressing cells) and HeLa-CD95-FL cells (CD95/c-FLIPL-overexpressing cells) are maintained in DMEM/Ham's F-12 media, supplemented with 10% heat-inactivated fetal calf serum, 1% penicillin-streptomycin, and 0.0001% puromycin in 5% CO₂ [1].
- Culture Conditions: Cells should be maintained under standard conditions (37°C, 5% CO₂) and passaged regularly to ensure optimal growth and experimental reproducibility.
- Preparation for Experiments: Plate cells at appropriate density (typically 1-2 × 10⁶ cells per 10 cm dish) and allow to adhere overnight before apoptosis induction.
Apoptosis Induction and DISC Immunoprecipitation
- Induction of Apoptosis: Induce apoptosis using recombinant LZ-CD95L (200 ng/mL) or TRAIL (100 ng/mL) for specified time points (typically 0-30 minutes) depending on experimental requirements [1].
- Cell Lysis: Following treatment, lyse cells in DISC immunoprecipitation buffer (20 mM Tris/HCl pH 7.4, 1% Triton X-100, 10% glycerol, 150 mM NaCl, supplemented with complete protease inhibitors).
- DISC Immunoprecipitation: Incubate cell lysates with anti-APO-1 antibody (2 μg per 500 μg of total protein) for 4 hours at 4°C with gentle rotation. Subsequently, add protein A/G sepharose beads and incubate for an additional 2 hours [2] [1].
- Bead Washing: Wash immunoprecipitates thoroughly with lysis buffer (3-4 times) to remove non-specifically bound proteins.
Caspase-8 Activity Assay
- Assay Setup: Resuspend immunoprecipitated DISC complexes in caspase assay buffer (100 mM HEPES, 10% sucrose, 0.1% CHAPS, 10 mM DTT, pH 7.4).
- Substrate Addition: Add caspase-8-specific fluorogenic substrate (IETD-AFC or IETD-AMC) at a final concentration of 50 μM.
- Fluorescence Measurement: Monitor fluorescence emission (excitation 400 nm, emission 505 nm for AFC; excitation 380 nm, emission 460 nm for AMC) over 60-120 minutes at 37°C using a fluorometric plate reader.
- Data Analysis: Calculate caspase-8 activity as pmol of substrate cleaved per minute per mg of total protein, using a standard curve of free AFC or AMC.
Western Blot Analysis
- Sample Preparation: Denature immunoprecipitated proteins in Laemmli buffer by boiling at 95°C for 5 minutes.
- Gel Electrophoresis: Separate proteins by SDS-PAGE (10-12% gels) and transfer to PVDF membranes.
- Antibody Probing: Probe membranes with primary antibodies against caspase-8 (clone C15), FADD (clone 1C4), c-FLIP (clone NF6), and CD95 (for normalization) [1].
- Detection: Use appropriate HRP-conjugated secondary antibodies and develop with enhanced chemiluminescence reagent.
- Data Interpretation: Analyze protein levels and processing using densitometry software.
Diagram 2: Experimental Workflow for Measuring Caspase-8 Activity at DISC
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for DISC and Caspase-8 Studies
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| Cell Lines | HeLa-CD95, HeLa-CD95-FL, Jurkat, MV4-11 | Model systems for studying DISC formation and caspase-8 activation [1] |
| Death Receptor Ligands | Recombinant LZ-CD95L, TRAIL (KillerTRAIL) | Induce DISC assembly and initiate extrinsic apoptosis pathway [1] |
| Antibodies for Immunoprecipitation | Anti-APO-1 (for CD95/Fas) | Specific antibodies for isolating native DISC complexes [1] |
| Antibodies for Western Blot | Anti-caspase-8 (C15), Anti-FADD (1C4), Anti-c-FLIP (NF6), Anti-caspase-3, Anti-PARP | Detect protein expression, processing, and activation markers [1] |
| Caspase Activity Substrates | IETD-AFC, IETD-AMC | Fluorogenic substrates for measuring caspase-8 activity in real-time [2] |
| Pharmacological Modulators | FLIPin small molecules (FLIPinQ, FLIPinR) | Enhance caspase-8 activity by targeting caspase-8/c-FLIPL heterodimer [1] |
| Computational Tools | Schrödinger Small-Molecule Drug Discovery Suite, Glide molecular docking | Structure-based drug design targeting DISC components [1] |
The formation of the Death-Inducing Signaling Complex and the subsequent assembly of DED filaments represent a crucial control point in the extrinsic apoptosis pathway. The emerging model of sequential procaspase-8 interactions forming an activating chain, supported by quantitative mass spectrometry data revealing the substoichiometric relationship between FADD and caspase-8, has fundamentally advanced our understanding of this process [4]. The development of sophisticated protocols for measuring caspase-8 activity directly at the DISC [2], combined with structure-based pharmacological approaches to target regulatory components like the caspase-8/c-FLIPL heterodimer [1], provides powerful tools for both basic research and therapeutic development. These advances offer promising avenues for manipulating this critical cell death pathway in pathological conditions, particularly in cancer where apoptosis resistance is a hallmark feature.
Caspase-8 Dimerization, Autoprocessing, and Full Activation
Caspase-8 functions as the critical initiator caspase in the extrinsic apoptosis pathway, triggering a proteolytic cascade that leads to programmed cell death. Activation of caspase-8 occurs through a tightly regulated process involving recruitment to death receptor complexes, dimerization, and autoproteolytic processing. Understanding this activation mechanism is fundamental to apoptosis research, with implications for cancer biology, autoimmune disorders, and therapeutic development. This application note details the molecular mechanisms of caspase-8 activation and provides standardized protocols for its detection and quantification in experimental systems, framed within the context of measuring caspase-8 activation for extrinsic pathway research.
Molecular Mechanism of Activation
Caspase-8 activation follows a sequential, multi-step process that transforms the inactive zymogen into a fully active protease.
Initial Recruitment and Oligomerization
The activation cascade begins when extracellular death ligands (e.g., FasL, TRAIL) bind to their cognate death receptors, triggering intracellular death-inducing signaling complex (DISC) formation. The adaptor protein FADD is recruited to activated receptors via death domain interactions [5]. FADD then recruits procaspase-8 through homotypic death effector domain (DED) interactions [6]. Recent structural studies using cryo-electron microscopy reveal that caspase-8 tandem DEDs (tDEDs) form helical filaments rather than simple linear chains within the DISC [6]. This filamentous assembly provides the structural framework for proximity-induced dimerization.
Table 1: Core Components of the Death-Inducing Signaling Complex (DISC)
| Component | Domain Architecture | Function in Caspase-8 Activation |
|---|---|---|
| Death Receptor | Extracellular CRD, transmembrane, intracellular DD | Receptor for death ligands; initiates DISC assembly |
| FADD | C-terminal DD, N-terminal DED | Adaptor protein; bridges death receptors and caspase-8 |
| Procaspase-8 | N-terminal tDED, large and small catalytic subunits | Zymogen form; undergoes activation through dimerization and cleavage |
| cFLIP isoforms | tDED (with/without caspase-like domain) | Key regulator; modulates caspase-8 activation outcomes |
Dimerization and Autoprocessing
Within the DISC filament architecture, caspase-8 catalytic domains form homodimers, enabling trans-autoproteolysis. Research demonstrates that neither dimerization nor cleavage alone is sufficient for full caspase-8 activation; both processes must occur coordinately [7]. The autoproteolytic processing occurs at specific aspartic acid residues: first between the linker and small subunit (D384 in humans, D387 in mice), then between DED2 and the large subunit, releasing the fully matured enzyme from the DISC [5].
The following diagram illustrates this sequential activation process:
Quantitative Analysis of Caspase-8 Activation Parameters
The activation mechanism of caspase-8 involves specific biochemical parameters that can be quantitatively measured. The following table summarizes key quantitative findings from mechanistic studies:
Table 2: Quantitative Parameters of Caspase-8 Activation
| Parameter | Experimental Finding | Experimental System | Significance |
|---|---|---|---|
| Activation Requirement | Neither dimerization nor cleavage alone sufficient; coordinated both required [7] | Inducible dimerization/cleavage system in caspase-8 deficient cells | Explains conflicting prior reports; establishes dual requirement |
| DISC Stoichiometry | ~6 procaspase-8 molecules per FADD protein [5] | Stoichiometric analysis of DISC components | Indicates significant amplification at signaling level |
| Filament Binding Affinity | Apparent KD of 175 ± 1.4 nM for FasDD/FADD complex to caspase-8 tDED [6] | Fluorescence polarization assay with purified components | Quantifies core protein interactions in DISC assembly |
| cFLIP Regulatory Effect | Concentration-dependent inhibition or promotion based on stoichiometry [5] [8] | Cellular and biochemical assays | Explains context-dependent regulatory function |
| Phosphorylation Regulation | Y380 phosphorylation blocks apoptosis without impairing initial DISC recruitment [5] | Phosphomimetic mutagenesis | Identifies post-translational regulatory mechanism |
Experimental Protocols for Monitoring Caspase-8 Activation
FRET-Based Caspase Activity Monitoring in Live Cells
Fluorescence Resonance Energy Transfer (FRET) reporters enable real-time monitoring of caspase activity in living cells [9] [10].
Protocol:
- Construct Design: Create a plasmid encoding CFP and YFP connected by a caspase-8-specific cleavage sequence (IETD). Tandem repeats enhance sensitivity [9].
- Cell Transfection: Transfect cells using appropriate method (lipofection, electroporation) and culture for 24-48 hours for expression.
- Treatment and Imaging:
- Seed transfected cells in imaging-compatible plates
- Treat with apoptosis inducers (e.g., TRAIL, 100 ng/mL; FasL, 50 ng/mL)
- Acquire time-lapse images using fluorescence microscopy with CFP (excitation 434 nm/emission 474 nm) and FRET (excitation 434 nm/emission 527 nm) channels
- Data Analysis: Calculate FRET ratio (FRET channel/CFP channel). Cleavage reduces FRET efficiency, decreasing ratio over time.
- Controls: Include caspase inhibitor (z-IETD-fmk, 20 µM) and cleavage-resistant mutant (IETD→IEAD) as negative controls [10].
Immunoblot Analysis of Caspase-8 Processing
Direct detection of caspase-8 cleavage fragments provides molecular evidence of activation.
Protocol:
- Sample Preparation:
- Treat cells with death receptor agonists for various durations
- Lyse cells in RIPA buffer supplemented with protease inhibitors
- Normalize protein concentrations (e.g., BCA assay)
- Electrophoresis and Transfer:
- Separate proteins (30-50 μg per lane) on 4-12% Bis-Tris gels
- Transfer to PVDF membranes using standard protocols
- Immunoblotting:
- Block with 5% non-fat milk in TBST
- Incubate with primary antibodies: anti-caspase-8 (recognizing pro-form and cleavage fragments)
- Use HRP-conjugated secondary antibodies and chemiluminescent detection
- Interpretation: Processed caspase-8 appears as cleaved fragments (p43/p41, p18) with corresponding decrease in pro-caspase-8 band [7] [11].
Flow Cytometric Detection with FLICA Assays
Fluorochrome-Labeled Inhibitors of Caspases (FLICAs) enable quantification of caspase activity at single-cell level.
Protocol:
- Cell Staining:
- Harvest cells after treatment
- Incubate with FITC-, FAM-, or SR-labeled IETD-fmk probe (10 μM) for 60 minutes at 37°C
- Washing and Analysis:
- Wash cells twice with wash buffer
- Resuspend in buffer containing viability dye (e.g., propidium iodide)
- Analyze by flow cytometry using appropriate channels
- Gating Strategy: Gate on viable cells, then analyze FLICA fluorescence intensity. Caspase-8 active cells show high fluorescence [12].
- Multiplexing: Combine with mitochondrial membrane potential dyes (TMRM) or phosphatidylserine exposure (Annexin V) for multiparametric apoptosis assessment [12].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Caspase-8 Research
| Reagent Category | Specific Examples | Research Application | Mechanistic Insight Provided |
|---|---|---|---|
| Activity Reporters | CFP-IETD-YFP FRET construct [9], CellEvent Caspase-3/7 Green [12] | Live-cell activity monitoring | Visualizes temporal dynamics of caspase activation in intact cells |
| Chemical Inhibitors | z-IETD-fmk (caspase-8 inhibitor) [10], emricasan (broad-spectrum) [8] | Pathway inhibition studies | Establages causal relationship between caspase-8 activity and apoptotic phenotype |
| Activity-Based Probes | FAM-IETD-fmk (FLICA) [12], SR-IETD-fmk [10] | Flow cytometric detection | Enables quantification of active enzyme populations at single-cell resolution |
| Antibodies | Anti-caspase-8 (pro-form and cleaved forms) [7] [11], anti-FADD [6] | Immunoblotting, immunoprecipitation | Detects molecular processing events and protein interactions |
| Expression Constructs | Caspase-8 cleavage-site mutants [7], inducible dimerization systems [7] | Mechanistic dissection | Separates functional contributions of dimerization versus cleavage |
| Recombinant Proteins | Purified caspase-8 tDED [6], FADD, FasDD | Structural and biochemical studies | Enables reconstitution of DISC assembly in vitro |
Regulatory Context and Implications
Caspase-8 activation is precisely regulated through multiple mechanisms, with important implications for experimental interpretation and therapeutic targeting. The cellular FLICE-inhibitory protein (cFLIP) exists in multiple isoforms that differentially regulate caspase-8 activation - cFLIPₛ inhibits activation while cFLIPₗ can promote limited activation [5] [8]. Phosphorylation at specific residues (e.g., Y380) can block apoptotic function without preventing initial DISC recruitment [5]. Recent research also reveals non-apoptotic functions of caspase-8 in regulating inflammation through cleavage of substrates like N4BP1, particularly relevant in pathological contexts such as severe SARS-CoV-2 infection [8].
The following diagram illustrates the regulatory networks controlling caspase-8 activity:
The activation of caspase-8 through coordinated dimerization and autoprocessing represents a critical control point in extrinsic apoptosis. The experimental approaches detailed herein provide researchers with robust methods to quantify and manipulate this process, enabling deeper investigation into cell death mechanisms and their therapeutic applications. As research continues to reveal non-apoptotic functions and regulatory complexities of caspase-8, these standardized protocols will facilitate comparison across studies and accelerate progress in the field of programmed cell death research.
In death receptor-mediated apoptosis, the classification of cellular responses into Type I and Type II pathways represents a fundamental paradigm for understanding downstream signaling complexity. This classification originated from observations that different cell types execute the extrinsic apoptosis program through distinct intracellular signaling routes following death receptor engagement [13]. The critical determinant separating these pathways lies in how cells process the initial caspase-8 signal generated at the Death-Inducing Signaling Complex (DISC) and whether they require mitochondrial amplification to fully activate the executioner phase of apoptosis [13].
In Type I cells, the extrinsic pathway activation generates sufficient amounts of active caspase-8 at the DISC to directly cleave and activate executioner caspases (caspase-3 and -7), enabling rapid apoptosis induction without mitochondrial involvement [13]. Conversely, in Type II cells, the initial caspase-8 signal requires amplification through the mitochondrial apoptotic pathway. This amplification occurs via caspase-8-mediated cleavage of the BH3-only protein BID, generating truncated BID (tBID), which translocates to mitochondria and induces BAX/BAK-mediated mitochondrial outer membrane permeabilization (MOMP) [13]. This mitochondrial phase enables the release of cytochrome c and other pro-apoptotic factors, leading to apoptosome formation and robust caspase activation.
The molecular basis for this differential signaling appears to be regulated by the expression levels of key inhibitory proteins. Cells with high levels of X-linked Inhibitor of Apoptosis Protein (XIAP) typically require the mitochondrial amplification pathway (Type II) to overcome XIAP-mediated caspase inhibition, whereas cells with lower XIAP levels can proceed directly through Type I signaling [13]. Additionally, the expression levels of cellular FLICE-inhibitory protein (c-FLIP), which regulates caspase-8 activation at the DISC, further contribute to determining pathway preference [13].
Key Molecular Distinctions Between Type I and Type II Pathways
Comparative Analysis of Signaling Characteristics
The differential features of Type I and Type II signaling pathways extend across multiple molecular and functional dimensions, as systematically compared in Table 1.
Table 1: Key Characteristics of Type I and Type II Signaling Pathways
| Characteristic | Type I Pathway | Type II Pathway |
|---|---|---|
| Initial Trigger | Death receptor activation (e.g., Fas, TRAIL-R) [13] | Death receptor activation (e.g., Fas, TRAIL-R) [13] |
| Key Signaling Hub | Death-Inducing Signaling Complex (DISC) [13] | DISC followed by mitochondrial amplification [13] |
| Caspase-8 Requirement | High activation levels [13] | Lower activation levels requiring amplification [13] |
| Mitochondrial Involvement | Not required [13] | Essential (BAX/BAK activation, MOMP) [13] |
| BID Cleavage | Not essential for apoptosis execution [13] | Critical for mitochondrial amplification [13] |
| XIAP Sensitivity | Lower (direct caspase activation bypasses inhibition) [13] | Higher (requires Smac/DIABLO release to counteract XIAP) [13] |
| c-FLIP Sensitivity | Highly sensitive to inhibition [13] | Less sensitive to c-FLIP-mediated regulation [13] |
| Kinetics of Apoptosis | Rapid initiation [13] | Delayed (requires mitochondrial amplification) [13] |
Molecular Regulators of Pathway Selection
The decision between Type I and Type II signaling is governed by several critical molecular determinants that collectively establish the cellular context for apoptosis execution:
Inhibitor of Apoptosis Proteins (XIAP): XIAP directly binds to and inhibits caspases-3, -7, and -9 [13]. In Type II cells, where XIAP expression is typically higher, the mitochondrial amplification phase enables the release of mitochondrial proteins such as Smac/DIABLO, which counteracts XIAP inhibition, thereby permitting full caspase activation [13].
Cellular FLIP (c-FLIP): This homolog of caspase-8 lacks catalytic activity and functions as a key regulator of DISC signaling by competing with caspase-8 for binding to FADD [13]. Elevated c-FLIP expression preferentially inhibits Type I signaling by preventing sufficient caspase-8 activation at the DISC.
BCL-2 Family Proteins: In Type II cells, the anti-apoptotic BCL-2 family members (including BCL-2 itself and BCL-XL) can inhibit apoptosis by preventing BAX/BAK activation and MOMP [14]. The ratio of pro-apoptotic to anti-apoptotic BCL-2 family proteins thus significantly influences cellular predisposition to Type II signaling.
Protocol: Measuring Caspase-8 Activation at the DISC
Experimental Rationale and Workflow
Accurate measurement of caspase-8 activation within its native complex is essential for delineating Type I versus Type II signaling mechanisms. This protocol enables researchers to directly quantify the initial caspase-8 signal generated at the DISC, providing critical insight into a cell's apoptotic pathway classification and potential therapeutic responses [15]. The procedure combines immunoprecipitation of the native DISC with a sensitive caspase activity assay, allowing specific assessment of caspase-8 activation kinetics and magnitude in different cellular contexts.
Table 2: Key Research Reagents for DISC and Caspase-8 Analysis
| Reagent/Category | Specific Examples | Function/Application |
|---|---|---|
| Cell Lines | HeLa-CD95 (CD95-overexpressing) [15] | Model system with robust DISC formation; other adherent or suspension lines sensitive to CD95L-induced apoptosis can be substituted |
| Critical Antibodies | Anti-CD95 (Fas) [15] | Immunoprecipitation of the DISC |
| Anti-caspase-8 (clone C15) [15] | Detection of caspase-8 recruitment and processing | |
| Anti-FADD (clone 1C4) [15] | Confirmation of DISC composition | |
| Anti-c-FLIP (clone NF6) [15] | Assessment of DISC regulation | |
| Apoptosis Inducers | Recombinant CD95L [15] | Direct activation of the extrinsic pathway through CD95 receptor engagement |
| Caspase Substrates/Inhibitors | IETD-based substrates (e.g., IETD-pNA) [15] | Colorimetric measurement of caspase-8 enzymatic activity |
| Z-IETD-FMK [15] | Specific caspase-8 inhibition for control experiments | |
| Lysis/IP Buffer Components | CHAPS detergent [15] | Maintains protein complexes while solubilizing membranes |
| DTT [15] | Maintaining reducing conditions for protein stability |
Step-by-Step Methodology
Cell Culture and Apoptosis Induction
- Cell Preparation: Culture HeLa-CD95 cells (or other CD95L-sensitive lines) in DMEM F12 medium supplemented with 10% fetal calf serum and appropriate antibiotics [15]. For each experimental condition, seed 5 × 10^6 cells in 14.5 cm plates and incubate overnight at 37°C with 5% CO₂ to achieve 70-80% confluence.
- Viability Assessment: Prior to experimentation, confirm cell viability exceeds 93% using Trypan Blue exclusion counting [15].
- Apoptosis Induction: Stimulate cells with 1-2 μg/mL recombinant CD95L for predetermined time points (typically 0-60 minutes) to trigger DISC formation [15]. Include untreated controls and "Beads control" for immunoprecipitation background assessment.
DISC Immunoprecipitation
- Cell Lysis: Following CD95L stimulation, immediately place plates on ice, wash cells with ice-cold PBS, and lyse using 1 mL of M-Per lysis buffer supplemented with 1× protease and phosphatase inhibitors [15].
- DISC Isolation: Centrifuge lysates at 16,000 × g for 15 minutes at 4°C. Incubate cleared supernatants with 2 μg anti-CD95 antibody (e.g., mouse monoclonal anti-APO-1) for 2 hours at 4°C with gentle rotation [15].
- Complex Recovery: Add 30 μL protein A/G agarose beads and incubate for an additional 2 hours. Pellet beads by gentle centrifugation (1,000 × g for 5 minutes) and wash three times with ice-cold lysis buffer [15].
Caspase-8 Activity Assay
- Sample Preparation: Divide immunoprecipitated complexes into two equal aliquots: one for caspase activity measurement and one for Western blot validation [15].
- Enzymatic Reaction: Resuspend the activity assay aliquot in 200 μL caspase assay buffer (100 mM HEPES, 10% sucrose, 0.1% CHAPS, 10 mM DTT, pH 7.4) containing 200 μM IETD-pNA substrate [15].
- Kinetic Measurement: Incubate at 37°C for 0-120 minutes and measure absorbance at 405 nm at 15-minute intervals using a plate reader [15].
- Data Calculation: Express caspase-8 activity as pmol pNA released per minute per mg of total protein used in the immunoprecipitation.
Western Blot Validation
- Sample Processing: Boil the reserved IP aliquot in 1× Laemmli buffer for 10 minutes, separate proteins by SDS-PAGE, and transfer to PVDF membranes [15].
- Membrane Probing: Block membranes with 5% non-fat milk, then incubate with primary antibodies against caspase-8 (1:20), FADD (1:10), and CD95 (1:500) [15].
- Signal Detection: After incubation with appropriate HRP-conjugated secondary antibodies, develop blots using enhanced chemiluminescence and image with a digital capture system [15].
Data Interpretation and Technical Considerations
- Type I vs. Type II Classification: Cells exhibiting rapid, robust caspase-8 activation at the DISC (within 15-30 minutes of CD95L stimulation) typically represent Type I signaling. In contrast, Type II cells demonstrate slower, less pronounced DISC-based caspase-8 activation, requiring longer stimulation periods for detectable activity [13].
- Quality Controls: Successful DISC immunoprecipitation should demonstrate time-dependent recruitment of both FADD and caspase-8 to CD95, with progressive caspase-8 processing to its active p18/p10 fragments [15].
- Troubleshooting: High background caspase activity may indicate excessive lysis or inadequate washing. Optimization of antibody concentrations and inclusion of caspase-8-specific inhibitors (Z-IETD-FMK) in control samples are essential for assay validation [15].
Signaling Pathway Visualizations
Type I and Type II Apoptotic Signaling Pathways
Experimental Protocol Workflow
Discussion: Therapeutic Implications and Research Applications
The distinction between Type I and II signaling pathways extends beyond theoretical interest to practical applications in drug discovery and therapeutic development. Understanding a particular cancer's apoptotic signaling classification enables more targeted therapeutic approaches. For instance, Type II cells, being dependent on mitochondrial amplification, demonstrate greater sensitivity to drugs targeting BCL-2 family proteins (such as venetoclax/ABT-199) [14], while Type I cells may respond better to agents that enhance DISC formation or caspase-8 activation.
Furthermore, the non-apoptotic functions of caspase-8 have emerged as significant factors in pathological inflammation. Recent research indicates that caspase-8 regulates inflammatory responses independent of its cell death functions, cleaving negative regulators like N4BP1 to potentiate NF-κB signaling and IL-1β production [8]. This mechanism appears particularly relevant in severe SARS-CoV-2 infection, where caspase-8 drives pathological inflammation independent of its apoptotic function [8].
From a methodological perspective, the protocol described herein for measuring caspase-8 activation at the DISC provides a robust framework for classifying cell signaling types and screening potential therapeutic compounds. This approach enables direct assessment of how pharmacological interventions affect the initial events in extrinsic apoptosis, facilitating more precise modulation of cell death pathways for therapeutic benefit. As research continues to elucidate the complex interplay between cell death and inflammatory signaling, these experimental approaches will remain essential tools for dissecting disease mechanisms and developing targeted interventions.
Caspase-8, a cysteine-aspartic protease, has been extensively studied for its pivotal role as an initiator of extrinsic apoptosis. However, emerging research has revealed that this enzyme possesses critical functions beyond apoptosis, serving as a key regulator of inflammatory processes and immune homeostasis [16]. Originally identified for its capacity to initiate programmed cell death upon death receptor engagement, caspase-8 is now recognized as a multifunctional protein that can modulate inflammatory signaling, cytokine production, and other non-apoptotic cellular responses [17]. This paradigm shift underscores caspase-8's role as a molecular switch that can direct cellular outcomes toward either death or inflammation, depending on cellular context, expression levels, and proteolytic status.
The broader thesis of measuring caspase-8 activation in extrinsic pathway research must now expand to encompass these non-apoptotic functions, requiring refined experimental approaches that can distinguish between caspase-8's proteolytic activity and its scaffolding capabilities. This application note details the mechanistic insights, experimental protocols, and research tools essential for investigating the non-apoptotic roles of caspase-8 in inflammation, providing a framework for researchers exploring this complex signaling node in health and disease.
Mechanistic Foundations: Molecular Basis of Non-Apoptotic Functions
The Scaffolding Role of Caspase-8 in Inflammation
Caspase-8 can function in a non-enzymatic capacity as a structural scaffold for the assembly of inflammatory signaling complexes. Upon TRAIL stimulation, caspase-8 recruits key signaling molecules to form a pro-inflammatory "FADDosome" complex, consisting of caspase-8, FADD, and RIPK1 [18]. This complex formation occurs independently of caspase-8's catalytic activity but is essential for subsequent NF-κB activation and pro-inflammatory cytokine production. Introduction of a catalytically inactive caspase-8 mutant into caspase-8 null cells restores TRAIL-induced cytokine production but not cell death, confirming the dissociation between its scaffolding and proteolytic functions [18].
The scaffolding function of caspase-8 extends to its regulation of the cytokine suppressor NEDD4-binding protein 1 (N4BP1). Caspase-8-mediated cleavage inactivates N4BP1, thereby relieving suppression of NF-κB signaling and promoting pro-inflammatory responses [8] [16]. This mechanism has been implicated in the pathological inflammation observed during severe SARS-CoV-2 infection, where caspase-8-dependent cleavage of N4BP1 drives excessive cytokine release independent of apoptotic cell death [8].
Regulation of Inflammatory Cytokines and Cell Death Crosstalk
Caspase-8 participates in the regulation of inflammatory cytokines through both direct and indirect mechanisms. It can directly process pro-IL-1β to its active form, creating a pathway for IL-1β maturation that operates independently of canonical inflammasome activation [17]. Additionally, caspase-8 regulates the transcription of numerous pro-inflammatory cytokines and chemokines through its scaffolding role in NF-κB activation [18].
The position of caspase-8 at the crossroads of multiple cell death pathways enables it to function as a critical arbitrator of cellular fate. Caspase-8 inhibits necroptosis by cleaving key necroptotic mediators including RIPK1 and RIPK3, thereby preventing the assembly of the necrosome and activation of MLKL [19] [20]. In macrophage models of atherosclerosis, inhibition of caspase-8 shifts the balance from apoptosis toward necroptosis, resulting in expanded necrotic cores within atheroma plaques due to impaired clearance of dead cells [20]. This demonstrates how caspase-8's regulatory function extends beyond single-cell fate decisions to influence tissue-level pathophysiology.
Table 1: Quantitative Parameters of Caspase-8 in Apoptotic vs. Non-Apoptotic Functions
| Parameter | Apoptotic Function | Non-Apoptotic Function | Experimental Evidence |
|---|---|---|---|
| Minimal activation threshold | <1% of total cellular caspase-8 sufficient to initiate apoptosis [21] | Not quantitatively determined | FRET-based biosensors in living single cells [21] |
| Auto-cleavage requirement | Required for full apoptotic activation [19] | Not required for scaffolding function [18] | Caspase-8 ΔE385 knock-in mice [19] |
| Primary molecular complexes | DISC homodimers [2] | FADDosome heterodimers with cFLIP [16] | Co-immunoprecipitation and complex analysis [18] |
| Key downstream substrates | Caspase-3, Caspase-7, Bid [21] | N4BP1, RIPK1, IL-1β [8] [17] | Western blot, cytokine measurements [8] [17] |
| Effect of catalytic inhibition | Blocks apoptosis [18] | Does not block inflammatory signaling [18] | Catalytically inactive mutants [18] |
Quantitative Analysis of Caspase-8 Functions
Table 2: Experimental Models for Studying Non-Apoptotic Caspase-8 Functions
| Experimental System | Key Findings | Methodological Approaches | References |
|---|---|---|---|
| SARS-CoV-2 infection models | Caspase-8 drives IL-1β-dependent inflammation independent of apoptosis | Gene-targeted mice (C8-/-/R3-/-), spatial transcriptomics, viral load measurement | [8] |
| Atherosclerosis models | Caspase-8 deficiency in macrophages shifts death from apoptosis to necroptosis, expanding necrotic cores | Bone marrow transplantation in Ldlr-/- mice, histological analysis, primary macrophage culture | [20] |
| TRAIL stimulation studies | Caspase-8 scaffolds FADDosome complex for NF-κB activation and cytokine production | Catalytically inactive caspase-8 mutants, affinity purification, cytokine arrays | [18] |
| Auto-cleavage mutant mice | Caspase-8 ΔE385 mutation impairs apoptosis but sensitizes to necroptosis | Knock-in mice, cell death assays, Western blot for phospho-MLKL | [19] |
| In vitro caspase-8 activity assays | Direct measurement of caspase-8 activity at DED filaments | Immunoprecipitation, fluorogenic substrates, Western blot | [2] |
Experimental Protocols and Methodologies
Protocol for Measuring Caspase-8 Activity at DED Filaments
The death-inducing signaling complex (DISC) serves as the primary activation platform for caspase-8, where procaspase-8 assembles into death effector domain (DED) filaments. This protocol enables specific measurement of caspase-8 activity within its native complex, providing insights into both apoptotic and non-apoptotic activation [2].
Key Steps:
- Cell Culture and Apoptosis Induction: Culture adherent cells of interest (e.g., HeLa, Jurkat) under standard conditions. Induce apoptosis using appropriate stimuli such as camptothecin (1-10 μM for 4-16 hours), Fas ligand (100 ng/mL for 2-8 hours), or TRAIL (50-100 ng/mL for 2-8 hours).
- DISC Immunoprecipitation: Harvest cells and lyse using mild non-denaturing lysis buffer (e.g., 1% CHAPS or 1% Triton X-100 in PBS) to preserve protein complexes. Incubate lysates with anti-FADD or anti-caspase-8 antibody conjugated to beads (4 μg antibody per 200 μg cell lysate) overnight at 4°C with gentle rotation.
- Caspase-8 Activity Assay: Wash immunoprecipitates thoroughly with lysis buffer. Resuspend beads in reaction buffer containing fluorogenic caspase-8 substrate (IETD-AFC or IETD-R110). Incubate at 37°C for 1-2 hours protected from light.
- Detection and Analysis: Measure fluorescence release (AFC: excitation 400 nm, emission 505 nm; R110: excitation 496 nm, emission 520 nm) using a plate reader. Normalize activity to protein concentration.
- Western Blot Validation: Analyze immunoprecipitates by SDS-PAGE and Western blotting for caspase-8 (proform: 55/50 kDa; cleaved: 40/36 kDa and 23 kDa) [22], FADD, and other complex components.
Applications: This protocol enables assessment of pharmacological inhibitors targeting caspase-8, differentiation between DISC-associated versus cytosolic caspase-8 activity, and investigation of caspase-8 activation in non-apoptotic contexts.
Protocol for Differentiating Scaffold versus Enzymatic Functions
To distinguish between caspase-8's scaffolding and enzymatic roles in inflammatory signaling, researchers can employ a combination of genetic and pharmacological approaches.
Key Steps:
- Genetic Models: Utilize caspase-8 null cells reconstituted with either wild-type or catalytically inactive caspase-8 (C362A or C362S mutants). Confirm expression levels by Western blot.
- Stimulation Conditions: Treat cells with TRAIL (50-100 ng/mL) or other death receptor ligands (TNF-α, FasL) for 4-24 hours in the presence or absence of broad-spectrum caspase inhibitors (z-VAD-FMK, 20-50 μM) or specific caspase-8 inhibitors (z-IETD-FMK, 10-50 μM).
- Inflammatory Readouts: Measure NF-κB activation by electrophoretic mobility shift assay (EMSA), luciferase reporter assays, or nuclear translocation. Quantify cytokine production (IL-1β, IL-6, TNF-α) by ELISA or multiplex assays.
- Cell Death Assessment: Evaluate apoptosis by Annexin V/propidium iodide staining and necroptosis by phospho-MLKL Western blot and propidium iodide uptake in the presence of caspase inhibition.
Interpretation: Inflammatory signaling in the presence of caspase inhibitors or in cells expressing catalytically inactive caspase-8 indicates scaffolding function, whereas dependence on catalytic activity suggests enzymatic role.
Signaling Pathway Visualization
Figure 1: Caspase-8 serves as a molecular switch directing cellular responses toward either inflammatory signaling or apoptotic death. Upon death receptor engagement, procaspase-8 is recruited to the receptor complex. When catalytic activity is inhibited or compromised, caspase-8 acts as a scaffold for RIPK1 recruitment, leading to NF-κB activation and cytokine production. When fully activated and cleaved, caspase-8 initiates apoptotic cascades.
Figure 2: Experimental workflow for measuring caspase-8 activity at DED filaments within the native death-inducing signaling complex (DISC). This protocol enables specific assessment of caspase-8 activation in its physiological context, crucial for distinguishing between its various functional roles.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for Caspase-8 Studies
| Reagent Category | Specific Examples | Applications | Considerations |
|---|---|---|---|
| Caspase-8 Antibodies | Purified Mouse Anti-Human Caspase-8 (Clone 3-1-9) [22] | Western blot (0.125-0.5 μg/mL), Immunoprecipitation (4 μg/200μg lysate) | Recognizes proform (55/50 kDa) and cleaved forms (40/36 kDa, 23 kDa) |
| Chemical Inhibitors | z-IETD-fmk (carbobenzoyl-Ile-Glu-Thr-Asp-fluoromethylketone) [21], Emricasan [8] | Inhibition of caspase-8 enzymatic activity | z-IETD-fmk not entirely specific for caspase-8; emricasan is broad-spectrum |
| Genetic Models | Casp8-/-/Ripk3-/- mice [8], Casp8ΔE385/ΔE385 knock-in [19], Casp8komac (myeloid-specific) [20] | Cell-type specific functions, differentiation of scaffolding vs catalytic roles | Embryonic lethality of Casp8-/- requires RIPK3 co-deletion to study postnatal functions |
| Activity Assays | Fluorogenic substrates (IETD-AFC, IETD-R110) [2], FRET-based biosensors [21] | Quantitative activity measurement in vitro and in live cells | FRET sensors enable single-cell dynamics; fluorogenic substrates suitable for population assays |
| Cell Lines | Jurkat T-cells (caspase-8 expression model) [22], Caspase-8 null cells reconstituted with mutants [18] | Structure-function studies, signaling pathway analysis | Reconstitution systems allow precise determination of functional domains |
The investigation of caspase-8's non-apoptotic functions, particularly in inflammatory signaling, has substantially expanded our understanding of this multifunctional protein. The emerging paradigm positions caspase-8 as a critical molecular switch that integrates signals from various pathways to determine cellular fate, directing outcomes toward apoptosis, necroptosis, or inflammatory activation depending on cellular context and activation status.
For researchers in the field of extrinsic pathway research, these insights necessitate more sophisticated experimental approaches that can differentiate between caspase-8's proteolytic and scaffolding functions, measure its activation in specific subcellular compartments, and account for its complex interactions with regulatory partners such as cFLIP. The protocols and reagents detailed in this application note provide a foundation for such investigations, enabling more precise dissection of caspase-8's diverse roles in physiological and pathological processes.
The therapeutic implications of targeting caspase-8 in inflammatory diseases continue to grow, with potential applications in autoimmune conditions, neurodegenerative diseases, sepsis, and severe viral infections such as COVID-19 [8] [16]. Future research directions should focus on developing strategies to selectively modulate specific aspects of caspase-8 function—such as inhibiting its enzymatic activity while preserving its scaffolding role—to achieve therapeutic efficacy without compromising essential homeostatic functions.
Protocols and Techniques for Measuring Caspase-8 Activity
Within the extrinsic apoptotic pathway, the formation of the Death-Inducing Signaling Complex (DISC) is the critical initiating event triggered by the ligation of death receptors, a subset of the TNF receptor superfamily [5]. The core components of the DISC include the trimerized death receptor, the adapter protein FADD (Fas-associated via death domain), and the initiator caspase-8 (procaspase-8) [5]. Caspase-8 is an aspartate-specific cysteine protease that is recruited to the complex as an inactive zymogen (procaspase-8). Its activation at the DISC occurs through proximity-induced dimerization and autoproteolysis, initiating the caspase cascade that leads to apoptotic execution [5] [21]. This protocol details methodologies for the native immunoprecipitation of the intact DISC and subsequent assays to quantify caspase-8 activity, providing crucial tools for research focused on modulating the extrinsic cell death pathway in therapeutic contexts, such as cancer and inflammatory diseases.
Quantitative Data on Key Apoptotic Components
Table 1: Key Protein Components in the Extrinsic Apoptotic Pathway
| Component | Function/Molecular Role | Key Characteristics & Quantitative Insights |
|---|---|---|
| Caspase-8 (CASP8) | Initiator cysteine protease; executes extrinsic apoptosis and inhibits necroptosis [5]. | Activated by homodimerization in the DISC [5]. Less than 1% of total cellular procaspase-8 is sufficient to initiate apoptosis [21]. |
| FADD | Adapter protein; bridges death receptor and procaspase-8 via homotypic domain interactions [5]. | Recruited to death receptors via its C-terminal death domain (DD). Binds procaspase-8 via its N-terminal death effector domain (DED) [5]. |
| Death Receptors | Trigger DISC assembly; include TNFR1, CD95/Fas, TRAIL-R1/2 [5]. | Ligation by cognate ligands (e.g., TNF, FasL, TRAIL) induces receptor trimerization and DISC nucleation [5]. |
| Procaspase-8 Zymogen | Inactive precursor of caspase-8 [5]. | Consists of two DEDs, a large (p18/p20) and a small (p10/p12) protease subunit. Autoproteolytically cleaves at D384 (human) and D387 (mouse) for activation [5]. |
| cFLIP | Regulatory protein; modulates caspase-8 activation [5]. | Encoded by CFLAR. Can heterodimerize with procaspase-8 at the DISC, influencing whether apoptosis or survival signals are generated [5]. |
Table 2: Recommended Lysis Buffer Compositions for DISC Immunoprecipitation
| Lysis Buffer Type | Composition | Recommended Application |
|---|---|---|
| Mild Lysis Buffer (e.g., NP-40) | 150 mM NaCl, 1% NP-40, 50 mM Tris-HCl (pH 8.0), 0.15% (w/v) BSA, 10% (v/v) Glycerol, Protease/Phosphatase Inhibitors [23]. | Ideal for native co-IP of membrane-localized or cytoplasmic protein complexes like the DISC. Preserves protein-protein interactions [23]. |
| Harsh Lysis Buffer (e.g., RIPA) | 50 mM Tris-HCl (pH 8.0), 150 mM NaCl, 1% NP-40, 0.5% Sodium Deoxycholate, 0.1% SDS, Protease/Phosphatase Inhibitors [23]. | Use for efficient lysis and extraction of nuclear or tightly bound proteins. May disrupt weaker protein interactions [23]. |
Experimental Protocols
Stage 1: Cell Culture, Stimulation, and Lysate Preparation
Materials:
- Cells of interest (e.g., HeLa, Jurkat)
- Appropriate cell culture medium
- Death receptor ligand (e.g., recombinant TRAIL, FasL)
- Ice-cold Phosphate-Buffered Saline (PBS)
- Selected lysis buffer (See Table 2), supplemented with protease and phosphatase inhibitor cocktails [23].
Procedure:
- Cell Stimulation: Culture and expand cells to the desired density. To induce DISC formation, treat cells with the appropriate death receptor ligand (e.g., 100 ng/mL TRAIL) for a time-course experiment (e.g., 0, 5, 15, 30, 60 minutes). Include an unstimulated control.
- Cell Harvesting and Washing: Collect cells by gentle scraping (adherent cells) or centrifugation (suspension cells). Wash the cell pellet 2-3 times with ice-cold PBS to remove residual media and serum [23].
- Cell Lysis: Resuspend the final cell pellet in ice-cold lysis buffer (e.g., 300 µL per 1–3 x 10⁷ cells). Incubate the suspension on ice for 10-30 minutes with occasional gentle vortexing to ensure complete lysis [23].
- Clarification: Centrifuge the lysate at high speed (8,000–12,000 x g) for 10 minutes at 4°C to pellet insoluble cellular debris. Carefully transfer the supernatant (the whole-cell lysate) to a fresh, pre-chilled tube [23].
- Protein Quantification: Determine the protein concentration of the lysate using a Bradford or BCA assay. Aliquot and snap-freeze lysates in liquid nitrogen for long-term storage at -80°C if not used immediately [23].
Stage 2: Immunoprecipitation of the Native DISC
Materials:
- Clarified cell lysate
- Antibody specific to the death receptor (e.g., anti-Fas) or a core DISC component (e.g., anti-caspase-8)
- Isotype control antibody (critical negative control)
- Protein A/G-coupled agarose or magnetic beads
- IP wash buffer (e.g., lysis buffer without BSA/glycerol)
Procedure:
- Pre-clearing (Optional): To reduce non-specific binding, incubate the lysate with protein beads alone or beads coupled with an isotype control antibody for 30-60 minutes at 4°C. Remove the beads by centrifugation [23].
- Antibody-Bead Complex Formation: While the lysate is pre-clearing, incubate the specific antibody (2-5 µg per 500 µg lysate) with the protein A/G beads for at least 1 hour at 4°C on a rotator. This pre-binding can increase capture efficiency.
- Immunoprecipitation: Incubate the pre-cleared lysate with the antibody-bound beads for 2-4 hours (or overnight) at 4°C with constant rotation.
- Washing: Pellet the beads gently and carefully aspirate the supernatant. Wash the beads 3-5 times with 1 mL of ice-cold wash buffer to remove non-specifically bound proteins. Resuspend the beads gently during each wash.
- Elution: After the final wash, completely remove the wash buffer. Elute the bound proteins from the beads by boiling in 2X Laemmli SDS-PAGE sample buffer for 5-10 minutes, or use a low-pH elution buffer followed by neutralization.
Stage 3: Caspase-8 Activity Assays
Method 1: In Vitro Caspase-8 Activity Assay from Immunoprecipitates
- Perform the DISC IP as described in Stage 2, but elute the proteins in a compatible activity assay buffer (e.g., 20 mM HEPES pH 7.4, 150 mM NaCl, 1% Triton X-100, 10% Glycerol).
- Incubate the eluted IP sample with a caspase-8-specific fluorogenic substrate (e.g., IETD-AFC or IETD-AMC).
- Measure the fluorescence emission (e.g., AFC: Ex ~400 nm, Em ~505 nm) over 30-120 minutes using a plate reader. The rate of fluorescence increase is proportional to caspase-8 activity.
Method 2: FRET-Based Caspase-8 Activity Monitoring in Live Cells
- Transfect cells with a FRET-based biosensor sensitive to caspase-8 cleavage (e.g., a construct with CFP and YFP linked by an IETD sequence) [21].
- Stimulate cells with the death receptor ligand while monitoring under a fluorescence microscope equipped with FRET filters.
- Caspase-8 activation and cleavage of the biosensor will result in a loss of FRET efficiency, quantified as a change in the CFP/YFP emission ratio over time, allowing for real-time, single-cell kinetic analysis of caspase-8 activation [21].
Experimental Workflow and Signaling Pathway Visualization
Caspase-8 Apoptosis Pathway
DISC IP and Activity Assay Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for DISC IP and Caspase-8 Assays
| Reagent / Material | Function / Application | Critical Notes for Experimental Success |
|---|---|---|
| Anti-Death Receptor or Anti-Caspase-8 Antibody | Target-specific capture for immunoprecipitation. | The key determinant of success. Must be validated for IP and capable of recognizing the native, non-denatured protein [23]. |
| Protein A/G-coupled Beads | Solid-phase matrix for antibody-antigen complex isolation. | Choose agarose for high binding capacity or magnetic beads for ease of washing and automation. |
| Protease & Phosphatase Inhibitor Cocktails | Preserve protein integrity and phosphorylation states during lysis. | Essential add-on to any lysis buffer to prevent proteolytic degradation and maintain post-translational modification signatures [23]. |
| Non-ionic Detergent (NP-40/Triton X-100) | Solubilizes cell membranes while preserving protein-protein interactions. | The foundation of a native lysis buffer for co-IP studies. Avoid ionic detergents like SDS for this application [23]. |
| Caspase-8 Fluorogenic Substrate (e.g., IETD-AFC) | Quantitative measurement of caspase-8 enzymatic activity. | The IETD sequence provides specificity for caspase-8. The released fluorophore (AFC) allows highly sensitive detection. |
| FRET-based Caspase Biosensor | Real-time, live-cell imaging of caspase-8 activation kinetics. | Enables single-cell analysis and reveals population heterogeneity in apoptotic response, providing dynamic data not available from endpoint assays [21]. |
The extrinsic apoptotic pathway is a fundamental process responsible for programmed cell death initiated by external signals, playing critical roles in tissue homeostasis, immune response, and cancer development. Central to this pathway is caspase-8, an initiator cysteine protease that becomes activated at multi-protein complexes known as Death-Inducing Signaling Complexes (DISCs) formed upon stimulation of death receptors like CD95/Fas or TRAIL receptors [15] [24]. Upon activation, caspase-8 initiates a proteolytic cascade that leads to the cleavage and activation of effector caspases (e.g., caspase-3 and -7), resulting in the organized dismantling of the cell [25]. Traditional endpoint measurements of apoptosis, such as Western blotting or flow cytometry, provide limited insight into the kinetic aspects of caspase-8 activation and fail to capture the dynamic, single-cell heterogeneity of this process in living systems.
Förster Resonance Energy Transfer (FRET)-based biosensors represent a powerful technological advancement that enables real-time monitoring of caspase-8 activity with high spatiotemporal resolution in live cells. FRET is a distance-dependent quantum mechanical phenomenon where energy is transferred from an excited donor fluorophore to a suitable acceptor fluorophore when they are in close proximity (typically 1-10 nm) [26]. In caspase-8 FRET biosensors, the donor and acceptor fluorophores are linked by a caspase-8-specific cleavage sequence. Upon caspase-8 activation and subsequent cleavage of this linker, the physical separation of the fluorophores results in a measurable decrease in FRET efficiency, providing a direct readout of enzymatic activity [21]. This approach allows researchers to quantitatively track the dynamics of caspase-8 activation in individual living cells, capturing the precise timing, amplitude, and heterogeneity of apoptotic signaling that would be lost in population-averaged endpoint measurements.
Principles and Design of FRET-Based Biosensors for Caspase-8
Molecular Architecture of Caspase-8 FRET Biosensors
The design of FRET-based biosensors for caspase-8 monitoring typically follows a modular architecture consisting of three essential components: a donor fluorophore, an acceptor fluorophore, and a caspase-8-specific cleavage motif serving as the linker. Commonly used fluorophore pairs include CFP/YFP (Cyan Fluorescent Protein/Yellow Fluorescent Protein) or newer variants such as mTurquoise2/sYFP2 that offer improved brightness and photostability [21] [26]. The selection of the caspase-8 cleavage sequence is critical for biosensor specificity. While caspase-8 exhibits preference for the LETD (Leu-Glu-Thr-Asp) motif, it can also cleave other tetra-peptide sequences, and the exact sequence must be carefully validated to ensure selective recognition by caspase-8 over other related proteases [15].
In the intact, uncleaved biosensor, the close proximity of the donor and acceptor fluorophores enables efficient FRET, resulting in emission from the acceptor when the donor is excited. Upon caspase-8 activation and subsequent cleavage of the linker sequence, the physical separation of the fluorophores reduces FRET efficiency, leading to decreased acceptor emission and increased donor emission (Figure 1). This ratiometric measurement (donor emission/acceptor emission) provides an internal control that minimizes artifacts from variations in biosensor expression level, focus drift, or excitation intensity, thereby enabling more robust quantitative analysis of caspase-8 dynamics [26].
Key Advantages Over Conventional Detection Methods
FRET-based biosensors offer several significant advantages for studying caspase-8 kinetics compared to traditional methods. Unlike Western blotting, which requires cell lysis and provides a single snapshot of caspase-8 activation in a population of cells, FRET biosensors enable continuous monitoring of caspase-8 activity in individual living cells over time, capturing the inherent heterogeneity in apoptotic responses [21]. This capability is particularly valuable for identifying subpopulations of cells with distinct activation kinetics that might be masked in bulk measurements.
Compared to fluorescence-based assays using fluorogenic substrates, FRET biosensors provide superior spatial resolution, allowing researchers to track the subcellular localization and translocation of caspase-8 activity, such as its movement from DISC complexes to the cytosol [21]. The genetically encoded nature of these biosensors enables cell-type-specific expression and long-term tracking of the same cells throughout the apoptotic process. Furthermore, FRET-based measurements are performed without the need for cell permeabilization or fixation, which can introduce artifacts and preclude true kinetic analysis [26]. The non-radiative nature of FRET makes these biosensors safer to use than radioactive assays and allows for repeated measurements in the same cells over extended time periods.
Quantitative Framework for Caspase-8 Activation Kinetics
Experimentally Determined Kinetic Parameters
The development and validation of mathematical models for caspase-8 activation have yielded crucial quantitative insights into the dynamics of extrinsic apoptosis. Table 1 summarizes key experimentally determined parameters that define caspase-8 activation kinetics in living cells.
Table 1: Experimentally Determined Kinetic Parameters of Caspase-8 Activation
| Parameter | Value | Experimental System | Significance |
|---|---|---|---|
| Minimal Active Caspase-8 Required for Apoptosis Commitment | <1% of total cellular caspase-8 | HeLa cells [21] | Demonstrates signal amplification in apoptotic pathway |
| Caspase-8 Concentration in HeLa Cells | ~100 nM (molar concentration) | Quantitative immunoblotting [21] | Provides absolute quantification for mathematical modeling |
| Half-life of Activated Caspase-8 | Short (minutes scale) due to proteasomal degradation | TRAF2-mediated K48-ubiquitination [27] | Sets temporal threshold for apoptosis commitment |
| Caspase-8 DED Filament Formation | Critical for full activation | DISC immunoprecipitation [15] | Explains high cooperativity in activation kinetics |
These quantitative parameters reveal several important characteristics of caspase-8 signaling. The finding that less than 1% of total cellular caspase-8 is sufficient to initiate apoptosis demonstrates the remarkable signal amplification capacity of the apoptotic cascade [21]. The short half-life of activated caspase-8, controlled by TRAF2-mediated K48-linked polyubiquitination and subsequent proteasomal degradation, establishes a critical temporal threshold that determines whether initial caspase-8 activation leads to full apoptotic commitment or sublethal signaling [27]. This ubiquitination process essentially functions as a "shutoff timer" that sets a critical barrier for extrinsic apoptosis commitment.
Regulatory Mechanisms Controlling Caspase-8 Activity
Caspase-8 activation is tightly regulated through multiple mechanisms that establish thresholds for apoptotic commitment. The formation of death effector domain (DED) filaments at the DISC provides a structural basis for high cooperativity in caspase-8 activation, creating a switch-like response to death receptor stimulation [15]. The regulatory protein cFLIPL competes with caspase-8 for binding to DED filaments and can either inhibit or promote caspase-8 activation depending on its expression level and stoichiometric ratio with caspase-8 [28] [29]. In non-small cell lung carcinoma (NSCLC), overexpression of cFLIPL correlates with poor prognosis and contributes to apoptosis resistance by inhibiting caspase-8 activation at the DISC [29].
The balance between caspase-8 activation and inactivation creates a dynamic system that processes extrinsic signals into life-or-death decisions. Quantitative measurements using FRET biosensors have been instrumental in revealing that this regulatory network operates as a digital switch rather than a linear response system, with a sharp threshold that separates survival from apoptosis commitment [21] [27].
Experimental Protocol: Monitoring Caspase-8 Dynamics Using FRET Biosensors
Biosensor Expression and Cell Preparation
This protocol describes the implementation of FRET-based biosensors for monitoring caspase-8 activation kinetics in living cells, adapted from established methodologies for live-cell compound screening [30].
Materials:
- FRET-based caspase-8 biosensor (e.g., pSCAT3 or similar construct)
- Appropriate cell line (HeLa, HEK293T, or other relevant model)
- Culture medium (DMEM supplemented with 10% FBS and 1% penicillin-streptomycin)
- Transfection reagents (calcium phosphate, lipofectamine, or comparable)
- Poly-D-lysine-coated multi-well plates (96-well format for screening)
- Phosphate Buffered Saline (PBS)
- Hank's Balanced Salt Solution (HBSS) or live-cell imaging medium
- Apoptosis inducers (e.g., CD95L, TRAIL, or other death receptor agonists)
- Inhibitors (e.g., z-VAD-fmk pan-caspase inhibitor, specific caspase-8 inhibitors)
- Fluorescence microplate reader or live-cell imaging system with appropriate filter sets
Procedure:
- Cell Culture and Transfection: Grow cells to 60-70% confluency in appropriate culture conditions. Transfect with FRET biosensor DNA using preferred transfection method (calcium phosphate, lipofection, or electroporation). For calcium phosphate transfection, mix 5 μg DNA with 55 μL of 2 M CaCl₂ and add ddH₂O to 500 μL final volume. Add this solution dropwise to 500 μL of 2× HBS while vortexing, incubate 30-40 minutes at room temperature, then add dropwise to cells [30].
- Validation of Transfection Efficiency: After 24 hours, assess transfection efficiency using fluorescence microscopy. Proceed only if transfection efficiency exceeds 70% to ensure adequate signal for FRET measurements [30].
- Cell Plating: Harvest transfected cells using trypsin/EDTA, count with hemocytometer using trypan blue exclusion to assess viability, and plate onto poly-D-lysine-coated 96-well plates at optimized density (typically 20,000-50,000 cells/well depending on cell type). Allow cells to adhere for 12-24 hours before imaging.
Live-Cell Imaging and FRET Quantification
- Experimental Setup: Replace culture medium with HBSS or specialized live-cell imaging medium to reduce background fluorescence. Add apoptosis inducers and/or inhibitors according to experimental design. Include appropriate controls (untreated cells, caspase inhibitor-treated cells).
- Image Acquisition: Place plate in live-cell imaging system maintained at 37°C with 5% CO₂. Acquire images at regular intervals (typically 5-30 minutes depending on experimental timeframe) using appropriate filter sets: excitation filter for donor (e.g., 420DF20 for CFP), and emission filters for donor (e.g., 470DF40 for CFP) and acceptor (e.g., 535DF25 for YFP) [30].
- FRET Calculation: Calculate FRET ratio for each time point using the formula: FRET Ratio = (Acceptor Emission Intensity) / (Donor Emission Intensity). Normalize FRET ratios to initial baseline values (time = 0) to account for cell-to-cell variation in biosensor expression.
- Data Analysis: Plot normalized FRET ratio versus time to generate kinetic curves of caspase-8 activation. Determine key parameters such as time to activation onset, maximum activation rate, and time to half-maximal activation. Perform statistical analysis across biological replicates.
Figure 2: Experimental workflow for FRET-based monitoring of caspase-8 activation
Advanced Applications in Extrinsic Apoptosis Research
Integration with Complementary Assays
FRET-based monitoring of caspase-8 activation can be powerfully combined with complementary approaches to obtain a comprehensive view of apoptotic signaling. Multiplexing with mitochondrial membrane potential dyes (e.g., TMRM, JC-1) allows simultaneous tracking of the caspase-8 initiation phase and subsequent mitochondrial amplification of the death signal. Similarly, combination with plasma membrane integrity dyes (e.g., propidium iodide) enables discrimination between apoptotic and necroptotic cell death outcomes [24]. For high-content screening applications, FRET biosensors can be integrated with nuclear labeling dyes (e.g., Incucyte Nuclight reagents) to concurrently monitor apoptosis and proliferation in the same cell population [31].
Recent technological advances have enabled the combination of FRET-based caspase-8 sensors with optogenetic tools to achieve precise spatiotemporal control of death receptor activation. This approach allows researchers to initiate apoptotic signaling at defined cellular locations and timepoints, providing unprecedented resolution for studying signal propagation and compartmentalization. Furthermore, the adaptation of FRET biosensors to 3D culture systems, including patient-derived organoids, offers more physiologically relevant models for investigating caspase-8 dynamics in tissue-like contexts [25].
Investigation of Non-Apoptotic Functions and Therapeutic Applications
Beyond its canonical role in apoptosis execution, caspase-8 participates in various non-apoptotic processes, including regulation of necroptosis, T-cell activation, and cytokine production. FRET biosensors have been instrumental in revealing that sublethal caspase-8 activation can trigger inflammatory responses through NF-κB activation and IL-8 production without committing cells to apoptosis [28]. This paradigm is particularly relevant in cancer biology, where tumor cells under metabolic stress (e.g., glutamine limitation) may activate caspase-8-dependent inflammatory signaling that promotes tumor progression rather than cell death [28].
From a therapeutic perspective, FRET-based caspase-8 biosensors provide valuable tools for screening compounds that modulate the extrinsic apoptotic pathway. HDAC inhibitors such as vorinostat have been shown to downregulate cFLIP expression and promote caspase-8-dependent apoptosis in NSCLC cells, suggesting potential combination strategies with TRAIL receptor agonists [29]. The ability to perform live-cell kinetic screening with FRET biosensors enables identification of compounds that specifically lower the threshold for caspase-8 activation, potentially overcoming apoptosis resistance in cancer cells [30].
Research Reagent Solutions for Caspase-8 FRET Studies
Table 2: Essential Research Reagents for FRET-Based Caspase-8 Monitoring
| Reagent Category | Specific Examples | Function/Application |
|---|---|---|
| FRET Biosensors | pSCAT3, mTurquoise2-sYFP2 based constructs | Genetically encoded reporters for caspase-8 activity |
| Cell Lines | HeLa-CD95, HEK293T, HCT116, A549 | Model systems with defined death receptor expression |
| Apoptosis Inducers | CD95L/FasL, TRAIL, TNF-α | Activate extrinsic pathway through death receptors |
| Caspase Inhibitors | z-IETD-fmk (caspase-8 specific), z-VAD-fmk (pan-caspase) | Specificity controls and pathway inhibition studies |
| Detection Instruments | Fluorescence microplate readers, Confocal microscopes | FRET signal acquisition and live-cell imaging |
| Validation Antibodies | Anti-caspase-8 (clone C15), Anti-cleaved caspase-8, Anti-PARP | Western blot validation of caspase-8 activation |
| Specialized Assay Kits | Incucyte Caspase-3/7 Dyes, Annexin V conjugates | Multiplexing with complementary apoptosis markers |
The selection of appropriate research reagents is critical for successful implementation of FRET-based caspase-8 monitoring. The choice of biosensor should consider factors such as brightness, photostability, and specificity for caspase-8 versus related proteases. Cell lines should be validated for expression of relevant death receptors and downstream apoptotic machinery. For pharmacological studies, caspase inhibitors should be titrated to establish optimal concentrations that provide specific inhibition without off-target effects. Instrument selection should prioritize sensitivity for detecting small changes in FRET efficiency and environmental control for maintaining cell viability during extended time-lapse experiments.
Figure 3: Caspase-8 in extrinsic apoptosis and cFLIP regulation
FRET-based biosensors represent a transformative technology for investigating caspase-8 activation dynamics in live cells, providing unprecedented temporal resolution and single-cell kinetic data that have fundamentally advanced our understanding of extrinsic apoptosis regulation. The quantitative framework established through these approaches has revealed critical parameters such as the minimal caspase-8 threshold required for apoptosis commitment, the role of regulatory ubiquitination in controlling activation kinetics, and the existence of sublethal caspase-8 signaling that drives inflammatory responses. The continued refinement of these biosensors, combined with advanced imaging platforms and computational analysis methods, promises to further elucidate the complex regulatory networks that govern cell fate decisions in health and disease. As these tools become increasingly accessible and adaptable to physiologically relevant model systems, they will undoubtedly continue to drive discoveries in fundamental cell biology and therapeutic development for cancer and other diseases characterized by dysregulated apoptosis.
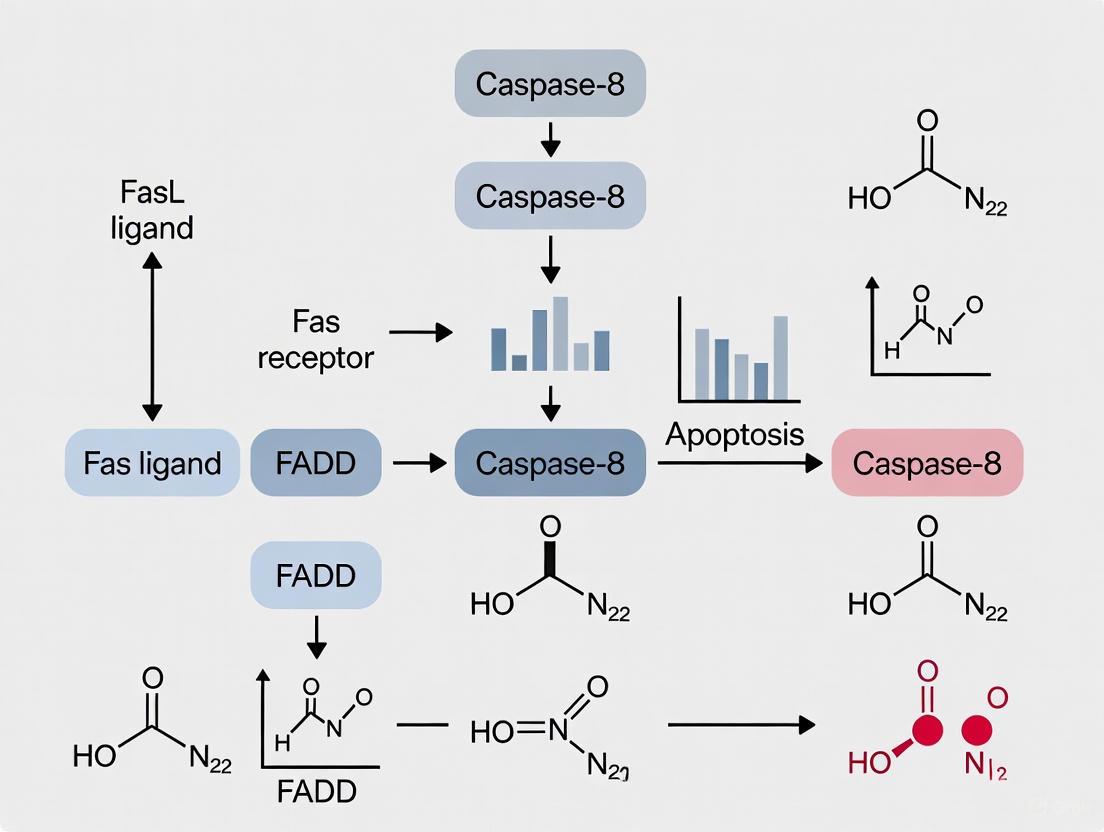
Caspase-8 plays a pivotal role as the initiator caspase in the extrinsic apoptosis pathway. This pathway is triggered when death ligands (e.g., CD95L/FasL) bind to their corresponding death receptors on the cell surface, leading to the formation of the Death-Inducing Signaling Complex (DISC) [15]. At the DISC, the adapter protein FADD recruits procaspase-8 via death effector domain (DED) interactions, facilitating its activation through proximity-induced dimerization and autocleavage [15]. The key stage of extrinsic apoptosis is the activation of procaspase-8 at the DISC, where it assembles into DED filaments [15]. Once activated, caspase-8 can cleave and activate downstream effector caspases (e.g., caspase-3, -6, -7), ultimately leading to the characteristic biochemical and morphological hallmarks of apoptosis [32]. Detecting the cleavage fragments of caspase-8 (p43/p41, p18, and p10) via Western blotting provides a critical readout for its activation status and is essential for research focused on measuring caspase-8 activation in extrinsic pathway research.
The following diagram illustrates the core signaling pathway of caspase-8 activation in extrinsic apoptosis, from the initial death ligand binding to the final cleavage events that can be detected by Western blot.
A successful Western blot experiment for detecting caspase-8 cleavage depends on the use of specific and validated reagents. The table below details essential research reagent solutions for this application.
Table 1: Key Research Reagents for Caspase-8 Western Blot Analysis
| Reagent / Resource | Source / Example Catalog Number | Specifications and Function in Detection |
|---|---|---|
| Cleaved Caspase-8 (Asp387) Antibody | Cell Signaling Technology (#9429) [32] | Rabbit monoclonal; detects endogenous levels of caspase-8 cleaved at Asp387; recognizes p43 (pro-domain + p18) and p18 subunits; ideal for confirming activation. |
| Caspase-8 Antibody (for total protein) | Santa Cruz Biotechnology (sc-8009) [15] | Mouse monoclonal; detects total caspase-8 (full-length and large fragments); useful for assessing the ratio of cleaved to full-length protein. |
| Anti-caspase-8 (clone C15) | Scaffidi et al. [15] | Mouse monoclonal; well-cited antibody for immunoprecipitation and detection of caspase-8. |
| Anti-FADD (clone 1C4) | Muzi et al. [15] | Mouse monoclonal; used for immunoprecipitation of the DISC to study caspase-8 activation in its native complex. |
| Anti-c-FLIP (clone NF6) | Scaffidi et al. [15] | Mouse monoclonal; detects c-FLIP, a key regulator of caspase-8 activation at the DISC. |
| HRP-conjugated Secondary Antibodies | SouthernBiotech (e.g., #1070-05, #4030-05) [15] | Species-specific antibodies conjugated to Horseradish Peroxidase for chemiluminescent detection. |
| Positive Control Lysate | Apoptotic cell lysate (e.g., CD95L-treated HeLa-CD95 cells) [15] | Essential control to validate antibody specificity and the experimental workflow. |
Detailed Experimental Protocol
This protocol provides a step-by-step guide for detecting caspase-8 cleavage, from sample preparation to imaging, incorporating both standard and innovative methods.
Sample Preparation and Apoptosis Induction
- Cell Culture and Treatment: Culture appropriate cell lines, such as HeLa-CD95 cells which are sensitive to CD95L-induced apoptosis [15]. Seed cells at a density of 5 x 10^6 cells per 14.5 cm plate and allow them to adhere overnight.
- Induction of Apoptosis: To activate the extrinsic pathway, treat cells with a death ligand such as CD95L for a predetermined time course (e.g., 0, 1, 2, 4, 8 hours) to capture the kinetics of caspase-8 activation [15]. Include an untreated control.
- Cell Lysis: Harvest cells and lyse them using a suitable lysis buffer, such as RIPA buffer, supplemented with protease and phosphatase inhibitors [33]. Determine the protein concentration of the lysate using a BCA assay kit, normalizing all samples to a consistent concentration (e.g., 1-2 µg/µL) [33].
Gel Electrophoresis and Protein Transfer
- Gel Preparation: Prepare an appropriate resolving gel (e.g., 8-12% acrylamide) to optimally separate caspase-8 cleavage products (full-length: 55/57 kDa; p43/p41: 43 kDa; p18: 18 kDa) [32] [33].
- Sample Loading: Mix normalized protein lysates (recommended load: 10-30 µg per well) with Laemmli sample buffer containing a reducing agent like β-mercaptoethanol [15]. Heat denature samples at 95°C for 5 minutes. Load samples alongside a pre-stained protein molecular weight marker.
- Electrophoresis: Run the gel in SDS-PAGE running buffer at a constant voltage until the dye front reaches the bottom of the gel.
- Protein Transfer: Transfer proteins from the gel to a nitrocellulose membrane using a wet or semi-dry transfer system. Confirm successful and even transfer using a reversible stain like Ponceau S [33].
Immunodetection: Conventional and Sheet Protector Strategies
The core step of probing the membrane with antibodies can be performed using a conventional large-volume method or a novel, resource-saving Sheet Protector (SP) strategy [33].
Table 2: Comparison of Conventional vs. Sheet Protector Antibody Incubation Methods
| Parameter | Conventional Method | Sheet Protector (SP) Strategy |
|---|---|---|
| Volume Required | ~10 mL for a mini-gel membrane [33] | 20 - 150 µL, adjusted to membrane size [33] |
| Primary Antibody Incubation | Overnight at 4°C with gentle agitation [15] | 15 minutes to 2 hours at room temperature, without agitation [33] |
| Key Steps | Membrane is submerged in antibody solution in a container. | Membrane is blotted to semi-dry, placed on a sheet protector leaflet, covered with a small antibody volume, and overlaid with the top leaflet to form a sealed unit. |
| Advantages | Well-established and familiar protocol. | Drastically reduces antibody consumption (up to 99%); faster incubation; no specialized equipment needed. |
| Considerations | High antibody consumption; long incubation time. | Requires optimization of antibody concentration; potential for uneven drying if not sealed properly. |
General Immunodetection Steps:
- Blocking: Incubate the membrane in 5% skim milk or BSA in TBST for 1 hour at room temperature with gentle rocking to block non-specific binding sites.
- Primary Antibody Incubation: Probe the membrane with the primary antibody against cleaved caspase-8 (e.g., #9429 at 1:1000 dilution [32]) or total caspase-8 using either the conventional or SP method as described in Table 2. If using the SP strategy for the first time, a concentration of 0.2 µg/mL is recommended as a starting point to achieve signal intensity comparable to the conventional method with 0.1 µg/mL [33].
- Washing: Wash the membrane three times with TBST for 5 minutes per wash.
- Secondary Antibody Incubation: Incubate the membrane with an HRP-conjugated secondary antibody (e.g., 1:2000 to 1:10000 dilution in 5% milk/TBST) for 1 hour at room temperature with agitation. This step is typically performed in a container.
- Washing: Repeat the washing step as after the primary antibody.
Signal Detection and Data Analysis
- Chemiluminescent Detection: Treat the membrane with a chemiluminescent HRP substrate according to the manufacturer's instructions. Image the membrane using a digital imaging system, such as an ImageQuant LAS-4000 mini, ensuring to capture multiple exposure times without signal saturation [33].
- Normalization and Quantification: For accurate quantification, normalize the signal intensity of the caspase-8 cleavage products to a total protein loading control. Total Protein Normalization (TPN) is now considered the gold standard over housekeeping proteins (HKPs) like GAPDH or actin, as HKP expression can be variable [34]. TPN can be achieved by using a reversible total protein stain (e.g., No-Stain Protein Labeling Reagent) on the membrane before immunodetection or by post-staining after imaging [35] [34].
- Densitometric Analysis: Analyze the band intensities using software such as FIJI (ImageJ). Calculate the ratio of cleaved caspase-8 (p43/p41 or p18) to total caspase-8 or to the total protein load to quantitatively assess activation [33].
The following workflow diagram summarizes the entire protocol, highlighting the key decision point between the conventional and SP methods.
Data Presentation and Publication Standards
Adherence to journal guidelines is critical for publishing robust and credible Western blot data. Leading journals now emphasize quantitative rigor and image integrity.
- Quantitative Normalization: As mentioned, prefer Total Protein Normalization (TPN). Journals are increasingly discouraging reliance on single housekeeping proteins due to their variable expression under different experimental conditions [34].
- Image Acquisition and Processing: Use digital imaging systems that provide a wide linear dynamic range (at least 4 logs) to ensure signals are proportional to protein abundance and not saturated [35]. Avoid over-cropping images and always retain molecular weight markers. Any image adjustments (e.g., brightness/contrast) must be applied uniformly to the entire image and never obscure, eliminate, or misrepresent any information [34].
- Data Transparency: Be prepared to provide original, uncropped blot images to journals upon request. Clearly indicate in figure legends if lanes from different parts of the same gel or from different blots have been reassembled [34].
Table 3: Summary of Caspase-8 Cleavage Products and Their Detection
| Protein Species | Approximate Molecular Weight | Biological Significance | Detected by Cleaved Caspase-8 (Asp387) Ab? |
|---|---|---|---|
| Procaspase-8 (full-length) | 55 / 57 kDa (isoforms) | Inactive zymogen form. | No [32] |
| p43 / p41 | 43 / 41 kDa | Initial cleavage intermediate, contains the pro-domain and the p18 subunit; indicates initial activation. | Yes (p43) [32] |
| p18 | 18 kDa | Large subunit of the active enzyme; definitive marker of activation. | Yes [32] |
| p10 | 10 kDa | Small subunit of the active enzyme. | No (Antibody #9429 is raised against the p18 subunit) [32] |
Troubleshooting and Pro-Tips
- No Signal: Confirm antibody specificity and compatibility with your species. Check the expiration of your chemiluminescent substrate. Ensure your positive control (e.g., CD95L-treated cells) is working.
- High Background: Increase the number and duration of washes. Optimize the concentration of primary and secondary antibodies. Ensure the blocking solution is fresh and effective.
- Non-Specific Bands: Verify the antibody's stated specificity. Use a blocking peptide (if available) to confirm the target band. Ensure samples are not degraded.
- Pro-Tip for Antibody Conservation: The Sheet Protector (SP) strategy is highly recommended for initial optimization and routine use with precious or expensive antibodies, as it can reduce consumption by up to 99% without compromising sensitivity [33].
In the context of caspase-8 activation research within the extrinsic apoptosis pathway, the analysis of downstream substrates provides the essential functional readouts that confirm pathway engagement and quantify cellular response. Caspase-8, the initiator caspase, acts as a molecular switch upon its activation at the Death-Inducing Signaling Complex (DISC). However, its activity is primarily confirmed by detecting the proteolytic cleavage of key downstream effector molecules. The cleavage of BID, caspase-3, and PARP serves as a definitive signature of successful signaling propagation, moving beyond mere initiator caspase activation to the actual execution of the apoptotic program. These substrates act as critical biomarkers, providing researchers with a quantifiable measure of cell death induction, which is vital for assessing drug efficacy, understanding resistance mechanisms, and evaluating toxicological outcomes in drug development.
Scientific Background: The Signaling Pathway from Initiation to Execution
Caspase-8: The Initiator Caspase
Caspase-8 is a crucial initiator caspase in the extrinsic death receptor pathway. It is activated upon ligation of death receptors like FAS, TNFR1, or TRAIL receptors. The process begins with the recruitment of the adapter protein FADD (Fas-associated protein with death domain), which then recruits procaspase-8 through homotypic death effector domain (DED) interactions, forming the DISC. Within the DISC, procaspase-8 molecules undergo proximity-induced dimerization and autoproteolytic cleavage, generating the fully active enzyme [5] [36]. This active caspase-8 then acts as the key upstream regulator that propagates the death signal by cleaving downstream substrates.
Key Downstream Substrates and Their Significance
The following diagram illustrates the core signaling pathway from caspase-8 activation to the cleavage of its key downstream substrates, BID, caspase-3, and PARP, culminating in the phenotypic outcomes of apoptosis.
The pathway delineates two primary routes for signal propagation from active caspase-8, culminating in the key functional readouts:
- Type I Pathway (Direct Activation): Active caspase-8 directly cleaves and activates executioner caspase-3 [5] [36].
- Type II Pathway (Mitochondrial Amplification): Active caspase-8 cleaves the BH3-only protein BID to its truncated active form (tBID). tBID then translocates to mitochondria, triggering MOMP and the release of cytochrome c, which leads to the formation of the apoptosome and activation of caspase-9, which in turn activates caspase-3 [9] [5] [37].
- Execution Phase: Activated caspase-3 cleaves a multitude of substrates, including PARP. Cleavage of PARP inactivates its DNA repair function and serves as a hallmark of irreversible commitment to apoptosis [38] [39].
The table below summarizes key quantitative findings and functional significance for each of the primary downstream substrates from published research.
Table 1: Quantitative Data and Functional Significance of Key Apoptotic Substrates
| Substrate | Cleavage Form Detected | Key Quantitative Findings | Functional Consequence of Cleavage |
|---|---|---|---|
| BID | Truncated BID (tBID) | • tBid activates Bax/Bak to induce MOMP [37].• In Type II cells, Bid cleavage links extrinsic to intrinsic pathway [5]. | • Activation of mitochondrial apoptosis pathway.• Amplification of initial death signal. |
| Caspase-3 | Active caspase-3 (17/12 kDa fragments) | • >5% active caspase-3+ Reed-Sternberg cells predicted favorable clinical outcome in Hodgkin's disease (P=.007) [40].• Active caspase-3 drives a positive feedback loop enhancing Bid activation [37]. | • Executioner caspase activity; cleaves ~100s of cellular proteins.• Key marker for cells undergoing apoptosis. |
| PARP | Cleaved PARP (89 kDa fragment) | • Cleavage by caspase-3 inactivates PARP's DNA repair function [38] [39].• Detection of PARP-1/p89 fragment correlates with active caspase-3 presence [40]. | • Inactivation of DNA repair.• Facilitation of cellular dismantling.• Hallmark of irreversible apoptosis commitment. |
Experimental Protocols & Workflows
This section provides detailed methodologies for assessing these key functional readouts, from sample preparation to data analysis.
Workflow for a Multi-Readout Apoptosis Assay
The following diagram outlines a generalized experimental workflow for the simultaneous analysis of multiple downstream substrates, integrating the specific protocols detailed thereafter.
Protocol 1: Flow Cytometric Analysis of Active Caspase-3
This protocol allows for the quantification of the percentage of cells undergoing apoptosis within a population by detecting active caspase-3 [41] [42].
Key Reagents:
- Inducer: Camptothecin (4-6 µM final concentration) as a positive control [41].
- Antibody: FITC-conjugated antibody specific for the active form of caspase-3 (does not recognize the pro-enzyme) [41].
- Buffers: BD Cytofix/Cytoperm Solution (contains 4.2% formaldehyde; handle with appropriate precautions), BD Perm/Wash Buffer (1X) [41].
Step-by-Step Procedure:
- Induction and Harvest: Treat cells (e.g., 1x10⁶/mL Jurkat cells) with your apoptotic stimulus or vehicle control for a predetermined time (e.g., 4 hours for camptothecin). Wash cells twice with cold PBS [41].
- Fixation and Permeabilization: Resuspend the cell pellet in 0.5 mL of BD Cytofix/Cytoperm Solution. Incubate for 20 minutes on ice [41].
- Washing: Pellet the cells, carefully aspirate the fixative solution, and wash twice with 0.5 mL of BD Perm/Wash Buffer (1X) per 1x10⁶ cells [41].
- Staining: Resuspend the cell pellet in 100 µL of BD Perm/Wash Buffer (1X) containing a pre-determined optimal concentration of the anti-active caspase-3 FITC antibody (e.g., 20 µL per test). Incubate for 30 minutes at room temperature, protected from light [41].
- Final Wash and Analysis: Wash the cells once with 1.0 mL of BD Perm/Wash Buffer (1X). Resuspend in 0.5 mL of buffer and analyze by flow cytometry. Use untreated and single-stained controls for proper gating and compensation [41].
Protocol 2: Western Blot Analysis for BID, Caspase-3, and PARP Cleavage
This protocol provides a multi-substrate assessment, confirming specific proteolytic cleavage events via molecular weight shifts.
Key Reagents:
- Primary Antibodies: Anti-BID (to detect full-length and tBID), anti-caspase-3 (to detect pro-form and cleaved 17/12 kDa fragments), anti-PARP (to detect full-length ~116 kDa and cleaved ~89 kDa fragment) [37] [40] [39].
- Lysis Buffer: A suitable RIPA buffer supplemented with protease and phosphatase inhibitors.
- Gel Electrophoresis: Standard SDS-PAGE equipment and transfer system.
Step-by-Step Procedure:
- Sample Lysis: Lyse harvested cells in an appropriate volume of ice-cold lysis buffer. Centrifuge at high speed (e.g., 12,000-15,000 x g) for 10 minutes at 4°C to remove insoluble material. Determine the protein concentration of the supernatant.
- Gel Electrophoresis and Transfer: Load 20-40 µg of total protein per lane onto an SDS-PAGE gel (e.g., 12-15% gradient gel for optimal separation of cleavage fragments). Separate proteins by electrophoresis and transfer to a PVDF or nitrocellulose membrane.
- Blocking and Antibody Incubation: Block the membrane with 5% non-fat milk or BSA in TBST for 1 hour at room temperature. Incubate with primary antibodies diluted in blocking buffer overnight at 4°C. Wash the membrane and incubate with an appropriate HRP-conjugated secondary antibody for 1 hour at room temperature.
- Detection: Develop the blot using a enhanced chemiluminescence (ECL) substrate and image with a digital imager. The cleavage of each substrate is confirmed by the appearance of the lower molecular weight band (tBID for BID, 17/12 kDa for caspase-3, 89 kDa for PARP) alongside or in place of the full-length protein.
Protocol 3: Live-Cell Imaging with FRET Reporters
This technique allows for real-time, kinetic analysis of caspase activity in live cells, providing dynamic data on the timing and heterogeneity of apoptosis.
Key Reagents:
- Reporters: Genetically encoded FRET-based constructs.
- Equipment: Live-cell fluorescence microscope with environmental control (CO₂, temperature, humidity) and appropriate filter sets for CFP and YFP.
Step-by-Step Procedure:
- Cell Preparation: Stably or transiently transfert cells with the desired FRET reporter (IC-RP to monitor initiator activity, EC-RP to monitor effector activity).
- Imaging Setup: Plate reporter cells in a glass-bottom dish and allow to adhere. Place the dish on the pre-warmed microscope stage. Establish imaging conditions (e.g., take images every 3-10 minutes to minimize phototoxicity) [9].
- Induction and Acquisition: Add the apoptotic stimulus directly to the dish during imaging. Continuously acquire CFP and FRET (YFP) channel images over the course of the experiment (e.g., 8-12 hours) [9].
- Data Analysis: Calculate the CFP/FRET (or YFP) emission ratio for each cell over time. Cleavage of the linker by the active caspase separates the FRET pair, resulting in a decrease in FRET and a corresponding increase in the CFP/FRET ratio. This kinetic trace reveals the precise timing of caspase activation for individual cells.
The Scientist's Toolkit: Essential Research Reagents
The table below catalogs key reagents and tools essential for conducting experiments focused on downstream apoptotic substrates.
Table 2: Key Research Reagents for Assessing Apoptotic Substrates
| Reagent/Tool | Specific Example | Function & Application Note |
|---|---|---|
| Active Caspase-3 Kits | BD Pharmingen FITC Active Caspase-3 Apoptosis Kit [41] | Flow cytometry-based quantification of apoptotic cells. Validated for human and mouse cells. |
| Selective Antibodies | Anti-cleaved caspase-3 (17/12 kDa); Anti-cleaved PARP (89 kDa); Anti-tBID [37] [40] [42] | Essential for Western blot and immunohistochemistry to confirm specific proteolytic events. |
| Live-Cell Reporters | EC-RP (Caspase-3 Reporter); IC-RP (Caspase-8 Reporter) [9] | FRET-based plasmids for real-time, kinetic analysis of caspase activity in live cells. |
| Pathway Inducers | Recombinant TRAIL/TNF-α; Agonistic anti-FAS antibody; Camptothecin [9] [41] | Positive control stimuli to reliably trigger the extrinsic or intrinsic apoptosis pathway. |
| Caspase Inhibitors | Z-VAD-FMK (pan-caspase inhibitor); Z-DEVD-FMK (caspase-3 inhibitor) | Essential control compounds to confirm caspase-dependent mechanisms. |
Optimizing Assay Conditions and Overcoming Common Challenges
The extrinsic apoptotic pathway is initiated when extracellular death ligands, such as CD95L or TRAIL, bind to their cognate death receptors (e.g., CD95/Fas), leading to the assembly of the Death-Inducing Signaling Complex (DISC) [15] [13]. At the heart of the DISC, procaspase-8 molecules oligomerize into death effector domain (DED) filaments, a key step that drives its activation [15]. Active caspase-8 then acts as the initiator caspase, propagating the death signal. In Type I cells, it directly cleaves and activates executioner caspases like caspase-3, while in Type II cells, the signal is amplified through the mitochondrial pathway via cleavage of the BID protein [13]. Given its pivotal role, precise measurement of caspase-8 activity at the DISC is fundamental for research in programmed cell death, cancer biology, and the development of novel anti-cancer therapeutics that seek to modulate this pathway [15] [43]. This application note details a robust protocol for measuring caspase-8 activation, emphasizing the critical experimental controls—beads control, cell viability, and specificity—that are essential for generating reliable and interpretable data.
Experimental Protocol: Measuring Caspase-8 Activity at the DED Filaments
The following protocol, adapted from König et al., outlines the steps for measuring caspase-8 activity in its native complex, from cell culture to data analysis [15] [2].
Materials and Reagents
- Cell Line: HeLa-CD95 cells (or other adherent/suspension cells sensitive to CD95L-induced apoptosis).
- Culture Medium: DMEM F12 supplemented with 10% Fetal Calf Serum (FCS), 0.1 mg/mL Penicillin/Streptomycin, and 0.2 μg/mL puromycin.
- Apoptosis Inducer: Recombinant CD95L [15].
- Lysis & Immunoprecipitation (IP) Buffer: Contains CHAPS, HEPES, NaCl, EDTA, and Triton X-100.
- Antibodies for IP: Anti-CD95 (e.g., mouse monoclonal anti-CD95, sc-8009) and anti-APO-1 [15].
- Protein A/G or Anti-Mouse IgG Coated Magnetic Beads: For immunoprecipitation.
- Caspase-8 Activity Assay Buffer: Contains HEPES, NaCl, sucrose, CHAPS, and DTT.
- Caspase-8 Fluorogenic Substrate: e.g., Ac-IETD-AFC.
- Western Blot Antibodies: Anti-caspase-8 (clone C15), anti-FADD (clone 1C4), anti-c-FLIP (clone NF6), anti-caspase-3, anti-PARP1 [15].
Step-by-Step Procedure
Cell Culture and Preparation (Timing: 2-3 days)
- Seed ( 2 \times 10^6 ) HeLa-CD95 cells in a T175 flask in 30 mL of complete medium and culture for two days at 37°C with 5% CO(_2) [15].
- On the day of the experiment, detach cells using trypsin, neutralize with medium, and centrifuge at 500 × g for 5 min.
- Resuspend the cell pellet in fresh medium and count using Trypan Blue staining. CRITICAL: Proceed only if cell viability exceeds 93% [15].
- Seed ( 5 \times 10^6 ) cells per 14.5 cm plate (in 20 mL medium) for experimental conditions. Include triplicates for Western Blot control and caspase-8 assay duplicates. Seed one additional plate for the "Beads control" [15].
- Incubate plates overnight.
Induction of Apoptosis (Timing: ~30 min)
- Stimulate cells with an appropriate concentration of CD95L (e.g., 100-500 ng/mL) for a predetermined time (e.g., 5-30 min) to activate the DISC. Include unstimulated controls.
Cell Lysis and Immunoprecipitation (IP) of the DISC (Timing: ~3 hours)
- Place culture plates on ice, discard medium, and wash cells with ice-cold PBS.
- Lyse cells in IP lysis buffer for 30 minutes on a rocking platform at 4°C.
- Scrape adherent cells and transfer the lysate to a precooled tube. Centrifuge at high speed (e.g., 16,000 × g) for 15 min at 4°C to remove insoluble debris.
- CRITICAL STEP: Split the supernatant for two purposes: one aliquot for direct Western Blot analysis (input control), and the other for DISC immunoprecipitation.
- Incubate the IP aliquot with anti-CD95 antibody (e.g., 1-2 μg) for 2 hours at 4°C with rotation.
- Add pre-washed magnetic beads (e.g., 25-50 μL bead slurry) and incubate for an additional 1-2 hours.
- Pellet the beads using a magnet and carefully aspirate the supernatant. Wash the beads 3-4 times with ice-cold lysis buffer.
Caspase-8 Activity Assay (Timing: ~1-2 hours)
- Resuspend the IP complex (beads-bound DISC) in caspase-8 assay buffer.
- Add the fluorogenic substrate Ac-IETD-AFC (final concentration ~50-100 μM).
- Incubate the reaction at 37°C and measure the fluorescence (excitation ~400 nm, emission ~505 nm) at regular intervals (e.g., every 5-10 min) for up to 1-2 hours using a plate reader.
- CRITICAL: Run the "Beads control" sample in parallel. This sample consists of beads incubated with lysate from unstimulated cells or a no-antibody control, processed identically to the test samples.
Western Blot Analysis (Timing: ~1 day)
- After the activity assay, denature the IP samples and input lysates in Laemmli buffer by boiling for 5-10 min.
- Separate proteins by SDS-PAGE and transfer to a PVDF membrane.
- Probe the membrane with antibodies against DISC components: caspase-8, FADD, c-FLIP, and the receptor (CD95) to confirm successful IP and specific protein recruitment [15].
The experimental workflow and the core signaling pathway being investigated are summarized in the diagrams below.
The Critical Control Framework
Rigorous experimental controls are non-negotiable for accurately interpreting caspase-8 activity data. The following controls address key sources of artifact and false positives.
Beads Control for Immunoprecipitation Specificity
The "Beads control" is designed to identify signal stemming from non-specific binding of proteins to the beads or antibody, rather than the specific DISC [15].
- Purpose: To distinguish specific caspase-8 activity isolated from the CD95-DISC from background activity caused by non-specifically bound proteins.
- Implementation: This control should be included for every experimental condition. It involves processing a sample (preferably from an unstimulated condition) through the IP procedure without the addition of the primary immunoprecipitating antibody (anti-CD95), or using an isotype control antibody [15]. The resulting caspase-8 activity and Western Blot signal from this control reflect non-specific interactions.
- Interpretation: A valid experiment will show significantly higher caspase-8 activity and clear enrichment of DISC components (caspase-8, FADD) in the specific IP sample compared to the Beads control. Similar signals in both indicate a failed IP and unreliable activity data.
Cell Viability Threshold at Seeding
Cell health at the experiment's outset is a fundamental prerequisite.
- Purpose: To ensure that the observed caspase-8 activation and subsequent apoptosis are specifically induced by the experimental stimulus (e.g., CD95L) and not by pre-existing stress or death in the cell population.
- Implementation: Before seeding cells for the main experiment, a small aliquot of the cell suspension must be mixed with Trypan Blue and counted using a hemocytometer or automated cell counter [15].
- Interpretation: The protocol mandates that experiments should proceed only if cell viability exceeds 93% [15]. A lower viability indicates suboptimal culture conditions, which can lead to aberrant baseline caspase activation and highly variable results.
Assay Specificity Verification
Specificity must be confirmed at multiple levels to attribute the measured activity unequivocally to caspase-8 within the DISC.
- IP Specificity (Western Blot): Confirming the successful and specific pull-down of the DISC is crucial. The Western Blot analysis of the IP sample must demonstrate the presence of caspase-8, FADD, and c-FLIP, and the absence of proteins not associated with the complex [15].
- Pharmacological Inhibition: Using well-characterized caspase-8 specific inhibitors (e.g., Z-IETD-FMK) in parallel experiments provides strong evidence for specificity. Effective inhibition of the measured activity confirms it is caspase-8-derived.
- Genetic Validation: Knockdown or knockout of caspase-8 (or its essential partner FADD) should abolish the activity signal, providing the most definitive proof of specificity.
Quantitative Data Presentation and Analysis
The quantitative data derived from the caspase-8 activity assay and supporting controls should be structured for clear interpretation. The table below provides a template for data organization.
Table 1: Example Structure for Caspase-8 Activity and Control Data
| Experimental Condition | Caspase-8 Activity (RFU/min) | Beads Control Activity (RFU/min) | Specific Activity (RFU/min) | Viability at Seeding (%) | DISC Recruitment (WB: Caspase-8) |
|---|---|---|---|---|---|
| Unstimulated | 150 ± 20 | 120 ± 15 | 30 ± 25 | 95.5 | - |
| CD95L (10 min) | 1250 ± 150 | 110 ± 20 | 1140 ± 152 | 96.0 | +++ |
| CD95L + Inhibitor | 200 ± 30 | 130 ± 10 | 70 ± 32 | 94.8 | + |
Data Analysis Guidelines:
- Specific Activity Calculation: Subtract the Beads control activity from the raw experimental activity for each replicate to obtain the specific caspase-8 activity attributable to the DISC.
- Statistical Testing: Perform appropriate statistical tests (e.g., t-tests, ANOVA) on the specific activity values, not the raw data, to compare experimental conditions.
- Correlation with Western Blot: Ensure that high specific activity correlates with strong caspase-8 and FADD recruitment visible in the Western Blot of the IP sample.
The Scientist's Toolkit: Essential Research Reagents
Successful execution of this protocol relies on high-quality, specific reagents. The following table details the essential materials required.
Table 2: Key Research Reagent Solutions for Caspase-8 Studies
| Reagent / Resource | Source / Catalog Number Example | Critical Function in the Protocol |
|---|---|---|
| Anti-CD95 Antibody | Santa Cruz, sc-8009 [15] | Key antibody for immunoprecipitating the DISC. |
| Anti-Caspase-8 Antibody | Clone C15 [15] | For detecting procaspase-8 and its cleaved forms in Western Blot. |
| Anti-FADD Antibody | Clone 1C4 [15] | Confirms successful DISC formation via Western Blot. |
| Recombinant CD95L | Fricker et al. [15] | The death ligand used to trigger extrinsic apoptosis and DISC assembly. |
| c-FLIP Inhibitor (FLIPinB) | N/A [43] | Small molecule that targets the caspase-8/c-FLIPL heterodimer, used to probe regulation and enhance DL-induced apoptosis [43]. |
| Caspase-8 Fluorogenic Substrate (IETD-based) | Commercially available (e.g., Ac-IETD-AFC) | The substrate hydrolyzed by active caspase-8, generating a measurable fluorescent signal. |
| Magnetic Beads (Protein A/G) | Various commercial suppliers | Solid support for antibody-mediated immunoprecipitation of the DISC. |
Troubleshooting Common Challenges
- Low Specific Activity: If the difference between experimental and beads control activity is small, optimize antibody concentration and washing stringency during IP. Verify CD95 receptor expression in your cell line.
- High Background in Beads Control: Pre-clear the lysate with beads alone before adding the primary antibody. Use a different lot or source of beads or antibody.
- No Activation Upon Stimulation: Titrate the concentration and duration of CD95L stimulation. Check cell line sensitivity to extrinsic apoptosis; some lines (Type II) require amplification through the mitochondrial pathway [13].
- Inconsistent Viability: Ensure consistent cell culture conditions, avoid over-confluency, and use fresh, pre-warmed medium for seeding.
Concluding Remarks
The precise measurement of caspase-8 activation is a cornerstone of extrinsic apoptosis research. The protocol detailed herein, with its embedded framework of critical controls, provides a reliable method for quantifying this key event in its native complex. Meticulous attention to the Beads control, cell viability threshold, and multi-layered specificity verification is what separates robust, publishable data from ambiguous results. This disciplined approach enables researchers to confidently probe the mechanisms of cell death, evaluate novel pharmacological inhibitors, and advance our understanding of cell death signaling in health and disease.
The extrinsic apoptosis pathway is a crucial mechanism of programmed cell death initiated by external stimuli, such as death receptor ligands (e.g., CD95L/FasL, TRAIL/Apo2L). A pivotal event in this pathway is the activation of caspase-8 at the multi-protein signaling platform known as the death-inducing signaling complex (DISC) [15] [44]. Upon ligand binding, death receptors recruit the adaptor protein FADD, which in turn recruits procaspase-8 via homotypic death effector domain (DED) interactions [44] [45]. This recruitment leads to the formation of DED filaments, where procaspase-8 undergoes dimerization, activation, and subsequent processing [45]. The activated caspase-8 then initiates a cascade of downstream events that ultimately lead to apoptotic cell death.
The classification of cells into Types I and II is based on their differential requirement for mitochondrial amplification in death receptor-mediated apoptosis [46] [47]. This classification has profound implications for experimental design, data interpretation, and the development of therapeutic agents targeting cell death pathways.
Type I vs. Type II Cells: Molecular Mechanisms and Characteristics
Defining Characteristics and Signaling Pathways
In Type I cells, robust caspase-8 activation at the DISC directly processes and activates effector caspases (such as caspase-3) without requiring mitochondrial amplification [46] [47]. This direct pathway is sufficient to execute the apoptotic program. In contrast, Type II cells generate less active caspase-8 at the DISC and require mitochondrial amplification to achieve full apoptosis commitment [46]. In these cells, caspase-8 cleaves the BH3-only protein Bid to generate truncated Bid (tBid), which translocates to mitochondria and activates Bax/Bak, leading to mitochondrial outer membrane permeabilization (MOMP), cytochrome c release, and activation of the caspase-9/-3 cascade [48] [47].
Table 1: Key Characteristics of Type I and Type II Cells
| Characteristic | Type I Cells | Type II Cells |
|---|---|---|
| Mitochondrial Dependency | Independent | Dependent |
| Caspase-8 Activation at DISC | High | Moderate/Low |
| Bid Cleavage and tBid Formation | Not essential | Essential |
| BAX/BAK Activation | Not required | Required |
| Cytochrome c Release | Minimal | Extensive |
| XIAP Sensitivity | Lower | Higher |
| BCL-2 Overexpression Effect | Minimal inhibition | Strong inhibition |
The reasons for these differential pathways are not fully understood but are influenced by the expression levels of key regulatory proteins. Cells with high levels of XIAP (X-linked inhibitor of apoptosis protein) often require mitochondrial amplification to overcome XIAP-mediated caspase inhibition, as mitochondria release pro-apoptotic proteins like Smac/DIABLO that neutralize XIAP [47]. The expression levels of caspase-8 and cellular FLICE-inhibitory proteins (c-FLIP) at the DISC also significantly influence this classification [49].
Experimentally Determined Cell Line Classifications
Research across different cell models has established specific classifications for common experimental cell lines:
Table 2: Experimentally Classified Type I and Type II Cell Lines
| Cell Line | Origin | Classification | Experimental Basis |
|---|---|---|---|
| Thymocytes | Mouse | Type I | Sensitive to Fas despite Bid deficiency [46] |
| SW480 | Human colon carcinoma | Type I | TRAIL-induced apoptosis not blocked by caspase-9 inhibitor [46] |
| SKW6.4 | Human lymphocytic | Type I | Established model for death receptor signaling [46] |
| Hepatocytes | Mouse/Human | Type II | Resistant to Fas in Bid-/- mice; require mitochondrial amplification [46] |
| HCT116 | Human colon carcinoma | Type II | TRAIL-induced apoptosis blocked by caspase-9 inhibitor [46] |
| Head & Neck Cancer Cells | Various | Mixed | Subset shows Type II with TRAIL sensitivity and high caspase-8/Bid [49] |
Practical Cell Culture Considerations: Adherent vs. Suspension
Experimental Workflow for Caspase-8 Activation Studies
The following diagram illustrates the generalized experimental workflow for measuring caspase-8 activation, incorporating considerations for different cell types:
Cell-Specific Protocol Adaptations
When studying caspase-8 activation, researchers must adapt their protocols based on whether they are using adherent or suspension cell lines:
For Adherent Cells (e.g., HeLa-CD95, HCT116, SW480):
- Seed 5-8×10^6 cells in 14.5 cm plates 24 hours before experimentation [15]
- Ensure cells are 80-90% confluent and firmly adherent at time of treatment
- For detachment during harvest, use trypsin (2 mL for a T175 flask, incubate 5 min at 37°C) followed by neutralization with 10 mL medium [15]
- Always include floating cells in analysis as they may represent apoptotic populations
For Suspension Cells (e.g., Jurkat, MV4-11, primary T-cells):
- Seed 1×10^7 cells per condition in 14.5 cm dishes immediately before treatment [15]
- For primary T-cells, use 1×10^8 cells per condition due to smaller cell size [44]
- No trypsinization required; proceed directly to centrifugation steps
- Cells can be used immediately without overnight incubation
The Scientist's Toolkit: Essential Research Reagents
Successful investigation of caspase-8 activation requires specific reagents tailored to the experimental approach:
Table 3: Essential Research Reagents for Caspase-8 Studies
| Reagent Category | Specific Examples | Application & Function |
|---|---|---|
| Death Receptor Ligands | Recombinant CD95L/FasL [15], TRAIL/Apo2L [49] | Induce DISC formation and initiate extrinsic apoptosis pathway |
| Caspase Inhibitors | z-VAD-fmk (pan-caspase) [49], Z-LEHD-FMK (caspase-9) [46] | Determine caspase dependency and pathway specificity |
| Antibodies for Immunoprecipitation | Anti-APO-1 [15], Anti-CD95 [44] | Isolate DISC complexes for composition analysis |
| Antibodies for Western Blot | Anti-caspase-8 (clone C15) [15], Anti-FADD (clone 1C4) [15], Anti-Bid [46], Anti-PARP [15] | Detect protein processing, cleavage, and complex composition |
| Cell Line Models | HeLa-CD95 (CD95-overexpressing) [15], HCT116 (colon carcinoma) [46], SW480 (colon carcinoma) [46] | Provide established systems for Type I/II comparative studies |
| Chemical Tools | FLIPin compounds [1], Smac mimetics [49] | Modulate specific pathway components for mechanistic studies |
Critical Experimental Parameters and Methodological Details
Death Receptor Stimulation Optimization
The efficacy of caspase-8 activation is highly dependent on proper death receptor stimulation. Key parameters to optimize include:
- Ligand Concentration: Titrate CD95L/FasL or TRAIL concentrations for each cell type [44]
- Stimulation Duration: Time courses from 15 minutes to several hours may be necessary to capture early activation events [15]
- Ligand Format: Isoleucine zipper-trimerized TRAIL (izTRAIL) enhances stability and efficacy [49]
- Inclusion of Cross-linking Agents: Enhances receptor clustering and DISC formation efficiency
DISC Immunoprecipitation and Analysis
The core protocol for analyzing native caspase-8 activation involves DISC immunoprecipitation:
- Cell Lysis: Use mild lysis buffers (1% CHAPS or Triton X-100) to preserve protein complexes [15]
- Immunoprecipitation: Incubate lysates with anti-receptor antibodies (e.g., anti-APO-1) coupled to protein A/G beads for 2-4 hours at 4°C [44]
- Wash Stringency: Vary salt concentration (150-500 mM NaCl) to reduce non-specific binding [15]
- Bead Controls: Always include "beads control" samples without receptor antibody to identify non-specific interactions [15]
Caspase-8 Activity Assessment
Multiple complementary approaches should be employed to fully characterize caspase-8 activation:
- In-situ Activity Assays: Fluorogenic substrates (IETD-based) directly measure enzymatic activity in immunoprecipitated complexes [15]
- Processing Analysis: Western blot detection of caspase-8 cleavage products (p55/53, p43/41, p30, p18) reveals activation status [44]
- Downstream Substrate Cleavage: Monitor PARP, caspase-3, and Bid cleavage to confirm functional signaling [46]
Signaling Pathway Architecture in Type I vs. Type II Cells
The molecular basis for differential caspase-8 signaling in Type I and Type II cells involves distinct protein complexes and signaling cascades:
The careful selection of appropriate cell models—considering both their growth characteristics (adherent vs. suspension) and apoptotic signaling classification (Type I vs. Type II)—is fundamental to successful research on caspase-8 activation in the extrinsic apoptosis pathway. The experimental protocols and considerations outlined here provide a framework for designing robust, reproducible studies that account for these critical cellular characteristics. As research in targeted cancer therapies continues to evolve, understanding these fundamental cell line considerations becomes increasingly important for both basic mechanistic studies and translational drug development efforts.
Caspase-8 is a crucial initiator caspase in the extrinsic apoptosis pathway, activated upon ligand binding to death receptors like CD95/Fas. This activation occurs within a multi-protein complex known as the Death-Inducing Signaling Complex (DISC), where procaspase-8 molecules aggregate via their death effector domains (DEDs) and undergo auto-proteolytic activation [15] [36]. The cellular FLICE-inhibitory protein (cFLIP) is a critical regulator of this process, existing in long (cFLIPL) and short (cFLIPS) splice variants. cFLIPL, a protease-deficient homolog of caspase-8, can hetero-dimerize with procaspase-8. Notably, cFLIPL exerts dual functions depending on its expression levels: at low physiological levels, it promotes procaspase-8 activation and apoptosis, while at high overexpression levels, it inhibits apoptosis [50] [36]. The synthetic peptide z-IETD-fmk serves as a potent and specific caspase-8 inhibitor, forming an irreversible covalent bond with the enzyme's active site cysteine residue, thereby blocking its proteolytic activity and subsequent apoptotic signaling [15].
Research Reagent Solutions
Table 1: Key Reagents for Caspase-8 and Extrinsic Apoptosis Research
| Reagent | Type/Form | Primary Function | Key Characteristics & Considerations |
|---|---|---|---|
| cFLIP (Endogenous Protein) | Long (cFLIPL) and Short (cFLIPS) isoforms | Dual regulator of caspase-8 activation at the DISC [50]. | cFLIPL: Promotes caspase-8 activation at low levels; inhibits at high levels [50].cFLIPS: Competitively inhibits procaspase-8 recruitment and activation [36]. |
| z-IETD-fmk | Cell-permeable synthetic peptide | Potent, irreversible caspase-8 inhibitor [15]. | Targets active site cysteine; used to confirm caspase-8-dependent phenotypes; requires validation of specificity. |
| Recombinant CD95L/FasL | Recombinant death ligand | Induces extrinsic apoptosis by triggering DISC formation [15]. | Used to stimulate the canonical caspase-8 activation pathway. Agonistic antibodies are an alternative. |
| FADD (Endogenous Protein) | Adaptor protein | Essential scaffold for DISC assembly; bridges death receptors and procaspase-8/cFLIP [51]. | Critical for initiating DED filament formation and caspase-8 activation. |
| HDAC Inhibitors (e.g., Vorinostat, Entinostat) | Small molecule inhibitors | Downregulate cFLIP expression, sensitizing cells to caspase-8-dependent apoptosis [29]. | Therapeutic agents that can overcome cFLIP-mediated apoptosis resistance. |
Table 2: Quantitative and Functional Effects of Caspase-8 Regulators
| Parameter / Experimental Context | Effect of cFLIP (Low Level) | Effect of cFLIP (High Level) | Effect of z-IETD-fmk |
|---|---|---|---|
| Caspase-8 Activation Kinetics | Potent enhancement (>50% processing at 2h vs. minimal in control) [50] | Inhibition of homo-dimerization and processing | Complete inhibition of enzymatic activity |
| Apoptosis Induction (e.g., in HeLa cells) | Promotion (dose-dependent enhancement, >30% cell death) [50] | Strong inhibition | Blockade of death receptor-mediated apoptosis |
| Downstream Substrate Cleavage (e.g., PARP, Caspase-3) | Increased cleavage indicative of apoptosis execution | Decreased cleavage | Abrogation of substrate cleavage |
| NSCLC Patient Prognosis | Not Applicable | High cytoplasmic expression correlates with shorter overall survival (HR=1.59) [29] | Not Applicable |
| Response to HDACi Treatment | Not Applicable | HDACi (e.g., Vorinostat) downregulates FLIP, inducing apoptosis; Resistance in FLIP-overexpressing cells [29] | Protects from HDACi-induced apoptosis |
Experimental Protocols
Protocol 1: Measuring Caspase-8 Activity at the Native DISC
This protocol enables the direct assessment of caspase-8 activation within the immunopurified DISC, providing a precise readout of the initial activation event [15].
I. Cell Culture and Apoptosis Induction
- Cell Lines: Use HeLa-CD95 or other adherent/suspension cells sensitive to CD95L-induced apoptosis.
- Preparation: Seed 5 x 10⁶ HeLa-CD95 cells per 14.5 cm plate in DMEM F12 medium with 10% FCS. Incubate overnight at 37°C with 5% CO₂. Use triplicate plates per condition.
- Stimulation: The next day, stimulate cells with 1 µg/mL recombinant CD95L for a defined period (e.g., 5-30 minutes) to trigger DISC assembly.
II. DISC Immunoprecipitation
- Lysis: Place plates on ice, discard medium, and wash cells with cold PBS. Lyse cells in 1 mL of mild lysis buffer (e.g., containing 1% Triton X-100, 30 mM Tris-HCl pH 7.5, 150 mM NaCl, 10% glycerol, and protease inhibitors) for 30 minutes at 4°C.
- Clarification: Scrape cells and transfer lysate to a microcentrifuge tube. Centrifuge at 15,000 × g for 15 minutes at 4°C to remove insoluble material.
- IP: Incubate the supernatant with 2 µg of anti-CD95 (e.g., mouse monoclonal anti-APO-1) antibody and 20 µL of Protein A/G Sepharose beads for 2-4 hours at 4°C with rotation.
- Washing: Pellet beads by brief centrifugation and wash 3-4 times with 1 mL of lysis buffer.
III. Caspase-8 Activity Assay
- Setup: Resuspend the immunoprecipitate (beads) from one plate in 50 µL of caspase assay buffer (e.g., 100 mM HEPES pH 7.5, 20% glycerol, 0.5 mM EDTA, 5 mM DTT).
- Reaction: Add a caspase-8-specific fluorogenic substrate (e.g., IETD-AFC) to a final concentration of 100 µM.
- Incubation: Incubate the reaction at 37°C for 1-2 hours protected from light.
- Measurement: Transfer the supernatant to a black 96-well plate and measure fluorescence (excitation 400 nm, emission 505 nm) using a plate reader. Include a beads-only control and a sample with the caspase-8 inhibitor z-IETD-fmk (e.g., 20 µM) to confirm signal specificity.
IV. Western Blot Analysis
- After the activity assay, elute proteins from the beads with Laemmli buffer by boiling for 5-10 minutes.
- Analyze samples by SDS-PAGE and western blotting to confirm immunoprecipitation efficiency and protein levels using antibodies against CD95, FADD, caspase-8, and cFLIP [15].
Protocol 2: Assessing the Efficacy of cFLIP-Targeting Drugs (e.g., HDAC Inhibitors)
This protocol evaluates how therapeutic agents that target cFLIP expression affect caspase-8 activation and cell death [29].
I. Cell Treatment and Viability Assessment
- Seeding: Seed NSCLC cell lines (e.g., H460, A549) and a normal lung fibroblast control (e.g., 34LU) in 96-well plates at a density of 5 x 10³ cells per well.
- Pre-treatment: The next day, pre-treat cells with a range of clinically achievable concentrations of an HDAC inhibitor like Vorinostat (e.g., 1-10 µM) for 6-24 hours. Include a DMSO vehicle control.
- Co-treatment (Optional): To test for synergy, co-treat cells with Vorinostat and recombinant TRAIL (e.g., 100 ng/mL) or a chemotherapeutic agent like cisplatin.
- Viability Assay: After 24-48 hours, measure cell viability using an MTT, MTS, or CellTiter-Glo assay according to the manufacturer's instructions.
II. Analysis of Apoptotic Markers by Western Blot
- Lysis: In parallel, lyse treated cells from a 6-well plate format in RIPA buffer.
- Blotting: Perform western blot analysis to detect:
- cFLIP Downregulation: Decrease in both cFLIPL and cFLIPS isoforms.
- Caspase-8 Activation: Cleavage/processing of procaspase-8 (p55/p53) to its active subunits (p43/p41, p18, p10).
- Apoptosis Execution: Cleavage of PARP-1 (from 116 kDa to 89 kDa) and caspase-3.
- Loading Control: Actin or GAPDH.
III. Pathway Dependency Validation
- Genetic Knockdown: Use siRNA or shRNA to knock down procaspase-8, FADD, or cFLIP in target cells. Repeat treatments to confirm that apoptosis induced by the HDAC inhibitor is dependent on caspase-8 and the extrinsic pathway.
- Pharmacological Inhibition: Pre-treat cells with z-IETD-fmk (20-50 µM) for 1 hour before adding Vorinostat. This should significantly reduce PARP cleavage and cell death, confirming caspase-8 dependency [29].
Signaling Pathway and Experimental Workflow Diagrams
Diagram 1: Caspase-8 regulation by cFLIP and inhibitors. The diagram shows how death receptor (DR) ligation leads to the assembly of the DISC via FADD. cFLIP hetero-dimerizes with procaspase-8, with the cellular concentration of cFLIP determining whether it promotes or inhibits caspase-8 activation. HDAC inhibitors target cFLIP for downregulation, while z-IETD-fmk directly inhibits active caspase-8.
Diagram 2: Workflow for assessing caspase-8 activation and drug efficacy. The protocol involves stimulating cells and treating them with compounds of interest, followed by DISC immunoprecipitation. The immunoprecipitate is then split for parallel analysis of caspase-8 enzymatic activity and protein processing/levels via western blot, with data integrated for a final assessment.
A fundamental challenge in extrinsic apoptosis research is the significant heterogeneity in the timing of cellular commitment to death, even within clonal populations exposed to identical death ligands. This variability complicates experimental reproducibility and the efficacy of chemotherapeutic agents designed to induce cell death. A critical source of this heterogeneity stems from the molecular events occurring at the death-inducing signaling complex (DISC), where the activation of caspase-8 serves as the crucial switch [15] [2]. This Application Note delineates strategies for measuring and analyzing caspase-8 activation dynamics, providing researchers with methodologies to dissect and address the origins of heterogeneous apoptotic commitment. By focusing on the core mechanisms of caspase-8 regulation and employing single-cell analytical approaches, scientists can better predict cellular responses and develop more effective therapeutic interventions.
Quantitative Analysis of Key Apoptotic Regulators
The following tables summarize core components and mathematical modeling insights that inform our understanding of heterogeneity in apoptotic signaling.
Table 1: Core Proteins Regulating Caspase-8 Activation and Heterogeneity at the DISC
| Protein/Component | Function in Apoptosis | Role in Generating Heterogeneity |
|---|---|---|
| Procaspase-8 | Inactive zymogen; undergoes activation at the DISC via DED filament formation [15]. | Stochastic fluctuations in expression and filament assembly kinetics create variable activation thresholds [52]. |
| c-FLIP isoforms (c-FLIPL, c-FLIPS) | Regulatory proteins structurally similar to caspase-8 but with limited activity; modulate caspase-8 activation [52] [8]. | Variable cellular concentrations and stoichiometric ratios with procaspase-8 can promote or inhibit death [52] [8]. |
| FADD | Adaptor protein that recruits procaspase-8 to the activated death receptor [15]. | Heterogeneous recruitment efficiency can lead to differences in DISC assembly completeness. |
| DED Filaments | Ordered structures of procaspase-8 DEDs that form at the DISC, essential for its activation [15]. | The kinetics of filament assembly and disassembly can introduce timing variability in caspase-8 activation. |
Table 2: Insights from Mathematical Models of Cell Fate Decisions
| Modeling Focus | Key Finding | Implication for Heterogeneity |
|---|---|---|
| BCL-2 Family Dynamics | Tristable regimes (survival, senescence, apoptosis) can emerge from cooperative interactions, with stochastic fluctuations driving fate commitment [53]. | Intrinsic noise in protein interactions can explain heterogeneous outcomes in genetically identical cells. |
| Caspase-8/ c-FLIP Regulation | Threshold mechanism where c-FLIP inhibits caspase-8 activation at low ligand concentrations [52]. | Cell-to-cell variation in inhibitor levels can create a bimodal population response (dying vs. surviving). |
| Single-Cell Cardiomyocyte Decisions | Individual cells commit early to either hypertrophy or apoptosis; initial cell size and morphology can predict the bias [54]. | Pre-existing cellular states, not just stochastic signaling, contribute to fate heterogeneity. |
Experimental Protocol: Measuring Caspase-8 Activity at the DISC
This protocol is adapted from König et al. (2025) to measure caspase-8 activity directly within its native complex, which is critical for understanding initial commitment heterogeneity [15] [2].
Background and Principle
The key step in extrinsic apoptosis is the activation of procaspase-8 at the DISC. Upon receptor activation, procaspase-8 is recruited and forms death effector domain (DED) filaments, which are crucial for its catalytic activity. This protocol uses immunoprecipitation (IP) to isolate the native DISC, followed by a caspase activity assay, allowing for the direct analysis of the activation platform and the evaluation of pharmacological inhibitors.
Materials and Reagents
- Cell Line: HeLa-CD95 (or other adherent/suspension cells sensitive to CD95L-induced apoptosis).
- Key Reagents:
- CD95L (soluble or membrane-bound) for apoptosis induction.
- Anti-CD95 antibody (e.g., mouse monoclonal anti-CD95) for immunoprecipitation.
- Protein G or A Sepharose beads.
- Caspase-8 assay buffer (containing HEPES, NaCl, CHAPS, DTT).
- Caspase-8 fluorogenic substrate (e.g., IETD-AFC).
- Lysis buffer (e.g., 1% Triton X-100, 150 mM NaCl, 20 mM HEPES pH 7.4, plus protease inhibitors).
- Western blot reagents and antibodies for caspase-8, FADD, c-FLIP, etc.
Step-by-Step Procedure
Cell Culture and Preparation (Timing: 2 days)
- Culture HeLa-CD95 cells in DMEM F12 medium supplemented with 10% FCS, penicillin/streptomycin, and puromycin.
- Seed 2 x 10^6 cells in a T175 flask and incubate for two days at 37°C with 5% CO2.
Cell Seeding and Stimulation (Timing: 1 day)
- Trypsinize, count, and seed 5 x 10^6 cells per 14.5 cm plate (in triplicate for conditions and controls). Incubate overnight.
- The next day, stimulate cells with CD95L for a predetermined time (e.g., 0-30 minutes) to trigger DISC formation.
DISC Immunoprecipitation (Timing: 3-4 hours)
- Place plates on ice and wash cells with cold PBS.
- Lyse cells in lysis buffer for 30 minutes with gentle agitation.
- Centrifuge lysates to remove nuclei and debris.
- Incubate the supernatant with anti-CD95 antibody-coupled beads for several hours or overnight at 4°C.
- Wash beads extensively with lysis buffer to remove non-specifically bound proteins.
Caspase-8 Activity Assay (Timing: 1-2 hours)
- Split the IP sample: one part for Western blot analysis, the other for the activity assay.
- Resuspend the beads for the activity assay in caspase-8 assay buffer containing the fluorogenic substrate.
- Incubate at 37°C for 1-2 hours, protecting from light.
- Measure the fluorescence (e.g., excitation 400 nm, emission 505 nm) in a plate reader. The rate of fluorescence increase is proportional to caspase-8 activity.
Western Blot Analysis (Timing: 1 day)
- Elute proteins from the other part of the IP beads and separate by SDS-PAGE.
- Transfer to a membrane and probe for DISC components (caspase-8, FADD, c-FLIP) to confirm IP efficiency and protein processing.
Data Interpretation and Heterogeneity Assessment
Compare the caspase-8 activity from the IP assay across multiple biological replicates. A high degree of variability in activity levels between replicates treated identically can be an indicator of underlying heterogeneity in DISC assembly and activation. This data should be correlated with Western blot analysis of procaspase-8 processing and c-FLIP recruitment. The effectiveness of caspase-8 inhibitors can be directly assessed by their ability to reduce the measured activity in this native complex.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Studying Caspase-8 Activation and Heterogeneity
| Reagent / Assay | Specific Function | Utility in Addressing Heterogeneity |
|---|---|---|
| Recombinant CD95L/FasL | Binds and activates CD95 receptor to initiate DISC formation. | Standardized, reproducible apoptosis induction is a prerequisite for studying variability. |
| DED-Filament Inhibitors (e.g., DEDid peptide) | Binds to procaspase-8 DED, blocking filament assembly and activation [15]. | Probe to test the contribution of filament kinetics to timing heterogeneity. |
| c-FLIP Expression Constructs/siRNA | Modulates cellular levels of caspase-8's key regulator. | Tool to manipulate the caspase-8 activation threshold and study its impact on population bimodality. |
| Caspase-8 Fluorogenic Substrates (IETD-) | Converted to a fluorescent product upon cleavage by active caspase-8. | Enables kinetic measurement of enzyme activity in purified DISC or live cells. |
| Annexin V Conjugates | Binds phosphatidylserine exposed on the outer leaflet of the plasma membrane in early apoptosis. | A flow cytometry-based endpoint assay to quantify the proportion of cells committed to death [55]. |
| TUNEL Assay Kit | Labels DNA strand breaks, a late-stage apoptotic marker. | Another endpoint assay to confirm apoptosis and its heterogeneity in a population [55]. |
| Flow Cytometry with Color Mapping | Visualizes a third parameter (e.g., caspase-8 activity) on a 2D plot using a color scale [56]. | Powerful tool for visualizing correlated expression of active caspase-8 and other proteins at the single-cell level. |
Advanced Strategy: Integrating Single-Cell Dynamics
To move beyond population averages, researchers should adopt single-cell approaches. As demonstrated in a study on cardiomyocytes, tracking individual cells revealed an early, biased commitment to either hypertrophy or apoptosis, supporting a "grow-or-die" model rather than a "grow-and-die" model [54]. Inhibition of caspase-3 shifted the single-cell probability from apoptosis towards the hypertrophy fate, highlighting how key nodes can be targeted to modulate heterogeneous fate decisions.
By combining the detailed molecular protocol for DISC analysis with single-cell fate tracking and computational modeling, researchers can develop a multi-scale understanding of apoptotic heterogeneity. This integrated approach is essential for identifying the key sources of variability in caspase-8 activation and developing strategies to overcome variable delays in apoptotic commitment for improved cancer therapy.
Validating Results and Placing Caspase-8 in a Broader Context
Within the broader research on the extrinsic apoptotic pathway, understanding the precise molecular link between initiator caspase activity and the pivotal point of cell death commitment is paramount. This application note details protocols for the correlative analysis of caspase-8 activity and its functional consequence, Mitochondrial Outer Membrane Permeabilization (MOMP). Caspase-8, the initiator caspase in the extrinsic pathway, plays a dual role: it can directly activate effector caspases and, crucially, can amplify the death signal by cleaving the BH3-only protein BID, leading to MOMP [5] [13]. MOMP is often considered the "point of no return" in apoptosis, as it results in the release of cytochrome c and other pro-apoptotic factors from the mitochondrial intermembrane space [57] [58]. This document provides detailed methodologies for quantitatively measuring caspase-8 activation and concurrently assessing MOMP, enabling researchers and drug development professionals to dissect this critical signaling axis.
Scientific Background
Caspase-8: More Than an Apoptotic Initiator
Caspase-8 is an aspartate-specific cysteine protease that serves as the apical enzyme in the extrinsic apoptotic pathway. Its activation occurs at the Death-Inducing Signaling Complex (DISC), formed upon ligation of death receptors like Fas/CD95 or TRAIL-R [59] [5]. Within the DISC, caspase-8 zymogens dimerize and undergo autoproteolytic cleavage, yielding the fully active enzyme [5]. Active caspase-8 then cleaves and activates downstream effector caspases, such as caspase-3 and -7. However, in many cell types (designated as Type II cells), this direct activation is insufficient for robust apoptosis and requires mitochondrial amplification [60] [13].
The critical link to the mitochondrial pathway is the caspase-8-mediated cleavage of BID, a BH3-only member of the Bcl-2 family. Cleavage generates truncated BID (tBID), which translocates to the mitochondria [5]. Recent research highlights the importance of a platform involving the mitochondrial lipid cardiolipin, which facilitates the full activation of caspase-8 and the subsequent cleavage of BID directly at the outer mitochondrial membrane [60] [13].
Mitochondrial Outer Membrane Permeabilization (MOMP): The Point of No Return
MOMP is the defining event of the mitochondrial apoptosis pathway. It is a tightly regulated process controlled by the Bcl-2 protein family, culminating in the formation of pores in the outer mitochondrial membrane by the pro-apoptotic effector proteins BAX and BAK [57] [58]. These pores allow proteins normally confined to the mitochondrial intermembrane space, such as cytochrome c and SMAC/DIABLO, to leak into the cytosol [57]. Cytochrome c, in combination with Apaf-1, forms the apoptosome, which activates caspase-9 and further amplifies the caspase cascade [57] [13]. The permeabilization of the mitochondrial outer membrane is a decisive step that typically commits the cell to death [58].
Table 1: Key Proteins Linking Caspase-8 to MOMP
| Protein | Function | Role in Caspase-8/MOMP Link |
|---|---|---|
| Caspase-8 | Initiator Protease | Cleaves and activates effector caspases; cleaves BID to tBID [5]. |
| BID | BH3-only Pro-apoptotic Protein | Substrate of caspase-8; once cleaved to tBID, activates BAX/BAK at MOM [5] [13]. |
| BAX/BAK | Multi-domain Pro-apoptotic Effectors | Oligomerize to form pores in the MOM, leading to MOMP and cytochrome c release [57] [58]. |
| Cardiolipin | Mitochondrial Phospholipid | Forms a platform at the MOM for caspase-8 activation and BID cleavage [60] [13]. |
| cFLIP | Regulatory Protein | Modulates caspase-8 activation at the DISC and MOM, can inhibit or promote activity depending on isoform and levels [59] [13]. |
| XIAP | Inhibitor of Apoptosis Protein | Binds and inhibits caspases-3, -7, and -9; its inhibition by SMAC/DIABLO (released during MOMP) is crucial for apoptosis in Type II cells [13]. |
The following diagram illustrates the core signaling pathway from caspase-8 activation to MOMP, integrating the key components described above.
Experimental Protocols
Protocol 1: Flow Cytometric Analysis of Caspase-8 Activity Using a FRET-Based Probe
This protocol enables real-time monitoring of caspase-8 activity in living cells, providing a quantitative readout of extrinsic pathway initiation.
Principle
A molecular probe is constructed from Cyan Fluorescent Protein (CFP) and Yellow Fluorescent Protein (YFP) linked by a peptide sequence containing the caspase-8 cleavage site (LEVD). When the probe is intact, FRET occurs from CFP to YFP upon excitation. Cleavage by caspase-8 separates the two fluorophores, abolishing FRET. The change in the FRET ratio can be precisely quantified by flow cytometry [61].
Reagents and Equipment
- FRET Probe: Plasmid encoding CFP-LEVD-YFP [61].
- Positive Control Inducer: Recombinant FasL, TRAIL, or caspase-8-RFP fusion protein expression vector [61].
- Caspase Inhibitor: pan-caspase inhibitor (e.g., z-VAD-fmk) or specific caspase-8 inhibitor.
- Cell Culture Reagents: Appropriate cell line (e.g., HeLa, Jurkat), culture medium, transfection reagent.
- Equipment: Flow cytometer capable of exciting CFP (~405-440 nm) and detecting emission at ~475 nm (CFP) and ~530 nm (YFP). Cell culture incubator.
Procedure
- Cell Seeding and Transfection: Seed cells in a 6-well plate at 60-80% confluency. The following day, transfect with the CFP-LEVD-YFP FRET probe plasmid using a standard transfection protocol. Include untransfected cells as a negative control for autofluorescence.
- Induction and Inhibition (24-48h post-transfection):
- Untreated Control: Leave one group of transfected cells untreated.
- Induced Group: Treat cells with the apoptotic inducer (e.g., 100 ng/mL FasL for 4-16 hours).
- Inhibited Control: Pre-treat cells with 20 µM z-VAD-fmk for 1 hour before adding the apoptotic inducer.
- Sample Preparation and Analysis:
- Harvest cells gently using non-enzymatic cell dissociation buffer.
- Wash cells once with ice-cold PBS and resuspend in PBS containing 2% FBS.
- Analyze cells immediately on the flow cytometer.
- Excite samples at 405 nm and collect fluorescence emission using 450/50 nm (CFP channel) and 535/30 nm (YFP channel) filters.
- Data Analysis:
- Gate on live, single cells that are positive for CFP fluorescence.
- For the gated population, calculate the mean fluorescence intensity (MFI) for both CFP and YFP channels.
- Compute the FRET ratio (YFP MFI / CFP MFI) for each sample.
- A decrease in the FRET ratio in induced samples compared to untreated controls indicates caspase-8 activation. This decrease should be suppressed in the inhibitor-treated group.
Protocol 2: Concurrent Measurement of MOMP via Cytochrome c Release and Mitochondrial Membrane Potential (ΔΨm)
This protocol allows for the correlative assessment of MOMP following the measurement of caspase-8 activity, typically performed in parallel or sequentially on the same cell population.
Principle
MOMP is assessed using two complementary methods:
- Immunofluorescence for Cytochrome c Localization: Cells are stained for cytochrome c and a mitochondrial marker (e.g., TOM20). In healthy cells, cytochrome c staining shows a punctate, mitochondrial pattern. Upon MOMP, cytochrome c is released into the cytosol, resulting in a diffuse staining pattern [57] [13].
- Mitochondrial Membrane Potential (ΔΨm) Measurement: The lipophilic cationic dye Tetramethylrhodamine Ethyl Ester (TMRE) accumulates in the mitochondrial matrix in a ΔΨm-dependent manner. A collapse in ΔΨm, which occurs rapidly after MOMP when caspases are active, leads to a loss of TMRE fluorescence [62].
Reagents and Equipment
- Antibodies: Anti-cytochrome c antibody, anti-TOM20 antibody, and suitable fluorescently-labeled secondary antibodies.
- ΔΨm Dye: TMRE (Tetramethylrhodamine Ethyl Ester).
- Permeabilization Buffer: PBS containing 0.1% Triton X-100.
- Fixative: 4% paraformaldehyde (PFA) in PBS.
- Equipment: Confocal or high-content fluorescence microscope. Microplate reader (for plate-based TMRE assays).
Procedure
Part A: Immunofluorescence Staining for Cytochrome c Release
- Cell Culture and Treatment: Seed cells on glass-bottom culture dishes or chamber slides. Treat cells according to the experimental design (e.g., untreated, induced, inhibited).
- Fixation and Permeabilization: At the desired timepoint post-induction, wash cells with PBS and fix with 4% PFA for 15 minutes at room temperature. Wash again and permeabilize with 0.1% Triton X-100 for 10 minutes.
- Immunostaining: Block cells with 5% BSA in PBS for 1 hour. Incubate with primary antibodies (anti-cytochrome c and anti-TOM20) diluted in blocking buffer overnight at 4°C. Wash thoroughly and incubate with fluorescent secondary antibodies for 1 hour at room temperature in the dark.
- Imaging and Analysis: Acquire high-resolution images using a confocal microscope. Score cells for cytochrome c localization: punctate (healthy, mitochondrial) vs. diffuse (MOMP has occurred). Count at least 200 cells per condition.
Part B: TMRE Staining for ΔΨm Loss
- Staining: Following treatment, load cells with 50-100 nM TMRE in culture medium for 30 minutes at 37°C.
- Analysis (Microscopy): For a direct correlation with cytochrome c staining, include TMRE in the final wash before fixation for the immunofluorescence protocol (Part A). Image TMRE fluorescence. A loss of bright, punctate TMRE staining indicates ΔΨm collapse.
- Analysis (Flow Cytometry): As a quantitative alternative, harvest TMRE-stained cells and analyze immediately by flow cytometry (Ex/Em: ~549/575 nm). A leftward shift in the TMRE fluorescence histogram indicates a loss of ΔΨm.
Table 2: Key Reagent Solutions for Correlative Analysis
| Research Reagent / Material | Function / Application | Example Product / Target |
|---|---|---|
| Recombinant Death Ligands | Activate the extrinsic pathway by engaging death receptors. | FasL, TRAIL |
| FRET-Based Caspase Probe | Real-time monitoring of caspase-8 activity in live cells. | CFP-LEVD-YFP construct [61] |
| Caspase Inhibitors | Pharmacological validation of caspase-specific effects. | z-VAD-fmk (pan-caspase), z-IETD-fmk (caspase-8) |
| Fluorescent ΔΨm Indicators | Measure mitochondrial membrane potential health/collapse. | TMRE, JC-1 |
| Antibody Panel (ICC/IF) | Detect subcellular localization of key proteins. | Anti-cytochrome c, Anti-BID/tBID, Anti-BAX, Mitochondrial markers (e.g., TOM20) |
| BID Knockout/Knockdown Tools | Genetically validate BID's essential role in the pathway. | siRNA, shRNA, CRISPR/Cas9 constructs |
| cFLIP Modulators | Investigate regulation of caspase-8 activation at DISC/MOM. | cFLIP expression vectors [59] [13] |
Data Interpretation and Correlative Analysis
Successful correlative analysis requires integrating data from both caspase-8 and MOMP assays. The workflow below outlines the logical sequence of experiments and decision points for interpreting the connection between caspase-8 and MOMP.
Expected Outcomes and Interpretation:
- In Type I Cells: A significant activation of caspase-8 may be sufficient to induce cell death with minimal MOMP, as effector caspases are directly activated [13].
- In Type II Cells: Activation of caspase-8 should be followed by a significant loss of ΔΨm and cytochrome c release. This indicates the pathway relies on mitochondrial amplification [13]. This outcome can be genetically validated by demonstrating that BID knockout/knockdown abrogates MOMP without affecting initial caspase-8 activation.
- Inhibited Pathway: If caspase-8 is not activated, check death receptor expression and DISC formation. If caspase-8 is activated but MOMP does not occur, investigate potential blocks in BID expression/cleavage, high levels of anti-apoptotic Bcl-2 proteins, or defective BAX/BAK activation.
Technical Notes and Troubleshooting
- Cell Type Considerations: The choice of cell line is critical. Preliminary experiments should determine whether the cells of interest behave as Type I or Type II in response to death receptor stimulation [13].
- Time Course: Perform a time-course experiment to establish the kinetics of caspase-8 activation relative to MOMP. Caspase-8 activation typically precedes detectable cytochrome c release.
- FRET Probe Specificity: While the LEVD sequence is sensitive to caspase-8, it can also be cleaved by other caspases (e.g., caspase-6). Use specific inhibitors and caspase-8 deficient cells to confirm the source of activity [61].
- TMRE Artifacts: Ensure TMRE concentrations are optimized, as high concentrations can artifactually depolarize mitochondria. Use a positive control (e.g., FCCP, an uncoupler) to validate the assay.
- Alternative MOMP Assays: SMAC/DIABLO release can be measured alongside cytochrome c for additional confirmation. The oligomerization status of BAX/BAK can be analyzed by cross-linking and western blotting [57].
Caspase-8 (CASP8) is a cysteine protease that functions as the critical initiator caspase in the extrinsic apoptotic pathway, which is triggered by the stimulation of cell surface death receptors (DRs) such as CD95/Fas/APO-1 and TRAIL receptors [63] [64]. Upon ligand binding, these receptors undergo oligomerization and form a multi-protein signaling platform known as the Death-Inducing Signaling Complex (DISC) [65] [64]. The DISC serves as the molecular machinery where the inactive zymogen procaspase-8 is recruited and undergoes activation through a process involving dimerization and subsequent interdomain cleavage [65] [64]. This activation is tightly regulated by cellular FLICE-inhibitory protein (c-FLIP), particularly its long isoform c-FLIPL, which can either promote or inhibit caspase-8 activation depending on its concentration and the cellular context [65]. Once activated, caspase-8 initiates a proteolytic cascade that leads to the activation of downstream effector caspases (e.g., caspase-3) and the execution of apoptosis [66] [64]. Mathematical modeling of these processes provides a powerful framework for making quantitative predictions about the kinetics of caspase-8 activation and subsequent cell fate decisions, enabling deeper insights into the dynamics of apoptotic signaling in both healthy and diseased states.
Mathematical Frameworks for Modeling Caspase-8 Kinetics
Deterministic and Stochastic Modeling Approaches
The dynamics of caspase-8 activation can be quantitatively described using different mathematical formalisms, each offering unique insights. Ordinary Differential Equations (ODEs) are particularly suited for modeling the temporal dynamics of signaling molecules in large populations where stochastic effects are minimal. ODE models assume well-mixed cellular environments and employ mass-action kinetics to describe biochemical reactions [64]. For instance, a reaction where A and B form complex C ((A+B \rightleftharpoons C)) can be translated into a system of coupled ODEs: (dA/dt = dB/dt = -k1 \cdot A \cdot B + k2 \cdot C) (dC/dt = k1 \cdot A \cdot B - k2 \cdot C) where (k1) and (k2) are kinetic rate constants. This approach has been successfully used to model the cascade of caspase-8 activation at the DISC and its downstream effects [65] [64].
In contrast, semi-stochastic models combine the strengths of both stochastic and deterministic frameworks. These are particularly valuable for understanding heterogeneous cell responses at low death receptor stimulation. One such model employs a direct Gillespie stochastic simulation algorithm to capture the slow, noise-prone assembly of the DISC/RIPoptosome platform, while using deterministic ODEs to model the downstream activation of caspases [67]. This hybrid approach accurately explains phenomena such as delayed cell death and fractional killing, where only a subset of cells undergoes apoptosis despite uniform death receptor stimulation [67].
Key Parameters and Variables in Caspase-8 Models
Mathematical models of caspase-8 activation incorporate specific, measurable biological parameters to generate quantitative predictions. Critical parameters include the absolute protein abundances of key signaling components. For example, in HeLa cells, quantitative studies have determined that DR4 and DR5 receptors are present on the cell surface at approximately 769 and 926 monomeric receptors per cell, respectively [67]. The table below summarizes key quantitative parameters used in these models.
Table 1: Key Quantitative Parameters for Caspase-8 Kinetic Models
| Parameter Description | Value/Range | Cellular Context | Reference |
|---|---|---|---|
| TNFR1 Surface Abundance | ~905 molecules/cell | HeLa cells | [67] |
| DR4 Surface Abundance | ~769 molecules/cell | HeLa cells | [67] |
| DR5 Surface Abundance | ~926 molecules/cell | HeLa cells | [67] |
| Minimal Active CASP8 | <1% of total CASP8 | HeLa cells | [63] |
| RIP1 Threshold for Apoptosis | <1,000 molecules/cell | L929 cells | [68] |
| RIP1 Threshold for Necroptosis | >46,000 molecules/cell | L929 cells | [68] |
These parameters are integrated into mathematical models that consist of multiple differential equations describing the dynamics of caspase-8 activation. For instance, a model focusing on the effects of the small molecule FLIPin on caspase-8 activation at the DISC is described by a system of 12 differential equations and incorporates 8 key kinetic parameters [65].
Experimental Validation of Model Predictions
Single-Cell Biosensors for Monitoring Caspase Activation
The validation of mathematical models predicting caspase-8 kinetics relies heavily on advanced experimental methodologies that enable real-time, quantitative monitoring of caspase activity in live cells. FRET-based biosensors are particularly powerful tools for this purpose. A sophisticated dual-FRET system, utilizing a triple fusion fluorescent protein (seCFP-Venus-mRFP1) with specific caspase cleavage sequences (IETD for caspase-8 and DEVD for caspase-3), allows for the simultaneous monitoring of both initiator and effector caspase activities within single living cells [66]. This system detects distinct activation patterns of caspase-8 and caspase-3 in response to various apoptotic stimuli, enabling researchers to correlate model predictions with actual temporal dynamics in live cells [66]. Furthermore, using a plasma membrane-targeted FRET-based biosensor, researchers have captured the spatiotemporal dynamics of caspase-8 activation, demonstrating that focal activation is sufficient to propagate apoptotic signals through death receptors via a diffusion-mediated process [66].
Quantitative Validation of Model Predictions
Experimental validation using these biosensors and other quantitative techniques has confirmed several key predictions generated by mathematical models. A central finding is the remarkable efficiency of the caspase-8 activation cascade. Quantitative experimental data validated a mathematical model showing that less than 1% of the total cellular pool of caspase-8 proteins is sufficient to initiate the apoptotic program once activated [63]. This high sensitivity underscores the amplification potential of the caspase cascade. Furthermore, combined modeling and experimental approaches have elucidated how the stochastic assembly of the DISC/RIPoptosome platform introduces variability in caspase-8 activation times, which in turn drives heterogeneous cell death responses at the population level, particularly under low levels of death receptor stimulation [67]. This stochasticity is a crucial factor in fractional killing, a phenomenon where only a fraction of cells undergoes apoptosis even at death-inducing ligand concentrations [67].
Table 2: Key Experimental Reagents for Studying Caspase-8 Kinetics
| Research Tool | Type/Composition | Primary Function in Research | Key Feature |
|---|---|---|---|
| Dual-FRET Biosensor (CYR83) | Triple fusion protein: seCFP-Venus-mRFP1 with IETD & DEVD linkers | Simultaneous monitoring of caspase-8 and caspase-3 activation in live cells | Enables differential activation profiling in single cells [66] |
| FLIPinBγ | Small molecule chemical probe | Binds caspase-8/c-FLIPL heterodimer to enhance caspase-8 activity after initial processing | Used to test model predictions on modulating DED filament function [65] |
| SWATH-MS | Mass spectrometry-based proteomics | Absolute quantification of proteins in dynamic signaling complexes (e.g., TNFR1, RIP1 complexes) | Provides sensitive, reproducible inputs for mathematical modeling [68] |
| QuantiBRITE PE Assay | Flow cytometry with phycoerythrin beads | Quantitative estimation of death receptor abundance on single cell surfaces | Enabled precise measurement of TNFR1, DR4, DR5 levels [67] |
Application Notes: Protocol for Kinetic Analysis of Caspase-8 Activation
Protocol: Integrating Mathematical Modeling with Experimental Validation
This protocol outlines a workflow for generating and experimentally testing quantitative predictions of caspase-8 activation kinetics using a combination of modeling, live-cell imaging, and biochemical analysis.
Step 1: System Definition and Model Selection
- Define the scope of the biological system (e.g., DISC-mediated initiation only, or inclusion of downstream effector caspases and mitochondrial amplification).
- Select the appropriate mathematical framework. For population-level average behavior, use an ODE-based model. For investigating heterogeneous single-cell responses, particularly at low stimulus levels, employ a semi-stochastic model [67] [64].
- Populate the model with initial kinetic parameters and protein concentrations from established literature or prior experiments (refer to Table 1 for examples).
Step 2: Model Simulation and Prediction
- Use computational software (e.g., Python with SciPy, MATLAB) to numerically solve the system of equations.
- Run simulations under various conditions, such as different death receptor ligand concentrations or genetic perturbations (e.g., c-FLIPL knockdown).
- Key outputs to analyze include: the time-to-activation of caspase-8 and caspase-3, the threshold of receptor stimulation required for commitment to cell death, and the predicted fraction of cells undergoing apoptosis in a population.
Step 3: Experimental Setup and Live-Cell Imaging
- Cell Preparation: Plate cells expressing the dual-FRET biosensor CYR83 [66] in an imaging-compatible chamber.
- Stimulation and Imaging: Apply the death receptor ligand (e.g., TRAIL, anti-FAS antibody) at the concentrations used in the simulations. Immediately begin time-lapse imaging using a fluorescence microscope equipped with filters appropriate for CFP, YFP (Venus), and RFP.
- Data Acquisition: Collect FRET ratio images over time. The seCFP/Venus ratio (excited at 440 nm) primarily reports caspase-8 activity (cleavage at IETD), while the Venus/mRFP1 ratio (excited at 500 nm) primarily reports caspase-3 activity (cleavage at DEVD) [66].
Step 4: Data Analysis and Model Validation
- Image Analysis: Process time-lapse images to quantify FRET ratios (e.g., 528 nm/476 nm for caspase-8 activity) for individual cells over time.
- Kinetic Profiling: Plot the FRET ratio versus time to generate activation curves for caspase-8 and caspase-3. Calculate metrics like delay time and activation rate.
- Model Refinement: Compare the experimental single-cell and population data with the model predictions. If discrepancies exist, refine the model parameters or structure (e.g., add regulatory feedback loops) and iterate the process.
Diagram: Integrated Workflow for Modeling and Validating Caspase-8 Kinetics
The following diagram illustrates the cyclic workflow of mathematical modeling and experimental validation, which is central to systems biology.
Discussion: Implications for Therapeutic Intervention
Quantitative mathematical models of caspase-8 activation have significant translational potential, particularly in the development of novel anti-cancer therapies. These models provide a powerful platform for in silico testing of therapeutic strategies aimed at manipulating the extrinsic apoptotic pathway to selectively induce cancer cell death. For instance, models that incorporate the detailed stoichiometry and composition of the DED filament have been used to predict the optimal cellular landscape (e.g., specific c-FLIPL to procaspase-8 ratios) that would favor the action of small molecule enhancers of caspase-8 activity, such as FLIPin [65]. This allows for the rational design of combination treatments that could sensitize resistant tumors to death receptor-targeted therapies.
Furthermore, understanding the crosstalk between apoptosis and necroptosis, another form of programmed cell death, is crucial. Models that quantify the recruitment of caspase-8 and RIP3 to the necrosome have revealed how the cellular levels of RIP1 dictate cell fate, with low levels promoting apoptosis and very high levels suppressing apoptosis to allow necroptosis [68]. This quantitative understanding is vital, as evidenced by in vivo studies showing that endothelial-specific deletion of caspase-8 in adult mice leads to fatal MLKL-dependent necroptotic hemorrhage in the small intestine, which can be prevented by TNF neutralization or MLKL deletion [69]. Such insights underscore the importance of caspase-8 in maintaining tissue homeostasis and highlight potential therapeutic targets for controlling pathological cell death processes.
Caspase-8, the initiator caspase of the extrinsic apoptotic pathway, has traditionally been studied within the confined context of apoptosis. However, emerging research has illuminated its pivotal role as a central molecular switch that governs crosstalk among multiple programmed cell death (PCD) pathways, including necroptosis, pyroptosis, and the recently characterized integrated cell death pathway known as PANoptosis [36]. This complex interaction network positions caspase-8 as a critical regulatory node in cell fate decisions, with significant implications for host defense, inflammatory diseases, and therapeutic development [70] [36].
PANoptosis represents an innovative conceptual framework in cell death biology, describing an intricately coordinated, inflammatory PCD pathway that integrates components and features from pyroptosis, apoptosis, and necroptosis without being fully explained by any one pathway alone [71] [72]. This pathway is orchestrated by multifaceted cytoplasmic complexes termed PANoptosomes, which serve as molecular platforms for simultaneously activating these three distinct cell death modalities [72] [73]. Within this integrated cell death paradigm, caspase-8 exhibits functional versatility that extends beyond its canonical apoptotic functions to include regulation of inflammatory responses and mediation of critical interactions among parallel PCD pathways [8] [74].
This protocol article provides detailed methodologies for investigating caspase-8 activation and its cross-pathway interactions within the emerging context of PANoptosis, offering researchers comprehensive tools to advance this rapidly evolving field.
Molecular Mechanisms of Caspase-8 in Cell Death Crosstalk
Caspase-8 in Apoptosis Initiation
Caspase-8 serves as the initiator caspase in the extrinsic apoptosis pathway. Upon activation by death receptors (e.g., FAS, TNFR1), it forms the death-inducing signaling complex (DISC) where it undergoes autocatalytic activation [36]. The active caspase-8 homodimer then cleaves and activates effector caspases-3 and -7, executing the apoptotic program [70] [36]. Additionally, caspase-8 can cleave the BH3-only protein BID to generate truncated tBID, which translocates to mitochondria and amplifies the death signal through the intrinsic apoptotic pathway [13] [75].
Table 1: Caspase-8 Functions in Cell Death Pathways
| Function | Mechanism | Pathway | Outcome |
|---|---|---|---|
| Initiator Caspase | DISC formation and self-activation | Extrinsic Apoptosis | Caspase-3/7 activation |
| Mitochondrial Amplification | Cleavage of BID to tBID | Intrinsic Apoptosis | Cytochrome c release |
| Inflammatory Regulation | Cleavage of IL-1β and GSDMD | Pyroptosis | Inflammation modulation |
| Necroptosis Checkpoint | RIPK1 and RIPK3 regulation | Necroptosis | MLKL activation suppression |
| PANoptosis Integration | PANoptosome assembly | PANoptosis | Coordinated cell death activation |
Caspase-8 in PANoptosis Regulation
The concept of PANoptosis has emerged as a unified cell death pathway that integrates components from pyroptosis, apoptosis, and necroptosis [72] [73]. PANoptosis is characterized by the assembly of PANoptosomes—supramolecular complexes that serve as cellular platforms for simultaneously activating multiple cell death pathways [71] [72]. These complexes incorporate molecular sensors (e.g., ZBP1, AIM2), adaptors (ASC, FADD), and catalytic effectors (RIPK1, RIPK3, caspase-8) that together coordinate the PANoptotic response [72].
Caspase-8 occupies a central position within several PANoptosome complexes, including ZBP1-, AIM2-, and RIPK1-PANoptosomes [72] [74]. In the ZBP1-PANoptosome, which forms in response to viral infection, caspase-8 interacts with ZBP1, RIPK3, RIPK1, FADD, and ASC to simultaneously drive apoptosis, pyroptosis, and necroptosis [71] [72]. Similarly, in the AIM2-PANoptosome, which activates in response to cytosolic DNA, caspase-8 collaborates with AIM2, ASC, and RIPK3 to orchestrate inflammatory cell death [72].
Table 2: Major PANoptosome Complexes and Their Components
| PANoptosome Type | Primary Activator | Key Components | Caspase-8 Role |
|---|---|---|---|
| ZBP1-PANoptosome | Viral RNA/DNA | ZBP1, RIPK1, RIPK3, FADD, ASC, NLRP3 | Apoptosis initiation, Complex assembly |
| AIM2-PANoptosome | Cytosolic DNA | AIM2, ZBP1, ASC, FADD, RIPK1, RIPK3 | Inflammatory regulation, Cell death execution |
| RIPK1-PANoptosome | TNF signaling | RIPK1, RIPK3, ASC, caspase-1, caspase-8 | Death receptor pathway integration |
| NLRP12-PANoptosome | Bacterial infection | NLRP12, ASC, caspase-8, RIPK3 | Innate immune response coordination |
Experimental Protocols
Detecting Caspase-8 Activation in PANoptosis
Principle: This protocol enables quantitative assessment of caspase-8 activation during PANoptosis induction by monitoring cleavage of specific substrates and formation of caspase-8-containing complexes.
Reagents:
- PANoptosis inducers: TNF-α (20-100 ng/mL), Poly(I:C) (1-5 μg/mL), nigericin (5-20 μM)
- Lysis buffer: RIPA buffer supplemented with protease and phosphatase inhibitors
- Caspase-8 activity assay kit (fluorometric)
- Antibodies: anti-caspase-8 (proform and cleaved), anti-RIPK1, anti-RIPK3, anti-ASC
- Co-immunoprecipitation kit
- Western blotting reagents
Procedure:
Cell Stimulation:
- Seed appropriate cells (e.g., THP-1, bone marrow-derived macrophages) in 6-well plates at 5×10^5 cells/well
- Stimulate cells with PANoptosis inducers for predetermined timepoints (typically 2-24 hours)
- Include controls: untreated cells, caspase-8 inhibitor (Z-IETD-FMK, 20 μM) treated cells
Caspase-8 Activity Measurement:
- Harvest cells and lyse in caspase assay buffer
- Incubate lysates with caspase-8 substrate IETD-AFC (50 μM) at 37°C for 1 hour
- Measure fluorescence (excitation 400 nm, emission 505 nm) using a plate reader
- Normalize activity to protein concentration
Caspase-8 Cleavage Analysis:
- Prepare whole cell lysates using RIPA buffer
- Separate proteins by SDS-PAGE (12% gel)
- Transfer to PVDF membrane and block with 5% BSA
- Incubate with anti-caspase-8 antibody (1:1000) overnight at 4°C
- Detect pro-caspase-8 (55/57 kDa) and cleaved fragments (43/41 kDa, 18 kDa) using HRP-conjugated secondary antibodies
PANoptosome Complex Immunoprecipitation:
- Lyse cells in mild lysis buffer (1% NP-40, 150 mM NaCl, 50 mM Tris pH 8.0)
- Pre-clear lysates with protein A/G beads for 30 minutes at 4°C
- Incubate with anti-ASC or anti-RIPK1 antibody overnight at 4°C
- Add protein A/G beads and incubate for 2 hours
- Wash beads 3 times with lysis buffer
- Elute proteins with 2× Laemmli buffer and analyze by western blotting for caspase-8, RIPK1, RIPK3, and ASC
Troubleshooting:
- High background in activity assays: Include appropriate controls and optimize cell lysis conditions
- Weak co-IP signals: Try crosslinking with DSS (1-2 mM) before lysis to stabilize transient interactions
- Non-specific bands in western blot: Validate antibodies using caspase-8 knockout cells
Functional Assessment of PANoptosis
Principle: This protocol provides a comprehensive approach to quantify simultaneous activation of apoptosis, pyroptosis, and necroptosis during PANoptosis.
Reagents:
- Propidium iodide (PI) and Hoechst 33342
- Annexin V-FITC apoptosis detection kit
- Anti-cleaved caspase-3 antibody
- Anti-GSDMD-NT antibody (pyroptosis marker)
- Anti-pMLKL antibody (necroptosis marker)
- LDH release assay kit
- IL-1β ELISA kit
Procedure:
Multiparametric Cell Death Analysis by Flow Cytometry:
- Harvest stimulated cells and wash with PBS
- Stain with Annexin V-FITC (apoptosis), PI (necrosis), and Hoechst 33342 (viability) according to manufacturer's protocols
- Analyze by flow cytometry with minimum 10,000 events per sample
- Identify distinct populations: viable (Annexin V-/PI-), early apoptotic (Annexin V+/PI-), late apoptotic (Annexin V+/PI+), and necroptotic/pyroptotic (Annexin V-/PI+)
PANoptosis Marker Analysis by Western Blotting:
- Prepare cell lysates as described in protocol 3.1
- Analyze parallel blots for:
- Apoptosis: cleaved caspase-3 (17/19 kDa) and PARP cleavage (89 kDa)
- Pyroptosis: GSDMD-NT (30 kDa) and caspase-1 p20
- Necroptosis: phosphorylated MLKL and RIPK3
Inflammatory Cytokine Measurement:
- Collect cell culture supernatants by centrifugation
- Analyze IL-1β, IL-18, and TNF-α release using commercial ELISA kits
- Measure LDH release as indicator of plasma membrane rupture
Morphological Assessment by Microscopy:
- Culture cells on glass coverslips and stimulate for PANoptosis
- Fix with 4% PFA and stain with specific antibodies for cleaved caspase-3, GSDMD-NT, and pMLKL
- Counterstain with DAPI and image using confocal microscopy
- Quantify cells positive for multiple cell death markers
Data Interpretation:
- PANoptosis is indicated by simultaneous activation of all three pathways in the same cell population
- Pathway-specific inhibitors can validate contributions of individual death mechanisms
- Temporal analysis may reveal sequential activation patterns
Signaling Pathway Visualization
Caspase-8 Regulatory Network in PANoptosis: This diagram illustrates the central role of caspase-8 in integrating signals from multiple PANoptosome complexes to coordinate apoptosis, pyroptosis, and necroptosis activation.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Caspase-8 and PANoptosis Studies
| Reagent Category | Specific Examples | Function/Application | Considerations |
|---|---|---|---|
| Caspase-8 Inhibitors | Z-IETD-FMK, Emricasan | Specific inhibition of caspase-8 activity | Emricasan is a broad-spectrum caspase inhibitor; Z-IETD-FMK is more specific |
| PANoptosis Inducers | TNF-α + SMAC mimetic + z-VAD, Influenza A virus, SARS-CoV-2 | Activate PANoptosis via different sensors | Concentration and timing require optimization for each cell type |
| Antibodies for Detection | Anti-caspase-8 (cleaved forms), anti-GSDMD-NT, anti-pMLKL, anti-RIPK1 | Detect activation of specific cell death pathways | Validate specificity using knockout controls |
| Cell Death Assays | Annexin V/PI staining, LDH release, IL-1β ELISA, SYTOX Green uptake | Quantify different cell death modalities | Multiparametric approaches recommended for PANoptosis |
| Genetic Tools | Caspase-8 knockout cells, cFLIP overexpression, ZBP1 siRNA | Modulate specific pathway components | CRISPR/Cas9 knockout preferred for permanent modification |
| PANoptosome Studies | ASC speck staining, Co-immunoprecipitation, Size exclusion chromatography | Analyze complex formation | Crosslinking may stabilize transient interactions |
Data Interpretation and Analysis
Quantitative Assessment of PANoptosis
When evaluating caspase-8 activation in the context of PANoptosis, researchers should employ multiple complementary approaches to confirm the simultaneous activation of apoptosis, pyroptosis, and necroptosis pathways. The following parameters provide evidence for bona fide PANoptosis:
Key Indicators:
- Biochemical Markers: Concurrent detection of caspase-8 and caspase-3 cleavage (apoptosis), GSDMD cleavage (pyroptosis), and MLKL phosphorylation (necroptosis) in the same cellular population [71] [72]
- Morphological Features: Cellular swelling (necroptosis/pyroptosis) with apoptotic body formation and membrane blebbing
- Inflammatory Profile: Significant release of IL-1β, IL-18, and DAMPs indicating inflammatory cell death
- Complex Formation: Immunoprecipitation evidence of PANoptosome assembly with caspase-8 incorporation
Experimental Controls:
- Include pathway-specific inhibitors to validate mechanism
- Use genetic knockouts of key PANoptosis components (ZBP1, ASC, caspase-8)
- Compare time courses to establish activation kinetics
Troubleshooting Common Challenges
Inconsistent PANoptosis Induction:
- Optimize stimulus concentration and duration for each cell type
- Ensure proper cell density and culture conditions
- Pre-test multiple PANoptosis inducers to identify most robust response for your system
Weak Caspase-8 Activation:
- Verify efficiency of stimulation conditions
- Check caspase-8 expression levels in your cell model
- Consider priming with IFN-γ to enhance ZBP1 expression [72]
Specificity Issues in Detection:
- Include appropriate controls for antibody validation
- Use multiple detection methods to confirm findings
- Employ genetic approaches to validate protein identities in complexes
The protocols outlined in this application note provide comprehensive methodologies for investigating the multifaceted roles of caspase-8 in regulating cell death crosstalk, particularly within the emerging paradigm of PANoptosis. As research in this field advances, these experimental approaches will enable deeper understanding of how caspase-8 serves as a molecular integrator of diverse cell death pathways and contributes to both physiological homeostasis and pathological inflammation. The continued refinement of these techniques will undoubtedly yield new insights into therapeutic targeting of cell death pathways in inflammatory diseases, cancer, and infectious disorders.
Caspase-8, a cysteine-aspartic protease, has traditionally been recognized for its initiator role in the extrinsic apoptosis pathway. However, emerging research has revealed its critical functions in regulating multiple cell death modalities (including pyroptosis and necroptosis), inflammatory responses, and innate immunity [36] [76]. This functional versatility positions caspase-8 as a molecule of significant therapeutic interest across diverse disease contexts, from cancer to inflammatory and infectious diseases [8] [36] [76]. This Application Note examines the therapeutic relevance of caspase-8 as both a biomarker and drug target, providing detailed experimental protocols for researchers investigating caspase-8 activation in extrinsic pathway research.
Caspase-8 in Disease Pathogenesis and Treatment Response
Regulatory Roles of Caspase-8 in Cell Death and Inflammation
Caspase-8 serves as a crucial molecular switch governing cell fate through its dual functions in cell death execution and restriction, as well as inflammation modulation [36] [76].
- Extrinsic Apoptosis: Caspase-8 is the initiator caspase in the extrinsic apoptosis pathway. Upon death receptor activation, it is recruited to the Death-Inducing Signaling Complex (DISC), where it undergoes activation and initiates a caspase cascade that leads to apoptotic cell death [15] [36].
- Necroptosis Restriction: In conditions of caspase-8 inhibition or deficiency, death receptor signaling can divert toward necroptosis, a lytic and inflammatory form of cell death. Caspase-8 activity normally cleaves critical necroptosis mediators like RIPK1 and RIPK3, thereby suppressing this pathway [76].
- Inflammation and PANoptosis: Recent research identifies caspase-8 as a core component of "PANoptosis," an integrated inflammatory cell death pathway that combines features of pyroptosis, apoptosis, and necroptosis. Caspase-8 can contribute to inflammatory responses by promoting IL-1β maturation and can form complexes within PANoptosomes, which regulate this multifaceted cell death process [36] [77].
Caspase-8 as a Therapeutic Target in Disease Models
Dysregulation of caspase-8 is implicated in the pathogenesis of numerous diseases, making it a promising therapeutic target.
- Cancer: Altered caspase-8 expression contributes to tumor resistance against chemotherapy. In ovarian cancer, for example, cisplatin-resistant cells exhibit decreased caspase-8 protein levels, and pre-exposure to cisplatin can resensitize cells to TRAIL-induced apoptosis by elevating caspase-8 expression [78]. In pancreatic cancer, targeting the caspase-8/c-FLIPL heterodimer with the small molecule FLIPinB enhances death ligand-induced apoptosis and synergizes with chemotherapeutic agents [79].
- Inflammatory Diseases: In severe SARS-CoV-2 infection, caspase-8 drives pathological inflammation and cytokine release independently of its apoptotic function. Caspase-8 deficiency reduces disease severity and viral load in murine models, linked to decreased IL-1β levels and pro-inflammatory responses [8].
- Ischemic Diseases: PANoptosis, with caspase-8 as a key regulator, contributes to tissue damage in cerebral, retinal, myocardial, and renal ischemia. Targeting these interconnected cell death pathways presents a promising therapeutic strategy [77].
Table 1: Caspase-8 Dysregulation and Therapeutic Targeting in Disease Models
| Disease Model | Caspase-8 Dysregulation | Therapeutic Approach | Experimental Outcome |
|---|---|---|---|
| Pancreatic Cancer [79] | Altered caspase-8/c-FLIPL heterodimer dynamics at the DISC | FLIPinB compound targeting c-FLIPL in the heterodimer + Gemcitabine + S63845 (Mcl-1 inhibitor) | Enhanced complex II assembly and synergistic elimination of cancer cells |
| Ovarian Cancer (Cisplatin-Resistant) [78] | Decreased caspase-8 protein expression | Cisplatin pre-exposure + recombinant human TRAIL (rhTRAIL) | Strongly elevated caspase-8 protein levels and enhanced apoptosis |
| Severe COVID-19 [8] | Increased caspase-8 activity, cleaving N4BP1 to promote NF-κB signaling | Genetic caspase-8 deficiency or broad-spectrum caspase inhibitor (emricasan) | Reduced disease severity, viral load, and IL-1β-driven inflammation |
| Hepatocellular Carcinoma (HCC) [76] | Dysregulation affecting PCD, inflammation, and tumor microenvironment | Targeting caspase-8 to restore PCD and modulate immune cell function | Potential strategy to overcome therapy resistance and improve prognosis |
Experimental Protocols for Measuring Caspase-8 Activation
Protocol: Measuring Caspase-8 Activity at the Death-Inducing Signaling Complex (DISC)
This protocol enables the specific analysis of caspase-8 activation within its native signaling complex, which is critical for understanding the initial events in extrinsic apoptosis [15].
Materials and Reagents
- Cell Line: HeLa-CD95 cells (or other adherent/suspension lines sensitive to CD95L-induced apoptosis).
- Induction: Soluble CD95L (e.g., leucine zipper-tagged).
- Lysis Buffer: 20 mM Tris/HCl pH 7.4, 1% Triton X-100, 10% glycerol, 150 mM NaCl, supplemented with protease inhibitors.
- Immunoprecipitation (IP): Anti-CD95 (e.g., mouse monoclonal anti-CD95, Santa Cruz sc-8009) and protein G Sepharose beads.
- Caspase-8 Activity Assay: Caspase-8 fluorogenic substrate (e.g., IETD-AFC) in assay buffer (100 mM HEPES pH 7.4, 10% sucrose, 0.1% CHAPS, 10 mM DTT).
- Western Blotting: Primary antibodies: anti-caspase-8 (e.g., clone C15), anti-FADD, anti-c-FLIP. Secondary HRP-conjugated antibodies.
Step-by-Step Procedure
- Cell Culture and Preparation:
- Seed HeLa-CD95 cells at a density of 5 x 10⁶ cells per 14.5 cm plate in DMEM F12 medium supplemented with 10% FCS. Use at least three plates per condition for triplicates (Western blot control and caspase-8 assay duplicates). Incubate overnight at 37°C with 5% CO₂ [15].
- Apoptosis Induction and Cell Lysis:
- Stimulate cells with 1 µg/mL CD95L for the desired time (e.g., 30-90 minutes). Include an unstimulated control.
- Discard medium, wash cells with cold PBS, and lyse them in 1 mL of ice-cold lysis buffer for 30 minutes. Centrifuge the lysates at 15,000 × g for 15 minutes at 4°C to remove insoluble material [15].
- DISC Immunoprecipitation:
- Incubate the pre-cleared supernatant with 2 µg of anti-CD95 antibody for 2 hours at 4°C with gentle rotation.
- Add protein G Sepharose beads and incubate for an additional hour.
- Pellet the beads by brief centrifugation and wash three times with lysis buffer [15].
- Caspase-8 Activity Measurement:
- Resuspend the IP beads in 100 µL of caspase-8 assay buffer containing 200 µM IETD-AFC substrate.
- Transfer the suspension to a 96-well plate and incubate at 37°C.
- Measure fluorescence (excitation 400 nm, emission 505 nm) at regular intervals over 1-2 hours using a plate reader [15].
- Western Blot Analysis:
- After the activity assay, add Laemmli buffer to the beads, boil for 5 minutes, and separate the proteins by SDS-PAGE.
- Transfer to a PVDF membrane and immunoblot for caspase-8, FADD, and c-FLIP to confirm IP efficiency and analyze protein processing [15].
Data Analysis
- Calculate caspase-8 activity from the linear range of the fluorescence increase, normalized to the protein concentration of the input lysate or the amount of immunoprecipitated receptor.
- Correlate enzymatic activity with the cleavage status of caspase-8 (appearance of p43/p41 and p18 fragments) and the composition of the DISC on Western blots.
Protocol: Spatial-Temporal Monitoring of Caspase-8 Activity in Live Cells
This protocol utilizes FRET-based biosensors to monitor the dynamics and subcellular localization of caspase-8 activity in real-time within single living cells [21] [80].
Materials and Reagents
- FRET-based Caspase-8 Biosensor: Plasmid encoding a caspase-8 sensor (e.g., CFP-IETD-Venus).
- Cell Line: HeLa or other transfectable cell lines.
- Transfection Reagent: Suitable for the cell line (e.g., lipofection).
- Live-Cell Imaging: Glass-bottom culture dishes and phenol-red free imaging medium.
- Microscopy: Confocal or widefield fluorescence microscope with environmental control (37°C, 5% CO₂) and appropriate filter sets for CFP and YFP.
Step-by-Step Procedure
- Sensor Expression:
- Transiently transfect cells with the CFP-IETD-Venus biosensor plasmid using standard transfection protocols. Allow 24-48 hours for expression [21].
- Image Acquisition:
- Prior to imaging, replace the medium with pre-warmed phenol-red free imaging medium.
- Mount the dish on the microscope stage. Select fields of view containing healthy, moderately expressing cells.
- Induce apoptosis by adding CD95L or TRAIL directly to the medium during image acquisition.
- Acquire time-lapse images of both CFP and FRET (YFP) channels every 5-10 minutes for several hours [21] [80].
- Data Processing:
- Using image analysis software, define regions of interest (ROIs) for the cytosol, plasma membrane, and/or other compartments.
- For each time point and ROI, calculate the FRET ratio (YFP channel intensity / CFP channel intensity) [80].
Data Analysis
- A decrease in the FRET ratio over time indicates caspase-8-mediated cleavage of the biosensor.
- Compare the kinetics and magnitude of FRET ratio changes between different subcellular compartments to determine spatial activation patterns. Studies have shown that caspase-8 is significantly more active at the plasma membrane than in the cytosol upon CD95 activation [80].
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Caspase-8 and Extrinsic Apoptosis Research
| Reagent / Assay | Specific Example | Function / Application |
|---|---|---|
| Anti-Caspase-8 Antibodies | Mouse monoclonal (clone C15) [15] | Immunoprecipitation and Western blot detection of caspase-8 |
| Anti-CD95/Fas Antibodies | Mouse monoclonal (e.g., sc-8009) [15] | Immunoprecipitation of the DISC; receptor activation studies |
| Death Ligands | Recombinant CD95L, TRAIL [78] [79] | Specific activation of death receptors to initiate extrinsic apoptosis |
| Caspase-8 Activity Assays | Fluorogenic substrate IETD-AFC [15] | Quantitative measurement of caspase-8 enzymatic activity in lysates or IP samples |
| Live-Cell Caspase-8 Sensors | FRET-based biosensor (CFP-IETD-Venus) [21] [80] | Real-time, spatial-temporal monitoring of caspase-8 activation in single living cells |
| Pharmacologic Inhibitors | Broad-spectrum (e.g., emricasan) [8]; Specific (z-IETD-fmk) | To dissect the contribution of caspase-8 to cell death and inflammation |
| Pharmacologic Activators | FLIPinB [79] | Small molecule that targets the caspase-8/c-FLIPL heterodimer to enhance caspase-8 activation |
Signaling Pathway and Experimental Workflow Diagrams
Caspase-8 in Death Receptor Signaling and Cell Fate
Experimental Workflow for DISC Caspase-8 Activity Assay
Conclusion
The precise measurement of caspase-8 activation is fundamental to understanding cell fate decisions in health and disease. This synthesis of foundational knowledge, methodological protocols, and validation strategies highlights that caspase-8 functions not only as a critical initiator of extrinsic apoptosis but also as a key regulator of non-apoptotic inflammatory signaling. Future research directions should focus on developing more sensitive, single-cell resolution tools to dissect the heterogeneity of caspase-8 activation within populations. Furthermore, exploring the therapeutic potential of modulating caspase-8—either to induce cell death in cancer or to curb pathological inflammation in diseases like COVID-19 and Alzheimer's—represents a promising frontier for biomedical and clinical research. The integration of quantitative models with advanced experimental data will continue to refine our ability to predict and control this pivotal cellular switch.
References
- 1. Dissecting DISC regulation via pharmacological targeting of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7308354/]
- 2. Protocol for measuring the caspase - 8 activity at the DED filaments in... [https://pubmed.ncbi.nlm.nih.gov/41066241/]
- 3. by the Caspase | SpringerLink Activation Extrinsic Pathway [https://link.springer.com/chapter/10.1385/1-59259-851-X:111]
- 4. A Death Effector Domain Chain DISC Model Reveals ... [https://www.sciencedirect.com/science/article/pii/S1097276512003875]
- 5. Caspase-8; regulating life and death - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5417704/]
- 6. Cryo-EM Structure of Caspase-8 Tandem DED Filament ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5089849/]
- 7. Inducible Dimerization and Inducible Cleavage Reveal a ... [https://www.sciencedirect.com/science/article/pii/S0021925819355899]
- 8. Non-apoptotic caspase-8 is critical for orchestrating ... [https://www.nature.com/articles/s41467-025-65098-z]
- 9. Quantitative analysis of pathways controlling extrinsic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2858979/]
- 10. Monitoring Caspase Activity in Living Cells Using Fluorescent ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1615789/]
- 11. Platelets express adaptor proteins of the extrinsic apoptosis ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0244848]
- 12. Caspase Assays | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/life-science/cell-analysis/cell-viability-and-regulation/apoptosis/caspase-assays.html]
- 13. Multiple entanglements of different cell death pathways, in ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2025.1667611/full]
- 14. Apoptosis: A Comprehensive Overview of Signaling ... [https://www.mdpi.com/2073-4409/13/22/1838]
- 15. Protocol for measuring the caspase-8 activity at the DED ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12539301/]
- 16. Caspase-8 in inflammatory diseases: a potential therapeutic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11463096/]
- 17. Multiple roles of caspase-8 in cell death, inflammation, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8664275/]
- 18. Caspase-8 Acts in a Non-enzymatic Role as a Scaffold for ... [https://www.sciencedirect.com/science/article/pii/S1097276517300485]
- 19. Caspase-8 auto-cleavage regulates programmed cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9345959/]
- 20. Macrophage caspase-8 inhibition accelerates necrotic core ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1513637/full]
- 21. The molecular mechanism of apoptosis upon caspase-8 activation: Quantitative experimental validation of a mathematical model - ScienceDirect [https://www.sciencedirect.com/science/article/pii/S0167488912001991]
- 22. BD Pharmingen™ Purified Mouse Anti-Human Caspase-8 [https://www.bdbiosciences.com/en-us/products/reagents/western-blotting-and-molecular-reagents/purified-mouse-anti-human-caspase-8.551242]
- 23. (IP) and co- Immunoprecipitation immunoprecipitation protocol [https://www.abcam.com/en-us/technical-resources/protocols/immunoprecipitation]
- 24. Extrinsic apoptosis and necroptosis in telencephalic ... [https://www.nature.com/articles/s41418-025-01594-5]
- 25. Integrated real-time imaging of executioner caspase dynamics ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12328661/]
- 26. FRET Based Biosensor: Principle Applications Recent ... [https://www.mdpi.com/2075-4418/13/8/1375]
- 27. TRAF2 Sets a Threshold for Extrinsic Apoptosis by Tagging ... [https://www.sciencedirect.com/science/article/pii/S109727651200860X]
- 28. Activation of the integrated stress response and loss ... [https://www.nature.com/articles/s41420-025-02625-3]
- 29. Prognostic and therapeutic relevance of FLIP and ... [https://www.nature.com/articles/cddis2013481]
- 30. FRET and BRET-Based Biosensors in Live Cell Compound ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4112949/]
- 31. Kinetic quantification of apoptosis using Incucyte® live-cell ... [https://www.news-medical.net/whitepaper/20251027/Kinetic-quantification-of-apoptosis-using-Incucytec2ae-live-cell-imaging-assays.aspx]
- 32. Cleaved Caspase-8 (Asp387) Antibody #9429 [https://www.cellsignal.com/products/primary-antibodies/cleaved-caspase-8-asp387-antibody/9429?srsltid=AfmBOor9BLCoW7pOhuWOWzIFo8MFbxuSODDc44opx0jyhzNJzGxAvdFY]
- 33. Sheet Protector Strategy for Western Blot to Reduce ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12462392/]
- 34. 2024 Guide to Quantitative Western Blot Publication [https://www.thermofisher.com/blog/life-in-the-lab/2023-guide-to-quantitative-western-blot-publication/]
- 35. Follow Publication Standards for Western Blot Data ... [https://www.licorbio.com/applications/quantitative-western-blots/produce-better-blots]
- 36. Caspase-8 in inflammatory diseases: a potential ... [https://cmbl.biomedcentral.com/articles/10.1186/s11658-024-00646-x]
- 37. Caspase-3 feedback loop enhances Bid-induced AIF ... [https://www.nature.com/articles/cddis2015276]
- 38. Caspases as master regulators of programmed cell death [https://www.nature.com/articles/s12276-025-01470-9]
- 39. Regulation of poly(ADP-ribose) polymerase-1 (PARP-1) gene ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2175517/]
- 40. High numbers of active caspase 3–positive Reed ... [https://www.sciencedirect.com/science/article/pii/S0006497120599692]
- 41. FITC Active Caspase-3 Apoptosis Kit [https://www.bdbiosciences.com/en-us/products/reagents/flow-cytometry-reagents/research-reagents/panels-multicolor-cocktails-ruo/fitc-active-caspase-3-apoptosis-kit.550480]
- 42. Detecting Cleaved Caspase-3 in Apoptotic Cells by Flow ... [https://pubmed.ncbi.nlm.nih.gov/27803251/]
- 43. Pharmacological targeting of caspase-8/c-FLIPL ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11698904/]
- 44. Measuring Composition of CD95 Death-Inducing Signaling ... [https://www.jove.com/t/62842/measuring-composition-cd95-death-inducing-signaling-complex]
- 45. Cryo-EM structural analysis of FADD:Caspase-8 ... [https://www.nature.com/articles/s41467-020-20806-9]
- 46. Defining Characteristics of Types I and II Apoptotic Cells in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1503670/]
- 47. Multiple entanglements of different cell death pathways, in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12644007/]
- 48. Cleavage by Caspase 8 and Mitochondrial Membrane ... [https://www.sciencedirect.com/science/article/pii/S0021925820432132]
- 49. Differential response of head and neck cancer cell lines to ... [https://www.nature.com/articles/bjc2014521]
- 50. c-FLIPL is a dual function regulator for caspase-8 activation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC125398/]
- 51. Deciphering DED assembly mechanisms in FADD ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11074299/]
- 52. Mathematical Modeling of Cell Death and Survival [https://pmc.ncbi.nlm.nih.gov/articles/PMC12651094/]
- 53. Dynamical analysis of a model of BCL-2-dependent ... [https://www.nature.com/articles/s41540-025-00615-w]
- 54. Single-cell dynamics reveal a stress-induced decision ... [https://www.sciencedirect.com/science/article/pii/S277297612500203X]
- 55. Apoptosis Protocols [https://health.usf.edu/medicine/corefacilities/flowcytometry/methods-apoptosis]
- 56. Color Mapping - FlowJo Documentation [https://docs.flowjo.com/flowjo/graphs-and-gating/data-visualization-and-display/color-mapping/]
- 57. Mitochondrial outer membrane permeabilization: a focus on the role of mitochondrial membrane structural organization - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5578938/]
- 58. Mitochondrial outer membrane permeabilization [https://en.wikipedia.org/wiki/Mitochondrial_outer_membrane_permeabilization]
- 59. Caspase-8: Arbitrating Life and Death in the Innate Immune ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11853578/]
- 60. Multiple entanglements of different cell death pathways, in ... [https://pubmed.ncbi.nlm.nih.gov/41306288/?utm_source=FeedFetcher&utm_medium=rss&utm_campaign=None&utm_content=1VSjW0JqT_v3o4ef_i7fczOFyon91YoRD2Oh6DG1jbIZYnq94z&fc=None&ff=20251127070858&v=2.18.0.post22+67771e2]
- 61. Monitoring Caspase Activity in Living Cells Using Fluorescent ... [https://pubmed.ncbi.nlm.nih.gov/15161627/]
- 62. Caspase-mediated loss of mitochondrial function and generation of reactive oxygen species during apoptosis - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2172744/]
- 63. The molecular mechanism of apoptosis upon caspase - 8 ... activation [https://pubmed.ncbi.nlm.nih.gov/22801217/]
- 64. of apoptosis | Cell Communication and... Mathematical modeling [https://biosignaling.biomedcentral.com/articles/10.1186/1478-811X-11-44]
- 65. Article [http://protein.bio.msu.ru/biokhimiya/contents/v85/full/85101331.html]
- 66. In Vivo Imaging of Hierarchical Spatiotemporal Activation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3503975/]
- 67. Heterogeneous responses to low level death receptor activation are... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6779275/]
- 68. RIP1-dependent linear and nonlinear recruitments of ... [https://link.springer.com/article/10.1007/s13238-020-00810-x]
- 69. Endothelial Caspase-8 prevents fatal necroptotic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9883523/]
- 70. Insights on the crosstalk among different cell death ... [https://www.nature.com/articles/s41420-025-02328-9]
- 71. PANoptosis: Cross-Talk Among Apoptosis, Necroptosis, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12184785/]
- 72. PANoptosis in neurological disorders: mechanisms ... [https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1579360/full]
- 73. PANoptosis in infectious Diseases: Mechanisms ... [https://www.sciencedirect.com/science/article/abs/pii/S0006291X25012203]
- 74. Evolutionary and Functional Analysis of Caspase-8 and ASC ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12066828/]
- 75. Mitochondrial orchestration of PANoptosis: mechanisms, ... [https://www.nature.com/articles/s41420-025-02750-z]
- 76. Targeting caspase-8: a new strategy for combating ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11669555/]
- 77. The molecular mechanisms and therapeutic implications of ... [https://link.springer.com/article/10.1007/s10495-025-02157-2]
- 78. Drug-induced caspase 8 upregulation sensitises cisplatin ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3078595/]
- 79. Pharmacological targeting of caspase-8/c-FLIP L ... [https://www.nature.com/articles/s42003-024-07409-6]
- 80. Caspase-8 cleaves its substrates from the plasma ... [https://www.nature.com/articles/cdd2012156]