Mitochondrial Membrane Potential Dye Redistribution Artifacts: From Mechanisms to Solutions in Biomedical Research
Accurate measurement of mitochondrial membrane potential (ΔΨm) is crucial for understanding cellular health, apoptosis, and drug mechanisms.
Mitochondrial Membrane Potential Dye Redistribution Artifacts: From Mechanisms to Solutions in Biomedical Research
Abstract
Accurate measurement of mitochondrial membrane potential (ΔΨm) is crucial for understanding cellular health, apoptosis, and drug mechanisms. However, dye redistribution artifacts frequently compromise data integrity, particularly during pharmacological treatments. This article examines the fundamental mechanisms underlying these artifacts, including concentration-dependent saturation effects and treatment-induced alterations in membrane properties. We present methodological frameworks for detecting and mitigating artifacts across experimental systems, from basic microscopy to advanced super-resolution techniques. Troubleshooting protocols address common challenges with uncouplers, inhibitors, and combination therapies. Finally, we establish validation strategies using orthogonal assays and comparative analysis of next-generation ΔΨm-insensitive probes. This comprehensive guide empowers researchers to improve experimental reliability in mitochondrial research and drug development.
Understanding Dye Redistribution: The Science Behind ΔΨm Measurement Artifacts
Fundamental Principles of ΔΨm-Sensitive Dye Accumulation Mechanisms
FAQ: Core Principles and Mechanisms
What is the fundamental principle that allows ΔΨm-sensitive dyes to accumulate in mitochondria? These dyes are typically lipophilic, cationic compounds that distribute across the mitochondrial inner membrane in response to the electrical gradient. The mitochondrial matrix is negatively charged relative to the intermembrane space, creating an electrical potential (ΔΨm) that drives the accumulation of positively charged molecules. Dyes equilibrate in a Nernstian fashion, accumulating in the mitochondrial matrix in inverse proportion to the ΔΨm. A more negative (i.e., more polarized) ΔΨm will accumulate more dye, and a less negative (depolarized) potential will accumulate less dye [1].
Why is the charge on the dye molecule so critical? The negative interior of the mitochondrial matrix (typically -150 to -180 mV) creates a strong electrophoretic force that attracts and concentrates positively charged (cationic) dyes. This charge-based accumulation allows the fluorescence intensity to serve as a proxy for the magnitude of the ΔΨm. The direction of the membrane potential favors inward transport of cations, which is the fundamental property exploited by these probes [2] [1].
What is the difference between "slow-response" and "fast-response" membrane potential probes? Slow-response dyes, which include most common ΔΨm probes like TMRM and JC-1, function by entering depolarized cells and binding to proteins or membranes. Increased depolarization results in additional dye influx and an increase in fluorescence, while hyperpolarization is indicated by a decrease in fluorescence. In contrast, fast-response probes are molecules that change their structure in response to the surrounding electric field and can detect transient (millisecond) potential changes. Slow-response probes are most often used to explore mitochondrial function and cell viability [3].
FAQ: Dye Selection and Operational Modes
How do I choose between different ΔΨm-sensitive dyes? The choice of dye depends on your experimental goals, detection method, and the need for quantitative vs. qualitative assessment. The table below summarizes key characteristics of common dyes.
Table 1: Comparison of Common ΔΨm-Sensitive Dyes
| Probe | Spectra (Ex/Em) | Primary Use & Strengths | Key Limitations & Considerations |
|---|---|---|---|
| TMRM / TMRE | ~549/573 nm (e.g., Rhodamine) | Best for acute studies measuring pre-existing ΔΨm; low mitochondrial binding and minimal inhibition of the Electron Transport Chain (ETC) [1]. | Fast equilibration can make it less suited for some quenching-mode studies. Requires careful concentration optimization [1]. |
| Rhodamine 123 | ~507/529 nm | Best for fast-resolution acute studies in quenching mode; slightly less ETC inhibition than TMRE [1]. | More slowly permeant than TMRM/TMRE [1]. |
| JC-1 | J-aggregates: ~585/590 nmMonomers: ~514/529 nm | Provides ratiometric (dual-color) assessment; well-suited for clear "yes/no" discrimination of polarization state, such as in apoptosis studies [1]. | Very sensitive to dye concentration; aggregate form can be influenced by factors other than ΔΨm (e.g., surface-to-volume ratios, H₂O₂) [1]. |
| DiOC₆(3) | ~484/501 nm | Often used for flow cytometry [1]. | Requires very low concentrations (<1 nM) to specifically monitor ΔΨm and to prevent respiratory toxicity [1]. |
| LDS 698 | Ex: 460/470 nm / Em: 580-700 nm | Novel dye with high sensitivity for detecting subtle ΔΨm changes; highly photostable with low cytotoxicity [4]. | Less established in the literature compared to traditional dyes; users may need to validate performance in their specific system [4]. |
| MitoTracker Probes | Varies by product | Some variants (e.g., MitoTracker Red CMXRos) are fixable, allowing for subsequent cell permeabilization and immunostaining [5]. | Many MitoTracker dyes (e.g., MitoTracker Red FM) covalently bind to thiol groups and do not respond to subsequent changes in potential after fixation [4]. |
What is the difference between "quenching" and "non-quenching" modes for these dyes? The operational mode depends on the concentration of the dye used [1]:
- Non-Quenching Mode: Achieved by using very low dye concentrations (e.g., 1-30 nM for TMRM). At these concentrations, the dye is dilute in the mitochondrial matrix and fluoresces brightly. A depolarization causes dye to leave the mitochondria, resulting in a decrease in fluorescence intensity.
- Quenching Mode: Achieved by using high dye concentrations (e.g., >50-100 nM for TMRM). The high intra-mitochondrial concentration leads to aggregation and self-quenching of fluorescence. A depolarization causes dye to redistribute out of the mitochondria, leading to a dilution of the dye and a paradoxical increase in fluorescence (unquenching).
Diagram: Dye Accumulation and Fluorescence Based on ΔΨm and Operational Mode
FAQ: Troubleshooting and Experimental Design
I am seeing high background fluorescence outside of my mitochondria. What can I do? High cytosolic or nuclear background is a common issue. Solutions include:
- Optimize Dye Concentration: Use the lowest possible dye concentration that provides a detectable signal. This is critical for reducing non-specific background [1].
- Use Background Suppressors: Some commercial kits provide background suppressor reagents designed to reduce extracellular and cytosolic background signals [3].
- Ensure Healthy Cells: Verify cell health, as compromised plasma membrane integrity can lead to non-specific dye accumulation.
- Proper Washing: Include careful washing steps after dye loading to remove excess dye from the media (particularly important for dyes used in quenching mode after washout) [1].
My dye signal is saturated and not reflecting changes in ΔΨm. What is wrong? Signal saturation is frequently a concentration issue. For dyes like TMRM, high concentrations (>40 nM) can saturate the cristae membranes, causing the signal to reflect dye distribution rather than the true potential gradient. To accurately measure potential gradients across mitochondrial sub-compartments (cristae vs. inner boundary membrane), use lower dye concentrations (e.g., 1.35-5.4 nM) [6]. Always perform a concentration curve to find the optimal, non-saturating level for your specific cell type and experimental setup [6] [1].
Why can't I use a ΔΨm-sensitive dye in fixed cells? ΔΨm-sensitive dyes require an active, energized mitochondrial membrane to distribute according to the potential. Fixation kills the cells and disrupts all metabolic activity and ionic gradients, including the ΔΨm. Once fixed, there is no mitochondrial activity to drive accumulation [5]. If you need to fix cells after staining, you must use a fixable structural mitochondrial dye (e.g., MitoTracker Green FM in some contexts, or antibodies against mitochondrial proteins like COX IV or TOMM20) that binds covalently or is retained through the fixation process [5].
My ΔΨm measurements are being affected by other factors. What are potential sources of artifact? A critical source of artifact is the influence of non-protonic charges. The ΔΨm is a measure of the total electrical gradient, not exclusively the proton gradient (ΔpH). Changes in the distribution of other ions, particularly calcium (Ca²⁺), can significantly alter ΔΨm independent of respiratory status. For example, a release of mitochondrial Ca²⁺ can cause hyperpolarization even when the proton gradient is collapsing [1]. Always consider parallel assays and controls to validate your findings, such as using ion chelators or measuring mitochondrial Ca²⁺ directly [1].
Experimental Protocols and Validation
Basic Protocol: Measuring ΔΨm with TMRM in Non-Quenching Mode This protocol is adapted for live-cell imaging and is suitable for tracking acute changes [1].
Dye Loading:
- Prepare a working solution of TMRM (1-30 nM) in pre-warmed cell culture medium or buffer. Note: The optimal concentration must be determined empirically for your cell type.
- Incubate cells with the dye for 15-30 minutes at 37°C and appropriate CO₂ levels.
- For some experiments, the dye can be maintained in the bath during imaging to allow for continuous equilibration.
Image Acquisition:
- Image using a fluorescence microscope with appropriate filter sets for tetramethylrhodamine (e.g., Ex/Em ~549/573 nm).
- Use low illumination intensity and short exposure times to minimize phototoxicity and dye bleaching.
Controls and Calibration:
- Full Depolarization Control: At the end of the experiment, apply a mitochondrial uncoupler such as FCCP (1-10 µM) to fully collapse the ΔΨm. This establishes the minimum fluorescence signal.
- Inhibitor Control: Application of oligomycin (1-5 µM), an ATP synthase inhibitor, can hyperpolarize the membrane by inhibiting proton flow through the synthase, providing a positive control for hyperpolarization.
Advanced Protocol: Analyzing Spatial Membrane Potential Gradients Super-resolution techniques like Structured Illumination Microscopy (SIM) can resolve ΔΨm differences between the cristae membrane (CM) and inner boundary membrane (IBM) [6].
Staining:
- Co-stain cells with a potential-sensitive dye (e.g., TMRM at a low, non-saturating concentration of 2.7-5.4 nM) and a potential-insensitive structural dye (e.g., MitoTracker Green FM, 500 nM).
Image Acquisition:
- Perform simultaneous dual-channel super-resolution imaging (e.g., using SIM).
Data Analysis:
- IBM Association Index: Use an automated threshold on the MitoTracker Green channel to define mitochondrial boundaries. Create inner (CM) and outer (IBM) regions and calculate the ratio of TMRM fluorescence intensity (IBM/CM) [6].
- ΔFWHM Method: Analyze the cross-section intensity profiles of both dyes. Calculate the difference in the Full Width at Half Maximum (FWHM) between the structural marker (MTG) and the potential probe (TMRM). A larger ΔFWHM indicates greater TMRM accumulation in the cristae [6].
Diagram: Workflow for Spatial Membrane Potential Analysis
The Scientist's Toolkit: Essential Reagents and Materials
Table 2: Key Research Reagents for ΔΨm Studies
| Reagent / Material | Function / Description | Example Use Case |
|---|---|---|
| TMRM / TMRE | Cationic, lipophilic dye for dynamic ΔΨm measurement. | Live-cell imaging of acute ΔΨm changes in non-quenching or quenching mode [1]. |
| MitoTracker Green FM | Cell-permeant dye that accumulates in mitochondria regardless of membrane potential; useful as a structural marker. | Labeling mitochondrial mass and morphology; can be used as a reference channel for ratiometric analysis with TMRM [6]. |
| FCCP | Protonophore and mitochondrial uncoupler. Collapses the proton gradient and ΔΨm. | Positive control for complete mitochondrial depolarization [1]. |
| Oligomycin | ATP synthase inhibitor. | Control to induce hyperpolarization by preventing proton flow through ATP synthase [1]. |
| Rotenone & Antimycin A | Inhibitors of Complex I and III of the Electron Transport Chain, respectively. | Used to inhibit proton pump activity and investigate the source of ΔΨm generation [6]. |
| Background Suppressor (e.g., BackDrop) | Reagent designed to reduce extracellular and cytosolic background fluorescence. | Improving signal-to-noise ratio in neuronal cells or other samples with high background [3]. |
| Ion Chelators (e.g., BAPTA-AM, EGTA) | Chelators of divalent cations like Ca²⁺. | Control experiments to dissect the contribution of non-protonic ions (e.g., Ca²⁺) to the measured ΔΨm [1]. |
Accurate measurement of the mitochondrial membrane potential (ΔΨm) is fundamental to assessing cellular health, metabolic activity, and the efficacy of therapeutic compounds. Fluorescent cationic dyes are indispensable tools for this purpose, as their distribution across the inner mitochondrial membrane follows the Nernst equation, accumulating within mitochondria in proportion to the ΔΨm [1] [7]. However, a significant and often overlooked source of experimental artifact stems from the concentration of the dye itself. Using a dye concentration outside its optimal linear range can lead to two primary artifacts: saturation effects, which mask true changes in potential, and false gradients, which create illusory spatial patterns of mitochondrial polarization that do not reflect biological reality. This guide details the mechanisms behind these artifacts and provides protocols for their identification and avoidance, ensuring data integrity in drug discovery and basic research.
Understanding the Artifacts: Mechanisms and Consequences
Saturation Effects: Masking True ΔΨm Changes
Saturation occurs when the intra-mitochondrial dye concentration reaches a level where its fluorescence intensity no longer increases linearly with ΔΨm. The relationship between potential and dye accumulation is logarithmic; a ~60 mV change in ΔΨm results in a 10-fold change in dye concentration [7]. At high dye concentrations, the mitochondrial matrix becomes saturated with the probe. Subsequent increases in ΔΨm cannot cause further proportional accumulation, leading to a ceiling effect where genuine hyperpolarization is undetectable [1]. Conversely, mild depolarization may not significantly reduce the fluorescence signal until the dye concentration falls below the saturation threshold, blunting the observed dynamic range of the assay.
False Gradients: The TMRM Distribution Paradox
Perhaps a more insidious artifact is the generation of false spatial gradients within mitochondria. Super-resolution microscopy studies have revealed that the distribution of dyes like TMRM between the inner boundary membrane (IBM) and the cristae membrane (CM) is highly concentration-dependent [6].
- At low, non-saturating concentrations (e.g., 1.35-5.4 nM), TMRM primarily accumulates in the cristae, where the proton pumps generate a more negative potential (ΔΨC) compared to the IBM (ΔΨIBM) [6]. This distribution accurately reflects the underlying physiology.
- At high, saturating concentrations (e.g., 40.5-81 nM), the cristae become saturated with TMRM. Excess dye then accumulates in the IBM, artificially increasing the fluorescence signal from this compartment [6]. This flattens the apparent ΔΨC/ΔΨIBM ratio and can be misinterpreted as a loss of cristae polarization or a change in cristae junction permeability, when it is merely a dye saturation effect.
The table below summarizes the key differences observed at low and high TMRM concentrations in HeLa and EA.hy926 cells, as quantified by super-resolution microscopy.
Table 1: Quantifying Concentration-Dependent Artifacts in TMRM Staining
| TMRM Concentration | ∆FWHM Value | IBM Association Index | Interpretation |
|---|---|---|---|
| Low (1.35 - 5.4 nM) | Higher [6] | Lower [6] | Accurate reflection of higher cristae potential |
| High (40.5 - 81 nM) | Lower [6] | Higher [6] | Saturation artifact; false gradient observed |
Impact on Data Interpretation in Drug Screening
In a drug development context, these artifacts can lead to severe misinterpretation. A test compound that genuinely hyperpolarizes mitochondria may show no effect if a saturating dye concentration is used. Furthermore, a drug-induced change in cristae structure or function could be masked by, or mistaken for, a saturation artifact. Relying on such flawed data can derail lead optimization and mechanism-of-action studies.
Troubleshooting FAQs: Identifying and Resolving Artifact Issues
Q1: Our JC-1 results show a strong green signal but very little red J-aggregate fluorescence. Could this be a concentration issue?
Yes, this is a classic symptom. For JC-1 to form red fluorescent J-aggregates, it must reach a critical concentration within the mitochondria, which is driven by a sufficiently negative ΔΨm [8]. Several concentration-related problems can prevent this:
- Insufficient dye loading concentration: The extracellular working concentration of JC-1 may be too low to achieve the intra-mitochondrial threshold for aggregation, even in healthy, polarized mitochondria [9]. It is critical to titrate the dye concentration for each cell type.
- Incorrect buffer preparation: JC-1 has limited solubility in aqueous buffer. If the stock solution is not properly prepared in DMSO first, or if the working solution contains particulate crystals, the effective dye concentration will be incorrect [9]. Always follow the manufacturer's preparation instructions precisely.
- Underlying depolarization: Of course, a lack of red fluorescence can also indicate true mitochondrial depolarization, such as during early apoptosis [8]. This highlights the necessity of including controls with healthy cells and depolarized controls (e.g., using CCCP or FCCP) to validate your staining protocol.
Q2: We observe heterogeneous staining in our cell population. Is this biological heterogeneity or an artifact?
It could be either, and careful controls are needed to distinguish them. Genuine biological heterogeneity in ΔΨm exists between cells and even between mitochondria within a single cell [10]. However, technical artifacts can mimic this.
- True Heterogeneity: Subpopulations of cells with different metabolic states or mitochondria in different subcellular locations can have different ΔΨm [10].
- Artifact-Driven Heterogeneity: Inconsistent dye loading due to variable cell density, poor reagent mixing, or prolonged staining times that lead to dye precipitation can create false heterogeneity. To rule this out, ensure consistent experimental conditions and include a depolarization control. If the "heterogeneous" pattern collapses to a uniformly low signal after uncoupler treatment, the original signal likely reflected true ΔΨm.
Q3: Our positive control (CCCP) does not fully collapse the fluorescence signal. What could be wrong?
An incomplete response to a potent uncoupler like CCCP is a strong indicator of artifact, often related to probe modification or concentration.
- Probe Saturation: At very high dye concentrations, even after depolarization, the absolute amount of dye remaining in the mitochondria due to non-specific binding or matrix saturation can produce a significant fluorescence signal that is independent of ΔΨm [11].
- Dye Modification and Trapping: Cationic dyes like Rhodamine 123 can be modified inside cells, for example, by esterases or cytochrome P450, converting them to forms that are trapped and whose fluorescence is no longer sensitive to ΔΨm [11]. This can be tested by using efflux inhibitors like amiodarone or by switching to more robust dyes like TMRM.
- Insufficient Uncoupler Concentration/Time: Verify that your uncoupler is active and used at an appropriate concentration with sufficient incubation time.
Recommended Experimental Protocols for Artifact Avoidance
Protocol 1: Determining Optimal Dye Concentration via Titration
This protocol is essential for any new cell line or experimental setup.
- Plate cells in a multi-well plate suitable for your detection method (e.g., 96-well plate for a plate reader, chambered coverslips for microscopy).
- Prepare a dilution series of your chosen dye (e.g., TMRM, JC-1) spanning at least two orders of magnitude. For TMRM, a range of 1 nM to 100 nM is a good starting point [6].
- Load the dye into cells according to standard protocols for your cell type and dye. Incubate at 37°C.
- Image or read the fluorescence. Include controls with a depolarizing agent (e.g., 10-20 µM CCCP) for each concentration.
- Analyze the data. Plot fluorescence intensity (or the red/green ratio for JC-1) against dye concentration. The optimal concentration is the highest value within the linear range of the curve for the control cells, which also shows a strong, significant signal loss upon depolarization.
Protocol 2: Validating Spatial Gradients with Super-Resolution Imaging
This protocol uses the dye MitoTracker Green FM (MTG) as a spatial reference to control for morphology.
- Cell Staining: Co-stain cells with 500 nM MTG and a low, non-saturating concentration of TMRM (e.g., 2.7 nM) in respiration buffer for 30 minutes [6].
- Image Acquisition: Perform simultaneous dual-channel structured illumination microscopy (SIM) imaging of both dyes.
- Image Analysis:
- Use the MTG channel to define the mitochondrial boundaries.
- IBM Association Index: Calculate the ratio of TMRM fluorescence intensity at the mitochondrial boundary (IBM) to the intensity in the interior (CM). A lower index indicates higher cristae polarization [6].
- ΔFWHM Method: Measure the Full Width at Half Maximum (FWHM) of fluorescence intensity profiles across individual mitochondria for both MTG and TMRM. A larger difference (ΔFWHM) indicates TMRM accumulation in the cristae [6].
- Validation: Repeat the experiment with a high, saturating TMRM concentration (e.g., 40 nM). The observed gradient will diminish or disappear, demonstrating the concentration-dependent nature of the artifact.
The Scientist's Toolkit: Essential Reagents & Materials
Table 2: Key Reagents for Investigating Mitochondrial Membrane Potential Artifacts
| Reagent / Material | Function / Description | Key Considerations |
|---|---|---|
| TMRM / TMRE | Cationic, potential-sensitive dye for live-cell imaging. | Preferred for minimal mitochondrial binding and ETC inhibition. Use in non-quenching mode (low nM) for acute studies [1]. |
| JC-1 | Ratiometric, potential-sensitive dye that forms J-aggregates. | Ideal for flow cytometry and yes/no discrimination of polarization. Very sensitive to concentration; requires careful titration and validation [1] [8]. |
| MitoTracker Green FM | Structural mitochondrial dye; accumulates in mitochondria independent of ΔΨm. | Used as a morphological reference in super-resolution imaging to control for shape and location [6]. |
| CCCP / FCCP | Protonophores that uncouple the mitochondrial proton gradient, collapsing ΔΨm. | Essential negative control for validating dye response. Use at sufficient concentrations (e.g., 10-20 µM) [9] [8]. |
| Rotename / Antimycin A | Inhibitors of Electron Transport Chain Complex I and III, respectively. | Used to inhibit proton pump activity and test the dependency of observed signals on respiration [6]. |
| Succinate | Substrate for Complex II. Used in respiration buffers to energize isolated mitochondria or permeabilized cells. | Ensures mitochondria are actively respiring and generating ΔΨm at the start of an experiment [10]. |
| Polyvinyl Alcohol (PVA) | Coating agent for capillaries in CE-LIF to reduce mitochondrial adhesion. | Critical for techniques like capillary electrophoresis to prevent loss of sample and ensure reproducible separations [10]. |
Visualizing the Workflow and Artifact Mechanism
The following diagram illustrates the core concepts of dye behavior at different concentrations and the recommended experimental workflow to avoid artifacts.
Treatment-Induced Membrane Alterations That Distort Dye Distribution
Accurate measurement of cellular and organellar membrane potential is fundamental to research in cell biology, drug discovery, and toxicology. Fluorescent potentiometric dyes, which distribute across membranes according to the Nernst equation, are a primary tool for these investigations. However, experimental treatments can induce physical and chemical alterations to membrane systems, leading to aberrant dye distribution and significant data misinterpretation. This guide addresses the common sources of these artifacts and provides methodologies for their identification and mitigation.
Core Concepts: How Dyes and Membranes Interact
The Principle of Nernstian Distribution
Cationic dyes, such as TMRE and TMRM, are widely used to measure membrane potential. These lipophilic, positively charged dyes passively diffuse across membranes and accumulate in compartments with a negative internal potential [12]. The distribution is governed by the Nernst equation, where a -60 mV potential results in an approximately 10-fold higher internal concentration, and a mitochondrial potential of around -180 mV leads to very high accumulation, making mitochondria light up in fluorescent images [12].
Potential-Independent Staining Mechanisms
Some dyes, described as "potential-insensitive" (e.g., MitoTracker Green, MitoView Green), are highly hydrophobic. While their initial accumulation is driven by the membrane potential, their lipophilicity causes them to be retained in mitochondrial membranes even after depolarization, as they are less likely to diffuse back into the cytoplasm [13]. This property makes them useful for measuring mitochondrial mass, but their signal is not a reliable indicator of functional potential.
Identifying and Troubleshooting Common Artifacts
Treatment-induced changes can disrupt the normal dye-cell interaction, leading to artifacts. The table below summarizes common issues, their causes, and solutions.
Table 1: Common Artifacts in Membrane Potential Dye Experiments
| Observed Artifact | Potential Causes | Recommended Solutions & Counter-Screens |
|---|---|---|
| Unexpected High Fluorescence (False "Healthy" Signal) | Treatment-induced autofluorescence of compounds [14]. Dye sequestration in non-target compartments due to altered membrane permeability [13]. | Perform control wells with dye but no cells to test for compound-dye interaction. Implement an orthogonal assay (e.g., plate reader vs. imager) to confirm signal [14]. |
| Unexpected Low Fluorescence (False "Depolarized" Signal) | Compound-mediated fluorescence quenching [14]. Treatment cytotoxicity causing massive cell loss or death [14]. | Statistically analyze nuclear counts and stain intensity to identify cytotoxic outliers [14]. Manually review images for signs of cell rounding, detachment, or death. |
| Altered Cellular or Dye Localization | Treatment disrupts cell adhesion, leading to substantial cell loss [14]. The fluorescent label itself interacts non-specifically with the growth substrate [15]. | Use an adaptive image acquisition to capture more fields until a cell threshold is met [14]. Coat culture surfaces with fibronectin instead of poly-L-lysine to minimize dye-substrate interactions [15]. |
| Poor Signal-to-Noise or High Background | Autofluorescence from culture media components like riboflavins [14]. Contamination from lint, dust, or plastic fragments [14]. | Use phenol-red free media or media without fluorescent components. Ensure labware and environment are clean to minimize particulate contamination [14]. |
Experimental Protocols for Artifact Identification
Protocol 1: Validating Signal Specificity with Pharmacological Depolarization
This protocol confirms that the dye signal is dependent on membrane potential.
- Seed cells in a multi-well plate and culture until they reach the desired confluency.
- Load the dye (e.g., 50 nM TMRE or TMRM) according to standard protocols and incubate to allow equilibration [12].
- Establish a depolarization control. Treat a subset of wells with a depolarizing agent:
- For plasma membrane: Add a high-K+ buffer.
- For mitochondria: Add 10 µM Carbonyl cyanide m-chlorophenyl hydrazone (CCCP) or 10 µM FCCP.
- Include an untreated control set of wells.
- Image the plate using a confocal microscope or high-content imager. For quantitative measurements, use a plate reader or flow cytometer.
- Interpretation: A genuine membrane potential signal will show a strong decrease in fluorescence in the depolarized control wells compared to the untreated wells. A lack of signal change suggests the dye is not reporting potential.
Protocol 2: A Counter-Screen for Compound Autofluorescence and Quenching
This orthogonal assay identifies if test compounds are directly interfering with optical detection [14].
- Prepare assay plates with only the culture medium, without any cells.
- Add your library of test compounds to the wells at the same concentrations used in your functional screens.
- Add the dye you use for membrane potential measurement to the wells.
- Read the plates using the same instrument settings (excitation/emission wavelengths, exposure time, etc.) as your primary HCS assay.
- Data Analysis: Statistically analyze the fluorescence intensity data. Compounds that produce outlier signals (extremely high or low) in this cell-free system are likely autofluorescent or quenching, and their data from cellular assays should be flagged for careful review [14].
Protocol 3: Assessing Treatment-Induced Cytotoxicity
This protocol controls for the confounding effects of general cell injury.
- In parallel with your dye assay, run a plate using a vital nuclear stain (e.g., Hoechst 33342) and a cell viability indicator.
- Treat cells with your test compounds as usual.
- Image the plates and use image analysis algorithms to quantify:
- Set a threshold for the minimum number of cells required for robust analysis. Flag or exclude data from wells where cell count falls below this threshold.
The following workflow integrates these protocols into a systematic approach for validating membrane potential data:
The Scientist's Toolkit: Essential Reagents and Materials
Table 2: Key Research Reagents for Membrane Potential Assays
| Reagent / Material | Function / Purpose | Key Considerations |
|---|---|---|
| TMRE / TMRM | Cationic, Nernstian dyes for measuring absolute membrane potential in cells and mitochondria [12]. | Suitable for slow potential changes. Not ideal for fast neuronal signaling. Can be used at very low concentrations (e.g., 5-50 nM) [12]. |
| MitoTracker Green / MitoView Green | Potential-insensitive dyes for staining mitochondrial mass [13]. | More hydrophobic. Retained after depolarization. Signal not a direct measure of function [13]. |
| CCCP / FCCP | Mitochondrial uncouplers used as depolarization controls to validate signal specificity. | Used at ~10 µM. Confirms that a loss of signal is due to loss of potential. |
| Fibronectin Coating | A substrate for cell culture that minimizes non-specific interactions of certain dyes (e.g., Alexa594) with the plate [15]. | Prevents artifactual immobilization of labeled membrane proteins compared to poly-L-lysine coating [15]. |
| Phenol-Red Free / Low-Fluorescence Media | Cell culture medium formulated to reduce background autofluorescence during live-cell imaging [14]. | Mitigates interference from fluorescent media components like riboflavins [14]. |
Frequently Asked Questions (FAQs)
Q1: My treatment seems to cause mitochondrial depolarization, but I'm not sure if the compounds are just quenching the dye. How can I tell? A1: Implement Protocol 2 as a counter-screen. By testing compounds in a cell-free system with your dye, you can directly identify optical interferers. Additionally, using a potential-insensitive dye like MitoTracker Green can help; if the signal is lost with your treatment using TMRE but not with MitoTracker Green, it is more indicative of true depolarization rather than quenching or cell loss.
Q2: Why do my cells sometimes show a bright, concentrated fluorescence after treatment that I wouldn't expect from healthy mitochondria? A2: This can be a sign of treatment-induced cytotoxicity. As cells die and round up, fluorescent probes (especially nucleic acid stains) can become concentrated, saturating the camera and creating a bright, aberrant signal [14]. Always correlate potential measurements with cell viability and morphology assays (Protocol 3).
Q3: Are there specific types of compounds known to cause these artifacts? A3: Yes. Screening libraries can be enriched for compounds with certain undesirable mechanisms that lead to artifacts. These include compounds that are intrinsically autofluorescent, act as redox cyclers, form colloidal aggregates, or are general cellular toxins (e.g., cytoskeletal poisons, mitochondrial toxins, lysosomotropic agents) [14]. These can all produce signals that obscure the true target or phenotype.
Q4: My dye localization looks patchy and uneven. Could this be a technical issue? A4: Yes. Beyond biological reasons, this can be caused by exogenous contaminants like lint, dust, or plastic fragments, which can cause focus blur and image saturation [14]. Ensure your lab environment and reagents are clean. Furthermore, if using labeled proteins, the choice of cell growth substrate (e.g., fibronectin vs. poly-L-lysine) can dramatically affect the apparent mobility and localization of the label [15].
Mitochondrial function is intrinsically linked to its complex internal architecture. The inner mitochondrial membrane (IMM) is extensively folded into cristae, which are dynamic, membrane-bound compartments that protrude into the mitochondrial matrix. These cristae are connected to the inner boundary membrane (IBM) – which runs parallel to the outer membrane – via narrow, tubular structures known as cristae junctions (CJs) [16]. The CJ acts as a fundamental switchboard, controlling the exchange of ions, metabolites, and proteins between the intermembrane space and the intracristal space [17]. This compartmentalization is crucial for establishing spatial H+ gradients and for the efficient function of the oxidative phosphorylation system [17] [16].
The permeability of the CJ is regulated by specific protein complexes. Key among these are:
- MICOS Complex: Located predominantly at CJ sites, it stabilizes contact sites with the outer membrane and is required for CJ formation [16].
- OPA1: Involved in fusion of the inner membrane and in stabilizing the tight curvature of CJs [17].
- F1FO-ATP Synthase: Dimers of this complex are found at the cristae ridges and are essential for shaping cristae membranes [16].
Within the context of your research, understanding this structure is vital. The mitochondrial membrane potential (ΔΨm), typically measured using potentiometric dyes, is not uniform across the entire inner membrane. The CJ functions as a physical barrier that can restrict the diffusion of ions and molecules, leading to sub-mitochondrial compartments with distinct electrochemical properties [17]. During treatments with pharmaceutical compounds, alterations in CJ permeability can cause dye redistribution artifacts, leading to misinterpretation of ΔΨm data.
Troubleshooting Guide: Dye Redistribution & CJ Permeability
Common Experimental Issues & Solutions
| Problem Phenotype | Potential Root Cause | Recommended Solution | Underlying Principle |
|---|---|---|---|
| Inconsistent ΔΨm readings (e.g., rapid signal loss or stabilization) during drug treatment. | Drug-induced opening of the mitochondrial permeability transition pore (mPTP), causing ion and small molecule flux [18] [19]. | Pre-incubate with mPTP inhibitor Cyclosporin A (CsA, 1 µM) for 15-30 minutes prior to assay. | CsA binds to cyclophilin D, inhibiting its interaction with the putative mPTP and preventing pore opening. |
| Unexpected punctate staining patterns with membrane potential dyes (e.g., TMRE, JC-1). | Alterations in CJ permeability, trapping dye in specific cristae compartments [17] [20]. | Combine dye imaging with cristae structure markers (e.g., immunostaining for MICOS subunits). | Validates whether dye pattern changes are coupled to physical cristae remodeling. |
| Overestimation of cell death when using metabolic assays (e.g., MTT) with suspected mitotoxic compounds. | Mitocans impair mitochondrial enzymes, leading to false-positive signals in metabolic assays [21]. | Switch to a differential nuclear staining assay (Hoechst 33342/Propidium Iodide) [21]. | This assay directly counts viable and dead cells based on membrane integrity, independent of metabolism. |
| Failure to detect ΔΨm loss in cells undergoing clear mitochondrial dysfunction. | Compensatory cristae remodeling, maintaining potential in some sub-compartments [17]. | Assess cristae morphology via electron microscopy or super-resolution microscopy. | Provides direct visual evidence of ultrastructural changes that confound bulk ΔΨm measurements. |
Advanced Assessment of CJ Function
To directly evaluate the functional state of the CJ and its regulators, consider these optimized methods:
A. Calcium Retention Capacity (CRC) Assay for mPTP Evaluation The opening of the mPTP is often linked to pathological cristae remodeling and CJ widening. The CRC assay quantitatively measures the susceptibility of mitochondria to Ca2+-induced permeability transition [19].
Protocol Summary:
- Isolate Mitochondria: Prepare mitochondria from your tissue of interest using differential centrifugation.
- Set Up Reaction: Place mitochondrial suspension (0.5-1 mg protein/mL) in assay buffer containing the fluorescent Ca2+ indicator Calcium Green-5N (1 µM).
- Induce Calcium Pulses: Add repetitive, small boluses of CaCl2 (e.g., 10 nmol/mg protein) to the continuously stirred suspension.
- Monitor Fluorescence: Record extramitochondrial Ca2+ via Calcium Green-5N fluorescence (excitation: 506 nm, emission: 532 nm).
- Determine CRC: The CRC is the total amount of Ca2+ accumulated before a massive, rapid Ca2+ release occurs, indicating mPTP opening. A lower CRC indicates higher mPTP opening sensitivity [19].
B. Swelling Assay for Inner Membrane Permeability This classic assay monitors the increase in mitochondrial volume due to osmotic swelling when the inner membrane becomes permeable to small solutes [19].
Protocol Summary:
- Prepare Mitochondria: Use isolated mitochondria (0.2-0.4 mg protein/mL) in a sucrose-based, isotonic buffer.
- Add Inducer: Introduce a mPTP inducer like 200 µM Ca2+ and 1-2 mM phosphate.
- Measure Absorbance: Monitor the decrease in absorbance at 540 nm over time. A faster decrease in absorbance indicates greater swelling and inner membrane permeability.
The Scientist's Toolkit: Essential Reagents & Methods
| Item / Reagent | Primary Function | Application Notes |
|---|---|---|
| Cyclosporin A (CsA) | Inhibits mPTP by binding to Cyclophilin D [18] [19]. | Control for mPTP-specific effects. Use 0.5-1 µM in assays. |
| MitoTracker Green FM | Cell-permeant mitochondria dye, labels regardless of membrane potential [22]. | Useful for visualizing overall mitochondrial network morphology. |
| TMRE / JC-1 | Potentiometric dyes for measuring ΔΨm [22]. | Be aware that redistribution artifacts are most likely with these dyes. |
| Hoechst 33342 | Cell-permeant nuclear dye, stains all nuclei [21]. | Used with PI for viability count; independent of metabolism. |
| Propidium Iodide (PI) | Cell-impermeant nuclear dye, stains only dead cells [21]. | Used with Hoechst for accurate viability assessment with mitocans. |
| CRISPRi/a Platform | For targeted gene knockdown (CRISPRi) or activation (CRISPRa) in primary cells [23]. | Ideal for validating roles of specific genes (e.g., OPA1, MICU1) in CJ regulation. |
| Super-resolution Microscopy (STED, STORM) | Imaging beyond the diffraction limit (~200 nm) to resolve mitochondrial ultrastructure [16]. | Enables visualization of CJ and cristae dynamics in live or fixed cells. |
Frequently Asked Questions (FAQs)
Q1: Why should I be concerned about cristae junctions when my primary readout is overall mitochondrial membrane potential? The ΔΨm is not uniform across the entire inner membrane. The cristae junctions create a diffusion barrier that can lead to the formation of sub-mitochondrial electrochemical microdomains [17] [20]. A treatment that alters CJ permeability can cause a redistribution of ions and dyes within these compartments, giving the appearance of a global ΔΨm change even if the potential in the cristae themselves is preserved or altered differently. This can lead to significant artifacts in data interpretation.
Q2: My drug candidate causes a drop in ΔΨm. How can I determine if this is linked to cristae junction opening? A multi-modal approach is recommended:
- Functional Tests: Perform the Calcium Retention Capacity (CRC) assay. A decreased CRC suggests increased susceptibility to mPTP opening, which is often associated with CJ widening and cristae remodeling [19].
- Morphological Analysis: Use electron microscopy to visualize cristae structure directly. Look for signs of cristae swelling, disorganization, or loss of defined junctions [16].
- Genetic Validation: Employ CRISPRi to knock down CJ-associated proteins like MIC60 or OPA1 in your model system. If your drug's effect is enhanced or mimicked by these knockdowns, it strongly implicates CJ dysfunction in the mechanism [23].
Q3: Are there specific types of compounds that are known to affect cristae junction permeability? Yes, several compound classes can impact CJs:
- Inducers of Oxidative Stress: Can trigger mPTP opening and cristae remodeling.
- Ca2+ Ionophores: Lead to mitochondrial calcium overload, a key activator of mPTP.
- Compounds targeting F1FO-ATP synthase: As this complex is implicated in both cristae shaping and mPTP formation, its inhibition or modification can directly alter CJ stability [18].
- Inhibitors of OPA1 or MICOS function: Compounds that disrupt these complexes will directly impair CJ integrity [17] [16].
Q4: My viability assay (e.g., MTT) shows cell death, but a nuclear stain assay (Hoechst/PI) does not. Which result should I trust? Trust the Hoechst/PI result. Metabolic assays like MTT rely on the activity of mitochondrial enzymes. Mitochondria-targeting compounds (mitocans) can inhibit these enzymes and reduce the MTT signal without immediately killing the cell, leading to a false-positive for cell death [21]. The Hoechst/PI assay directly assesses plasma membrane integrity, a more reliable indicator of necrosis.
Visualizing the Relationship: Cristae Junctions, Dye Redistribution, and Experimental Assessment
The following diagram illustrates the core concepts and recommended experimental pathways for troubleshooting artifacts related to cristae junction permeability.
FAQs: Addressing Common TMRM Redistribution Challenges
Question: During calcium stimulation experiments, my TMRM fluorescence shows a rapid increase, but I expected a decrease due to depolarization. What is happening?
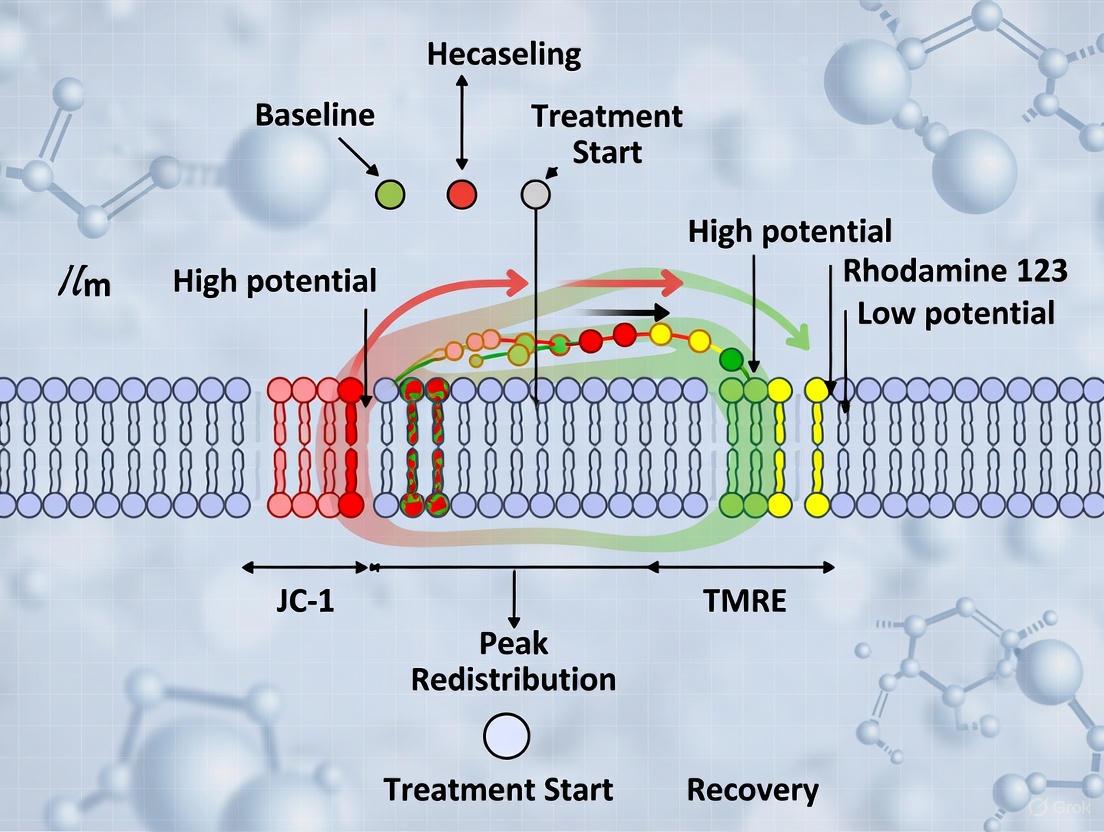
This phenomenon often indicates mitochondrial hyperpolarization, not depolarization. An increase in cytosolic calcium can activate calcium-sensitive dehydrogenases in the mitochondrial matrix, boosting TCA cycle activity and electron transport chain function [6]. This enhanced proton pumping increases ΔΨm, causing additional TMRM accumulation from the cytosol into the mitochondrial matrix, thereby increasing fluorescence intensity [6]. This is a physiologically relevant response, not an artifact.
Question: Why does TMRM fluorescence distribution across my mitochondrial network become heterogeneous after histamine stimulation?
Calcium uptake can trigger localized hyperpolarization of cristae membranes (ΔΨC) relative to the inner boundary membrane (ΔΨIBM) [6]. The cristae junction functions as a barrier, and the proton pumps (Complexes I, III, and IV) are primarily located in the cristae membranes. Calcium-induced activation of metabolism thus hyperpolarizes the cristae first, leading to a spatial gradient of TMRM accumulation that is visible with high-resolution microscopy [6].
Question: I am observing high background fluorescence outside of my cells. How can I reduce this?
Consider using a background suppressor reagent like BackDrop Background Suppressor to reduce extracellular background signal [24]. Furthermore, ensure you are using an appropriate TMRM concentration for your assay mode (low nanomolar for non-quenching mode) and that you wash the cells after loading to remove excess dye from the medium [1] [25].
Question: My untreated control cells are fluorescing, and I'm not seeing a significant difference in my test sample. Is this normal?
Yes, this is expected. Healthy, untreated cells with a polarized mitochondrial membrane potential will accumulate TMRM and fluoresce [24]. The critical factor is the degree of change relative to a proper control. It is essential to include both an untreated control and a positive control treated with a depolarizing agent like FCCP or CCCP to validate your assay and establish a dynamic range [24].
Troubleshooting Guide: TMRM Redistribution Artifacts & Solutions
Table 1: Common TMRM Redistribution Artifacts and Resolution Strategies
| Observed Problem | Potential Cause | Solution | Supporting Controls |
|---|---|---|---|
| Unexpected Fluorescence Increase | Metabolic activation causing hyperpolarization [6]. | Interpret increase as hyperpolarization; confirm with metabolic inhibitors. | Use rotenone (Complex I inhibitor) to block metabolic hyperpolarization [6]. |
| Heterogeneous Intramitochondrial Staining | Spatial membrane potential gradients between cristae and IBM [6]. | Use super-resolution microscopy (e.g., SIM) to validate; employ concentration-dependent distribution analysis [6]. | Analyze TMRM distribution with ∆FWHM or IBM association index methods at different dye concentrations [6]. |
| High Background Fluorescence | Excess dye in extracellular medium [24]. | Include wash steps after loading; use background suppressor reagents [24]. | Image after washes; signal should be predominantly cytosolic and mitochondrial. |
| Poor Response to Stimuli | Dye concentration too high (saturation) [6]; unhealthy cells. | Titrate TMRM to optimal concentration (e.g., 1-50 nM for non-quenching mode) [1] [6]. | Validate system response with FCCP/CCCP (depolarizer) and oligomycin (hyperpolarizer) [25]. |
| Signal Loss Over Time | Photobleaching; dye leakage; genuine depolarization. | Include vehicle control; use photostable imaging buffers; minimize laser exposure. | Compare signal decay in untreated vs. treated cells under identical imaging conditions. |
Quantitative Data & Experimental Protocols
Reference ΔΨm Values Under Physiological Challenges
Table 2: Quantified Mitochondrial Membrane Potential (ΔΨm) in Neurons Under Various Metabolic States
| Metabolic State / Treatment | Absolute ΔΨm (mV) | Change vs. Rest | Key Driver |
|---|---|---|---|
| Resting State | -139 ± 5 [26] | Baseline | Baseline energy demand |
| Sustained PM Depolarization (High K⁺) | -108 ± 4 [26] | ↓ ~31 mV depolarization | Increased ATP demand [26] |
| Metabolic Activation (Ca²⁺) | -158 ± 7 [26] | ↑ ~19 mV hyperpolarization | Ca²⁺-dependent substrate oxidation [26] |
| FCCP (Maximal Depolarization) | ~0 [1] | Complete dissipation | H⁺ ionophore uncoupler |
Detailed Protocol: Measuring ΔΨm Redistribution During Calcium Signaling
This protocol is adapted for investigating TMRM redistribution in response to agonists like histamine that induce calcium release from the endoplasmic reticulum [6].
Materials:
- TMRM: Prepare a stock solution and working concentration in the range of 1.35 to 50 nM for non-quenching mode imaging [6] [25].
- MitoTracker Green FM (MTG): 500 nM stock for morphological reference [6].
- Agonist: e.g., Histamine (concentration as optimized for your cell type).
- Inhibitors: Rotenone (Complex I inhibitor), Antimycin A (Complex III inhibitor).
- Imaging Buffer: Hanks' Balanced Salt Solution (HBSS) with HEPES, pH 7.4.
Procedure:
- Cell Preparation: Plate cells on glass-bottom dishes or chambered cover slips suitable for high-resolution or super-resolution microscopy.
- Dye Loading: Incubate cells with both 500 nM MTG and an optimized concentration of TMRM (e.g., 13.5 nM) for 30 minutes at 37°C in growth media or imaging buffer [6].
- Wash and Equilibration: Wash cells twice with pre-warmed imaging buffer to remove excess dye. Add fresh imaging buffer and allow the dye to equilibrate for an additional 15-20 minutes before imaging.
- Baseline Imaging: Acquire simultaneous dual-channel images (e.g., using SIM) of TMRM and MTG to establish the baseline membrane potential distribution and mitochondrial morphology.
- Stimulation: Add the calcium-mobilizing agonist (e.g., histamine) directly to the imaging chamber and continue time-lapse imaging.
- Inhibition Control (Optional): In parallel experiments, pre-treat cells with rotenone (e.g., 1 µM) for 15-30 minutes before following steps 2-5. This inhibits the electron transport chain and prevents metabolic hyperpolarization, serving as a control [6].
Data Analysis:
- IBM Association Index: This automated method uses the MTG channel to define mitochondrial boundaries. It creates inner (cristae membrane, CM) and outer (inner boundary membrane, IBM) regions and calculates the ratio of TMRM fluorescence in these regions (IBM/CM). A decrease in this index after stimulation indicates relative hyperpolarization of the cristae [6].
- ∆FWHM Method: This semi-automated method analyzes the cross-section intensity profiles of MTG and TMRM. It calculates the difference in the Full Width at Half Maximum (FWHM) of these profiles. A larger ∆FWHM indicates greater TMRM accumulation in the cristae, signifying a higher ΔΨC [6].
Visualizing the Pathways and Workflows
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Investigating TMRM Redistribution
| Reagent / Tool | Function / Purpose | Key Consideration |
|---|---|---|
| TMRM | Cationic, fluorescent potentiometric probe for measuring ΔΨm. | Use low nM (1-50 nM) for non-quenching mode to monitor real-time dynamics; higher concentrations can saturate cristae [6] [25]. |
| MitoTracker Green FM (MTG) | Mitochondrial morphology reference dye; accumulates in IMM independent of ΔΨm after binding. | Use to control for mitochondrial morphology changes and as a spatial reference for TMRM distribution analysis [6]. |
| FCCP / CCCP | Proton ionophores; positive control for complete ΔΨm dissipation. | Validates TMRM response; should cause rapid and complete loss of mitochondrial TMRM signal in non-quenching mode [1] [25]. |
| Oligomycin | ATP synthase inhibitor; causes hyperpolarization by blocking proton reflux. | Used to test the integrity of the electron transport chain and to investigate coupling between ATP demand and ΔΨm [25]. |
| Rotenone / Antimycin A | Inhibitors of Complex I and III, respectively. | Used to inhibit electron transport chain and block metabolic hyperpolarization signals [6]. |
| BackDrop Suppressor | Reduces extracellular background fluorescence. | Improves signal-to-noise ratio by quenching background signal from free dye in solution [24]. |
Best Practices for Accurate ΔΨm Assessment Across Experimental Systems
Optimizing Dye Concentration and Loading Conditions for Different Cell Types
Frequently Asked Questions (FAQs)
Q1: Why is dye concentration so critical in measuring mitochondrial membrane potential (ΔΨm)?
The concentration of potentiometric dyes like TMRM is paramount because it directly affects the measurement's accuracy. At high concentrations (e.g., 40.5-81 nM), the dye saturates the cristae membranes and spills over into the inner boundary membrane (IBM), masking the true potential gradient. At low concentrations (e.g., 1.35-5.4 nM), TMRM preferentially accumulates in the cristae due to their more negative potential, allowing for accurate spatial measurement of the ΔΨm gradient between the cristae and IBM [6].
Q2: What are the signs of dye overloading or underloading in my experiment?
Signs of overloading include a homogenously bright mitochondrial signal without clear structural definition and high background cytosolic fluorescence, indicating saturation. Signs of underloading are a faint, patchy signal that does not adequately resolve the mitochondrial network, potentially leading to an underestimation of ΔΨm [6].
Q3: My ΔΨm measurements are inconsistent between cell lines. What could be the cause?
Different cell types have varying metabolic profiles (e.g., glycolytic vs. oxidative phosphorylation-dependent), which directly influence their basal ΔΨm and dye-loading kinetics [6]. Furthermore, cell types differ in the expression of efflux pumps like P-glycoprotein, which can actively remove dyes from the cell, requiring optimization of loading conditions and potential use of efflux pump inhibitors for consistent results [27].
Q4: How do I validate that a change in dye signal is due to a real ΔΨm shift and not an artifact?
A multi-parameter approach is recommended. Correlate the ΔΨm dye signal with direct functional assays like mitochondrial ATP production [6]. Additionally, use control experiments with known depolarizing agents (e.g., CCCP) and hyperpolarizing agents to establish the dynamic range of the dye in your specific cell model. Inhibition of electron transport chain complexes (e.g., with Rotenone or Antimycin A) can also confirm that signal changes are linked to proton pump activity [6].
Troubleshooting Guide
Table 1: Common Staining Issues and Solutions
| Problem | Potential Cause | Recommended Solution |
|---|---|---|
| High background fluorescence | Dye concentration too high; insufficient washing after loading. | Titrate dye to lowest effective concentration; perform multiple careful washes with dye-free buffer [6]. |
| Weak or no signal | Dye concentration too low; insufficient loading time; inactive dye. | Increase dye concentration incrementally; extend incubation time; test dye viability on a control cell line [6]. |
| Inconsistent results between replicates | Uneven dye loading; variable cell confluency; fluctuations in temperature during loading. | Ensure consistent cell seeding density; pre-warm all buffers; use the same loading protocol for all samples [28]. |
| Signal loss over time | Photobleaching; dye leakage; active dye efflux. | Minimize light exposure during and after staining; use imaging chambers that maintain temperature and CO₂; consider efflux pump inhibitors [27]. |
| Artificially low ΔΨm reading | Dye-induced toxicity; cell death. | Use the lowest possible dye concentration that gives a robust signal; check cell viability with a co-stain like propidium iodide [28]. |
Table 2: Optimized Dye Concentrations for Different Cell Types
This table summarizes research-grade concentration ranges. Exact optimal concentration must be determined empirically for your specific experimental setup.
| Cell Type | Dye | Optimized Concentration Range | Key Considerations |
|---|---|---|---|
| HeLa (Glycolytic) | TMRM | 1.35 - 13.5 nM [6] | Lower concentrations (1.35-5.4 nM) reveal cristae/IBM potential gradients [6]. |
| EA.hy926 (Slightly OXPHOS-dependent) | TMRM | 1.35 - 13.5 nM [6] | Similar to HeLa, concentration dictates spatial resolution of ΔΨm [6]. |
| General Cell Lines (e.g., for apoptosis) | JC-1 | Consult manufacturer's protocol | Ratio of J-aggregates (red) to monomers (green) is concentration-dependent; requires careful titration [28]. |
| General Viability Assessment | Propidium Iodide (PI) | 1-5 µg/mL [27] | Penetrates only cells with compromised membranes. Often used in combination with other dyes [28]. |
Experimental Protocols
Protocol 1: Optimizing TMRM Concentration for Spatial ΔΨm Gradients
This protocol is adapted from super-resolution microscopy studies to analyze mitochondrial sub-structure [6].
Key Reagent Solutions:
- TMRM Stock Solution: Prepare a mM stock in DMSO and store at -20°C.
- MitoTracker Green FM (MTG) Stock Solution: 1 mM in DMSO, store at -20°C.
- Imaging Buffer: Hanks' Balanced Salt Solution (HBSS) or phenol-red free culture medium, pre-warmed to 37°C.
Step-by-Step Methodology:
- Cell Preparation: Seed cells on high-quality glass-bottom dishes and culture until they reach 60-80% confluency.
- Dye Loading:
- Create a series of TMRM working solutions in imaging buffer across a range of concentrations (e.g., 1.35, 2.7, 5.4, 13.5, 40.5 nM).
- Add a constant concentration of MTG (e.g., 500 nM) to each TMRM solution to serve as a mitochondrial morphology reference that is largely insensitive to ΔΨm changes after accumulation.
- Incubate cells in the dye-containing solution for 15-30 minutes at 37°C in the dark.
- Washing: Gently wash the cells three times with pre-warmed, dye-free imaging buffer.
- Image Acquisition: Immediately image the cells using structured illumination microscopy (SIM) or high-resolution confocal microscopy. Acquire images in both the TMRM and MTG channels simultaneously.
- Data Analysis:
- IBM Association Index: Use automated thresholding on the MTG channel to define mitochondrial boundaries. Measure fluorescence intensity in an inner (cristae) and outer (IBM) region. The IBM association index is the ratio of intensities (IBM/CM) [6].
- ∆FWHM Method: Analyze cross-section intensity profiles of MTG and TMRM. Calculate the difference in their Full Width at Half Maximum (FWHM). A larger ∆FWHM indicates greater TMRM accumulation in the cristae [6].
Protocol 2: Multiparametric Flow Cytometry for ΔΨm and Cell Health
This protocol allows for the correlated analysis of ΔΨm with apoptosis, cell cycle, and proliferation from a single sample [28].
Key Reagent Solutions:
- JC-1 Stock Solution: 1 mg/mL in DMSO, store at -20°C.
- Annexin V Binding Buffer: 10 mM HEPES, 140 mM NaCl, 2.5 mM CaCl₂, pH 7.4.
- BrdU (Bromodeoxyuridine): Prepare as per manufacturer's instructions for adding to culture medium.
- CellTrace Violet Stain: Reconstitute in DMSO to make a stock solution.
Step-by-Step Methodology:
- Cell Staining:
- Proliferation Tracing (Optional): Prior to treatment, stain cells with CellTrace Violet according to the manufacturer's protocol to track cell divisions.
- BrdU Incorporation: Add BrdU to the culture medium for the desired pulse period to label S-phase cells.
- ΔΨm Staining: Harvest cells and stain with JC-1 at the pre-optimized concentration (e.g., 2-5 µM) for 20-30 minutes at 37°C in the dark.
- Apoptosis/Necrosis Staining: Wash cells and resuspend in Annexin V Binding Buffer containing fluorescently-labeled Annexin V and Propidium Iodide (PI). Incubate for 15 minutes at room temperature in the dark.
- Fixation and Permeabilization (for BrdU staining): Fix cells with paraformaldehyde, then permeabilize with ice-cold ethanol or a detergent-based permeabilization buffer.
- BrdU Detection: Denature DNA (e.g., with HCl) and stain with a fluorescent anti-BrdU antibody.
- Flow Cytometry Acquisition: Acquire data on a flow cytometer capable of detecting multiple fluorescence channels. Collect at least 10,000 events per sample.
- Data Analysis:
- Analyze JC-1 signal: healthy mitochondria show high red (J-aggregates) and low green (monomers) fluorescence; depolarization causes a shift to high green fluorescence.
- Gate populations based on Annexin V and PI: Viable (Annexin V-/PI-), Early Apoptotic (Annexin V+/PI-), Late Apoptotic/Dead (Annexin V+/PI+), Necrotic (Annexin V-/PI+).
- Analyze CellTrace Violet dilution to determine proliferation rates and BrdU incorporation to identify cell cycle phases.
Signaling Pathways and Experimental Workflows
Diagram: Mitochondrial ΔΨm Regulation & Dye Response
Cellular Signaling Impacting Dye Distribution
Diagram: Experimental Workflow for Dye Optimization
Dye Optimization and Validation Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Mitochondrial Dye Studies
| Reagent | Function/Application | Key Considerations |
|---|---|---|
| TMRM (Tetramethylrhodamine, Methyl Ester) | Potentiometric dye for measuring ΔΨm; used for live-cell imaging. | Reversible dye; concentration is critical to avoid artifacts and toxicity. Excitation/Em: ~548/573 nm [6]. |
| JC-1 | Ratiometric potentiometric dye for flow cytometry and imaging. | Forms J-aggregates (red) in high ΔΨm and monomers (green) in low ΔΨm. The red/green ratio is indicative of ΔΨm [28]. |
| MitoTracker Green FM (MTG) | Mitochondria-selective stain that accumulates regardless of membrane potential. | Useful as a morphological reference stain. Covalently binds to thiol groups, allowing fixation [6]. |
| MitoTracker Red CMXRos | Mitochondria-selective stain that requires membrane potential for accumulation. | Like MTG, but potential-sensitive. Can be used in conjunction with other dyes [29]. |
| Propidium Iodide (PI) | Cell-impermeant dye that stains nucleic acids in dead cells. | Used to assess viability and in apoptosis assays (Annexin V/PI). Excitation/Em: ~535/617 nm [27] [28]. |
| Annexin V (Fluorophore-conjugated) | Binds to phosphatidylserine (PS) exposed on the outer leaflet of apoptotic cells. | Used in combination with PI to distinguish early and late apoptotic cells [28]. |
| Carbonyl Cyanide m-Chlorophenyl Hydrazone (CCCP) | Protonophore that uncouples oxidative phosphorylation, causing ΔΨm collapse. | Essential control for validating ΔΨm dye response and inducing depolarization [6]. |
| Rotenone & Antimycin A | Inhibitors of Electron Transport Chain Complex I and III, respectively. | Used to probe the link between ETC activity, ΔΨm, and dye distribution [6]. |
Experimental Protocols & Methodologies
Protocol for Combined JC-1 and TMRM Staining in Live-Cell Imaging
This protocol is designed for the simultaneous assessment of mitochondrial membrane potential (ΔΨm) using two complementary dyes, providing a ratiometric (JC-1) and a single-emission (TMRM) readout [30] [31].
Materials:
- JC-1 Dye: Prepare a 200 µM stock solution in DMSO [32].
- TMRM Dye: Prepare a 100 µM stock solution in DMSO [31].
- Staining Buffer: Hanks' Balanced Salt Solution (HBSS) or an appropriate cell culture medium without serum [33] [32].
- Control Reagents: Carbonyl cyanide 4-(trifluoromethoxy)phenylhydrazone (FCCP, 10-50 µM) or carbonyl cyanide m-chlorophenyl hydrazone (CCCP, 50 µM) for depolarization [32] [30].
Procedure:
- Cell Preparation: Plate cells on glass-bottom dishes or coverslips suitable for microscopy. Ensure cells are at an appropriate confluence (e.g., 60-80%) at the time of imaging [33] [31].
- Dye Loading:
- Wash cells gently with warm staining buffer.
- For TMRM (non-quenching mode): Incubate cells with 20-200 nM TMRM in staining buffer for 15-30 minutes at 37°C, 5% CO₂ [33] [30] [31].
- For JC-1: Add 2 µM JC-1 directly to the TMRM-containing staining buffer and co-incubate for an additional 15-30 minutes at 37°C, 5% CO₂ [32] [34].
- Washing and Imaging:
- After incubation, gently wash the cells 2-3 times with warm staining buffer to remove excess dye.
- For TMRM, maintain a low concentration of the dye (e.g., 20 nM) in the imaging buffer during the experiment to prevent signal loss due to dye equilibration [33] [31].
- Acquire images using a fluorescence microscope equipped with appropriate filter sets:
Critical Notes:
- Order of Addition: Staining with TMRM first followed by JC-1 can minimize potential interactions.
- Light Sensitivity: Protect stained samples from light to prevent photobleaching and dye-mediated cytotoxicity.
- Controls: Always include a negative control (e.g., cells treated with FCCP/CCCP for 10 minutes prior to imaging) to confirm depolarization-dependent dye redistribution [32] [30].
Protocol for Fixing Cells after Staining with Morphological Markers
This protocol is for experiments requiring subsequent immunostaining or analysis at a later time point, using fixable mitochondrial dyes.
Materials:
- MitoTracker Probes: MitoTracker Red CMXRos, MitoTracker Orange CMTMRos, or MitoTracker Deep Red FM [35].
- Fixative: 3-4% formaldehyde or paraformaldehyde in PBS.
- Permeabilization Buffer: PBS containing 0.1-0.5% Triton X-100.
Procedure:
- Dye Loading: Incubate live cells with 50-200 nM of the desired MitoTracker probe in culture medium for 15-45 minutes at 37°C, 5% CO₂ [35].
- Fixation:
- After staining, wash cells briefly with warm PBS.
- Fix cells with 3-4% formaldehyde solution for 15 minutes at room temperature.
- Wash cells 3 times with PBS to remove residual fixative.
- Permeabilization and Immunostaining (Optional):
- If subsequent immunostaining is required, permeabilize cells with 0.1-0.5% Triton X-100 in PBS for 10-15 minutes.
- Proceed with standard immunostaining protocols using antibodies against morphological markers like TOM20, COX IV, or ATP synthase [36].
Critical Notes:
- Dye Retention: MitoTracker dyes with a chloromethyl moiety (e.g., CMXRos, CMTMRos) are well-retained after aldehyde fixation, unlike JC-1 or TMRM [35].
- MitoTracker Green FM: This dye labels mitochondrial mass independently of ΔΨm but is less well retained after permeabilization [30] [35].
- Validation: Confirm that fixation does not alter the mitochondrial morphology under investigation.
Troubleshooting Guides & FAQs
Frequently Asked Questions (FAQs)
Q1: Why do I observe a loss of TMRM signal upon illumination, and how can I mitigate this? A: The phenomenon you describe is likely photo-induced "flickering" or transient depolarization [30] [31]. TMRM is sensitive to light, and excessive illumination can cause local phototoxicity, leading to reversible ΔΨm loss in individual mitochondria [30]. To mitigate this:
- Reduce the intensity of the excitation light using neutral density filters.
- Shorten the exposure time during image acquisition.
- Increase the time interval between image captures for time-lapse experiments.
- Use a TMRM concentration in the non-quenching mode (5-20 nM) to minimize photodynamic stress [31].
Q2: My JC-1 red/green ratio is low, but my TMRM signal remains bright. What could explain this discrepancy? A: Discrepancies can arise from the fundamental differences in how the dyes operate.
- JC-1 Artifacts: JC-1 aggregation is concentration-dependent and can be influenced by factors other than ΔΨm, such as mitochondrial viscosity and pH [34] [37]. A hidden multidrug resistance (MDR) phenotype in some cell types can actively export JC-1, preventing J-aggregate formation and leading to a low red/green ratio even in healthy cells [38].
- TMRM Specificity: TMRM distribution is more directly governed by the Nernst equation for ΔΨm [31]. To investigate, treat cells with an MDR inhibitor like verapamil or cyclosporin A and repeat the JC-1 staining. If the red signal increases, an MDR artifact is likely [38].
Q3: Can I use JC-1 and TMRM in fixed cells? A: No. Both JC-1 and TMRM are ΔΨm-dependent dyes and will leak out of mitochondria upon loss of membrane potential, which occurs during fixation [34] [35]. Their distribution is not preserved in fixed cells. For fixed-cell experiments, use MitoTracker probes (e.g., CMXRos, CMTMRos), which contain a thiol-reactive chloromethyl group that covalently binds to mitochondrial proteins, allowing retention after fixation [35].
Q4: How do I choose between Mitotracker dyes and TMRM for automated morphology analysis? A: Both can be used, but with caveats. A 2023 study found that TMRM and Mitotracker Red CMXRos are all suited for automated morphology quantification but do not deliver numerically identical results for parameters like area or aspect ratio [30]. Crucially, upon FCCP-induced depolarization, the mitochondrial localization of TMRM is lost most rapidly, followed by the Mitotrackers, while Mitotracker Green FM (a mass marker) is largely unaffected [30]. Therefore, TMRM is best for integrated analysis of ΔΨm and morphology under normal potential conditions, while Mitotracker Green is better for pure morphology assessment regardless of potential [30].
Troubleshooting Common Problems
Table 1: Troubleshooting Dye Redistribution Artifacts
| Problem | Possible Cause | Solution |
|---|---|---|
| High background fluorescence in JC-1/TMRM channels | Incomplete washing of excess dye; dye precipitation. | Increase number of washes; filter dye stock solutions through a 0.2 µm filter before use. |
| Uneven staining between cells in a population | Hidden MDR phenotype; variable dye loading [38]. | Use MDR inhibitors (e.g., verapamil, cyclosporin A); ensure uniform dye incubation conditions and cell confluency. |
| Rapid photobleaching | Excessive light exposure; high dye concentration. | Use lower dye concentrations; reduce exposure time/light intensity; include an oxygen-scavenging system in the buffer. |
| Loss of TMRM signal over time in live imaging | Dye equilibration with the bath solution; genuine ΔΨm depolarization. | Maintain a low concentration of TMRM in the perfusion/imanging buffer [33] [31]; include a positive control (FCCP) to validate the signal loss. |
| Poor correlation between JC-1 ratio and TMRM intensity | JC-1 aggregation artifacts; differential sensitivity to ΔΨm fluctuations [30]. | Validate JC-1 performance with FCCP/CCCP controls; use the dyes as complementary, not redundant, measures. |
Data Presentation: Quantitative Comparisons
Table 2: Comparison of Key Mitochondrial Dyes for Multi-Parameter Assays
| Dye | Primary Readout | ΔΨm Sensitivity | Fixable? | Compatible Morphology Analysis | Key Advantages | Key Limitations / Artifacts |
|---|---|---|---|---|---|---|
| JC-1 | Ratiometric (Red/Green) | High | No [34] | Possible in live cells [36] | Internal rationing minimizes artifacts from mitochondrial density [32] [34] | Prone to MDR export; aggregation is non-linear and concentration-dependent [38] [34] |
| TMRM | Intensity-based | High | No [35] | Yes (in live cells) [30] [31] | Reversible binding, suitable for kinetics; reliable for ΔΨm measurements [30] [31] | Sensitive to photo-induced flickering; requires careful concentration control [33] [30] |
| MitoTracker Red CMXRos | Intensity-based | High | Yes [35] | Yes (post-fixation) [30] [35] | Retained after fixation, enabling immunostaining [35] | Potential toxicity; covalent binding may not reflect rapid ΔΨm changes [30] [35] |
| MitoTracker Green FM | Intensity-based | Low (Mass marker) | Partial [35] | Yes (post-fixation, pre-permeabilization) [30] | Labels mitochondrial mass independently of ΔΨm [30] [35] | Not a reliable indicator of ΔΨm; signal lost upon permeabilization [30] [35] |
| CellLight Mitochondria-GFP/RFP | Fluorescent Protein | No (Genetic tag) | Yes | Yes (pre- and post-fixation) [35] | Excellent for morphology; not dependent on ΔΨm; can be expressed long-term [35] | Requires transfection/transduction; does not report on functional state (ΔΨm) [35] |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Tools for Mitochondrial Morphofunctional Analysis
| Item | Function | Example Usage |
|---|---|---|
| JC-1 Assay Kit (e.g., MitoProbe) | Provides optimized dye and controls for ratiometric ΔΨm assessment by flow cytometry or imaging. | Apoptosis studies, high-throughput screening of compounds affecting ΔΨm [32] [34]. |
| TMRM | lipophilic cationic dye for sensitive, kinetic measurement of ΔΨm in live cells. | Real-time monitoring of ΔΨm fluctuations (e.g., "flickering") and spatial heterogeneity [33] [30] [31]. |
| MitoTracker Probes (CMXRos, Deep Red) | Fixable dyes for correlating ΔΨm-sensitive staining with immunocytochemistry. | Co-staining with antibodies (e.g., TOM20) to link potential and morphology in fixed samples [36] [35]. |
| FCCP / CCCP | Protonophores that uncouple the electron transport chain, collapsing ΔΨm. | Essential negative control for validating ΔΨm-dependent dye localization [32] [30] [31]. |
| Oligomycin | ATP synthase inhibitor, causes hyperpolarization by blocking proton re-entry. | Tool to test the coupling state of mitochondria and the response of dyes to hyperpolarization [33] [31]. |
| MDR Inhibitors (e.g., Verapamil) | Inhibit multidrug resistance pumps that can export dyes like JC-1. | Confirming suspected dye export artifacts in cell lines with high MDR activity [38]. |
Experimental Workflow and Signaling Pathways
Experimental Workflow for Multi-Parameter Analysis
Mitochondrial Signaling in Apoptosis and Dye Redistribution
Technical Support Center
Frequently Asked Questions (FAQs)
FAQ 1: What are the most common artifacts when using potentiometric dyes for mitochondrial membrane potential (ΔΨm) measurement in drug treatment studies? The most common artifacts arise from dye concentration, non-protonic ion fluxes, and incorrect interpretation of fluorescence changes.
- Dye Concentration Artifacts: Using high concentrations of dyes like TMRM can saturate the mitochondria, preventing the detection of genuine hyperpolarization and causing dye redistribution to the inner boundary membrane (IBM), which masks the true potential gradient between the IBM and cristae membrane (CM) [6]. For TMRM/TMRE, use the lowest possible concentration (e.g., 1–30 nM for non-quenching mode) [1].
- Non-Protonic Charge Artifacts: Cationic ΔΨm dyes respond to the total electrical gradient, not exclusively the proton gradient. For example, a massive efflux of calcium ions (Ca²⁺) from the mitochondrial matrix can cause a hyperpolarization of ΔΨm, even while the proton gradient (ΔpHm) is decreasing. Relying solely on ΔΨm dyes can thus lead to incorrect conclusions about the proton motive force and ATP synthesis capacity [1].
- Interpretation Artifacts: With JC-1, the formation of J-aggregates is sensitive to factors beyond ΔΨm, such as mitochondrial size/volume (S/V) ratios and the presence of oxidants like H₂O₂. If S/V ratios differ between cell types or treatments, it could falsely imply differences in ΔΨm [1].
FAQ 2: How can I validate that my observed ΔΨm dye redistribution is due to a true change in membrane potential and not an artifact? A robust validation requires a series of controlled pharmacological challenges.
- Induce Depolarization: Apply a known uncoupler like FCCP or carbonyl cyanide-p-trifluoromethoxyphenylhydrazone. A genuine ΔΨm-dependent dye will show a rapid and reversible redistribution (e.g., a loss of JC-1 J-aggregates, or a decrease in TMRM intensity) [1] [8].
- Induce Hyperpolarization: Application of the ATP synthase inhibitor oligomycin, which inhibits proton flow back into the matrix, should cause a measurable hyperpolarization and corresponding dye response (e.g., increased JC-1 J-aggregation) [1].
- Parallel Assays: Correlate your findings with direct measurements of other parameters. For instance, use a pH-sensitive dye like SNARF-1 to measure mitochondrial pH and confirm whether ΔpHm changes in parallel with ΔΨm [1].
FAQ 3: My super-resolution images show heterogeneous TMRM distribution within a single mitochondrion. Is this a real potential gradient or a staining artifact? This is likely a real biological phenomenon. Super-resolution microscopy has revealed that the inner mitochondrial membrane (IMM) is compartmentalized into the inner boundary membrane (IBM) and the cristae membrane (CM), which can maintain distinct electrical potentials (ΔΨIBM and ΔΨC) [6]. The cristae junction (CJ) acts as a barrier, separating these compartments. The distribution of TMRM between them is concentration-dependent [6]:
- At low concentrations (e.g., 1.35–5.4 nM), TMRM preferentially accumulates in the CM, which typically has a higher (more negative) potential [6].
- At high concentrations (e.g., 40.5–81 nM), the CM becomes saturated, and TMRM staining increases relatively in the IBM, making the potential gradient less visible [6]. Therefore, using low dye concentrations is critical for visualizing this spatial gradient.
FAQ 4: We need to fix cells after staining for subsequent immunocytochemistry. Which ΔΨm dyes are compatible with fixation? Most cationic potentiometric dyes (e.g., Rhodamine 123, TMRM, JC-1) are washed out upon fixation because the loss of membrane potential prevents their retention [35]. The MitoTracker Orange and Red probes (e.g., CMXRos, CM-H2XRos) are designed to overcome this limitation. They contain a thiol-reactive chloromethyl moiety that covalently binds to mitochondrial proteins, allowing the staining pattern to be preserved after aldehyde fixation [35]. Note that MitoTracker Green FM accumulation is less dependent on membrane potential and may serve as a marker for mitochondrial mass in some fixed-cell applications [35].
Troubleshooting Guides
Problem: Inconsistent JC-1 Ratios After Drug Treatment
Potential Causes and Solutions:
- Cause 1: Incorrect Dye Loading Concentration or Time. JC-1 behavior is highly sensitive to concentration. Aqueous solutions above 0.1 µM can lead to aggregation unrelated to potential [8].
- Solution: Titrate the JC-1 concentration for your specific cell type. Standard protocols often use 2–5 µM with a 15–30 minute incubation at 37°C [8]. Always include a control with an uncoupler like FCCP or CCCP to validate the signal.
- Cause 2: Drug Interference with Dye. The test compound may directly interact with the JC-1 dye or alter mitochondrial volume independently of potential.
- Solution: Perform a control experiment where cells are loaded with JC-1, then treated with the drug and immediately analyzed. A very rapid change in the ratio may indicate direct chemical interference. Consider validating key findings with an alternative dye like TMRM.
- Cause 3: Inadequate Washing or Background Fluorescence.
- Solution: Ensure proper washing after dye loading to remove excess extracellular JC-1 monomers, which contribute to green background fluorescence [8].
Problem: Excessive Photobleaching and Phototoxicity in Live-Cell STED Imaging
Potential Causes and Solutions:
- Cause 1: High Laser Intensities. STED microscopy inherently uses high-intensity depletion lasers, which can damage dyes and living cells [39].
- Cause 2: Suboptimal Dye Selection. Some conventional dyes are not photostable enough for STED.
- Solution: Employ STED-optimized dyes. For example, the pkMitoRed dye has been successfully used for STED imaging of the inner mitochondrial membrane [40].
- Cause 3: Prolonged Imaging.
- Solution: Minimize the number of frames and the field of view. Use faster scanning modes if available and consider alternative super-resolution techniques like structured illumination microscopy (SIM) for live-cell imaging, as it generally has lower photodamage, though with a more modest resolution gain [39].
Experimental Protocols
Protocol 1: Validating Drug-Induced ΔΨm Changes Using JC-1 and Flow Cytometry
This protocol is optimized for detecting shifts in the red/green fluorescence ratio indicative of apoptosis or other stress responses [8].
1. Reagent Setup:
- Prepare JC-1 working solution: Dissolve JC-1 in DMSO to make a 1–2 mM stock. Further dilute in cell culture medium to a final working concentration of 2–5 µM.
- Positive control: Prepare 50 µM carbonyl cyanide-m-chlorophenyl hydrazone (CCCP) in DMSO.
- Assay Buffer: 1x Phosphate-Buffered Saline (PBS) or your preferred cell culture medium.
2. Staining Procedure:
- Harvest and wash cells, then resuspend at a density of 0.5–1 x 10⁶ cells/mL.
- Add the JC-1 working solution to the cell suspension. Incubate for 15–30 minutes at 37°C in the dark.
- Centrifuge cells at low speed and gently resuspend in pre-warmed assay buffer.
- (Optional) Drug Treatment: Cells can be treated with the drug of interest before staining and then analyzed with JC-1 present, or treated after staining and washing.
3. Flow Cytometry Data Acquisition:
- Use 488 nm excitation.
- Collect green monomer fluorescence with a 530/30 nm bandpass filter (FITC channel).
- Collect red J-aggregate fluorescence with a 585/42 nm bandpass filter (PE channel).
- Acquire data for at least 10,000 events per sample.
4. Data Analysis:
- Gate on viable cells based on forward and side scatter.
- Create a bivariate dot plot of PE (J-aggregates, red) vs. FITC (monomers, green) fluorescence.
- Calculate the ratio of median fluorescence intensity in the PE channel to the FITC channel. A decrease in this ratio indicates mitochondrial depolarization.
Protocol 2: Analyzing Spatial Membrane Potential Gradients with SIM and TMRM
This protocol details a method for visualizing the membrane potential difference between the cristae membrane (CM) and inner boundary membrane (IBM) using super-resolution structured illumination microscopy (SIM) [6].
1. Cell Staining:
- Culture cells (e.g., HeLa or EA.hy926) on high-performance glass-bottom dishes.
- Dual-Labeling: Co-stain cells with 500 nM MitoTracker Green FM (MTG) and a low concentration of TMRM (e.g., 1.35–13.5 nM) in culture medium for 30 minutes at 37°C [6].
- Note: MTG acts as a potential-insensitive reference for mitochondrial morphology, while TMRM reports on potential.
2. SIM Imaging:
- Perform simultaneous dual-channel SIM imaging.
- Settings: Maintain identical laser power, exposure time, and SIM reconstruction parameters for all samples within an experiment.
3. Image Analysis:
- IBM Association Index Method (Automated) [6]:
- Use the MTG channel to define the mitochondrial boundary using an automated threshold (e.g., Otsu).
- Generate two masks by shrinking and widening this boundary to create IBM and CM regions.
- Measure the mean TMRM fluorescence intensity in both regions.
- Calculate the IBM Association Index = TMRM IntensityIBM / TMRM IntensityCM. A decrease in this index indicates relative hyperpolarization of the CM.
- ΔFWHM Method (Semi-Automated) [6]:
- Draw a line scan perpendicular to the long axis of a mitochondrion on both the MTG and TMRM channels.
- Plot the fluorescence intensity profile for each channel.
- Measure the Full Width at Half Maximum (FWHM) for each profile.
- Calculate ΔFWHM = FWHMMTG - FWHMTMRM. A higher ΔFWHM indicates greater TMRM accumulation in the cristae (CM hyperpolarization).
Table 1: Concentration-Dependent Behavior of Common ΔΨm Dyes
| Dye | Recommended Concentration | Mode/Readout | Key Considerations and Artifacts |
|---|---|---|---|
| TMRM / TMRE [1] [6] | 1–30 nM (non-quenching)>50–100 nM (quenching) | Intensity-based; higher potential = more accumulation | Low mitochondrial binding. High concentrations saturate cristae, masking CM-IBM gradients [6]. Fast equilibration. |
| JC-1 [1] [8] | 2–5 µM | Ratiometric (590 nm Aggregates / 529 nm Monomers) | Sensitive to concentration, S/V ratios, and oxidants like H₂O₂. Aggregate form not solely dependent on ΔΨm [1]. |
| Rhodamine 123 [1] | ~1–10 µM (quenching) | Intensity-based (quenching/unquenching) | Slowly permeant; depolarization causes unquenching (increased fluorescence). Less ETC inhibition than TMRE [1]. |
| DiOC₆(3) [1] | <1 nM | Intensity-based | Requires very low conc. to monitor ΔΨm rather than plasma membrane potential (Δψp). Toxic at higher concentrations [1]. |
Table 2: Super-Resolution Microscopy Techniques for Mitochondrial Analysis
| Technique | Practical Resolution | Suitability for Live-Cell ΔΨm Imaging | Key Limitations |
|---|---|---|---|
| STED [40] [39] | ~50 nm (lateral) | Variable (lower for large fields); can be used with pkMitoRed and TMRM [40] | High photodamage and photobleaching. Tuneable resolution vs. damage trade-off [39]. |
| SIM [6] [39] | 90–130 nm (lateral) | High (for 2D-SIM); used with TMRM/MTG [6] | High susceptibility to reconstruction artifacts. Lower resolution gain than STED [39]. |
| Pixel Reassignment (e.g., AiryScan) [39] | 140–180 nm (lateral) | Intermediate to High | Moderate resolution improvement. Lower susceptibility to artifacts than SIM [39]. |
Research Reagent Solutions
Table 3: Essential Reagents for Mitochondrial Membrane Potential and Super-Resolution Imaging
| Item | Function | Example Product(s) |
|---|---|---|
| Potentiometric Dyes | Accumulate in mitochondria in a membrane potential-dependent manner to report on ΔΨm. | TMRM, TMRE, JC-1 (MitoProbe JC-1 Assay Kit) [1] [8], Rhodamine 123 [1], MitoTracker Red CMXRos (fixable) [35] |
| Morphology Reference Dyes | Label mitochondrial structure independently of membrane potential, serving as a spatial reference. | MitoTracker Green FM [6], CellLight Mitochondria-GFP/RFP (BacMam) [35] |
| Pharmacological Controls | Used to validate dye response by artificially modulating ΔΨm. | FCCP (uncoupler, depolarizes) [1], Oligomycin (ATP synthase inhibitor, hyperpolarizes) [1], CCCP (uncoupler) [8] |
| STED-Optimized Dyes | Fluorescent dyes with high photostability and brightness suitable for the high-intensity lasers in STED microscopy. | pkMitoRed [40] |
| Mitochondrial Superoxide Indicator | Detects mitochondrial superoxide production, a key parameter in cell health and stress. | MitoSOX Red [35] |
Signaling Pathways and Experimental Workflows
Diagram 1: Mechanism of Dye Redistribution Artifacts in Mitochondrial Sub-Compartments
Diagram 2: Experimental Workflow for Validating Drug Effects on ΔΨm
Flow Cytometry Protocols for High-Throughput ΔΨm Screening
Mitochondrial membrane potential (ΔΨm) serves as a crucial indicator of mitochondrial health and cellular viability, with its dissipation being a hallmark early event in apoptosis [8]. In high-throughput screening (HTS) environments for drug discovery, flow cytometry has emerged as a powerful technology for quantifying ΔΨm across thousands of samples. The integration of fluorescent potentiometric dyes like JC-1 enables rapid assessment of mitochondrial function in response to pharmacological treatments [28] [41]. However, researchers frequently encounter dye redistribution artifacts that can compromise data interpretation, particularly when screening compounds that affect mitochondrial function or membrane integrity. This technical support center addresses the specific challenges of ΔΨm screening, providing detailed protocols, troubleshooting guidance, and reagent specifications to ensure robust, reproducible results in high-throughput drug development workflows.
Experimental Protocols for ΔΨm Assessment
JC-1 Staining Protocol for Flow Cytometry
The JC-1 dye represents a ratiometric probe that exhibits potential-dependent accumulation in mitochondria, forming red fluorescent "J-aggregates" at hyperpolarized potentials and green fluorescent monomers at depolarized potentials [8]. This protocol is optimized for high-throughput screening applications.
Sample Preparation:
- Plate cells at a density of 1.0 × 10^5 cells per well in a 96-well or 384-well plate format suitable for HTS [42] [41].
- Apply experimental treatments following specific research objectives (e.g., compound libraries for drug discovery).
- For hypoxia/reoxygenation models mimicking ischemia/reperfusion injury: incubate cells in an airtight hypoxic jar with oxygen-scavenging catalyst for 18 hours, followed by 2 hours reoxygenation [41].
Staining Procedure:
- Prepare JC-1 working solution by diluting JC-1 stock solution (200×) in pre-warmed (37°C) staining buffer to a final concentration of 2-5 μM [41] [8].
- After treatments, collect cells using mild trypsinization (0.25% trypsin-EDTA for approximately 1 minute) and neutralize with complete medium.
- Pellet cells by centrifugation at 500 × g for 3 minutes at room temperature and carefully aspirate supernatant.
- Resuspend cell pellet in 0.5 mL complete medium and add 0.5 mL JC-1 working solution.
- Incubate cells for 15-30 minutes at 37°C in the dark to prevent fluorochrome photobleaching [41] [8].
- Pellet cells again by centrifugation at 500 × g for 3 minutes at 4°C.
- Wash cells twice with 2 mL ice-cold staining buffer to remove unincorporated dye.
- Resuspend final cell pellet in 300 μL ice-cold staining buffer and analyze by flow cytometry within 30 minutes [41].
Flow Cytometry Acquisition:
- Configure flow cytometer with 488 nm excitation laser.
- Use FITC (530/30 nm) and PE (575/26 nm) emission filters to detect JC-1 monomer and J-aggregate signals, respectively [8].
- Analyze a minimum of 10,000 events per sample at low flow rate settings to ensure high-quality data [28] [43].
- Include appropriate controls: unstained cells, CCCP-treated cells (20 μM for 1 hour) as depolarization control, and valinomycin-treated cells for K+-induced depolarization [41] [8].
Multiparametric Assessment of Cell Death and Proliferation
For comprehensive screening, ΔΨm measurement can be integrated with additional cellular parameters to distinguish primary mitochondrial effects from secondary consequences of cell death or proliferation changes [28].
Annexin V/PI Staining for Apoptosis:
- Combine JC-1 staining with Annexin V/PI staining to correlate ΔΨm dissipation with apoptosis progression.
- After JC-1 staining, resuspend cells in Annexin V binding buffer containing fluorescently conjugated Annexin V and propidium iodide (PI).
- Incubate for 15 minutes at room temperature in the dark before flow cytometric analysis [28].
BrdU/PI Staining for Cell Cycle Analysis:
- Prior to JC-1 staining, incubate cells with bromodeoxyuridine (BrdU) for 30-60 minutes to label S-phase cells.
- After JC-1 staining, fix and permeabilize cells, then denature DNA to expose BrdU epitopes.
- Stain with anti-BrdU antibody and PI to assess cell cycle distribution and proliferation status [28].
Troubleshooting Guides and FAQs
Common Technical Issues and Solutions
Table 1: Troubleshooting Flow Cytometry Issues in ΔΨm Screening
| Problem | Possible Causes | Recommended Solutions |
|---|---|---|
| Weak or no fluorescence signal | Low mitochondrial membrane potential; Inadequate dye loading; Incorrect instrument settings | Optimize dye concentration (2-5 μM) and incubation time (15-30 min); Verify laser alignment and PMT voltages; Include CCCP-treated positive control [43] [44] |
| High background or non-specific staining | Excessive dye concentration; Presence of dead cells; Inadequate washing | Titrate JC-1 concentration; Include viability dye to gate out dead cells; Increase washing steps after staining [43] [44] |
| Poor resolution of cell cycle phases | High flow rate; Insufficient PI staining; Suboptimal cell preparation | Reduce flow rate to minimum setting; Ensure adequate PI concentration and RNase treatment; Harvest cells during exponential growth phase [43] |
| Abnormal scatter profiles | Cell clumping; Bacterial contamination; Incorrect instrument settings | Filter cells through mesh before analysis; Practice sterile technique; Calibrate instrument with reference beads [44] |
| Loss of epitope signal | Over-fixation; Methanol permeabilization issues | Limit fixation time to <15 minutes; Chill cells on ice before adding ice-cold methanol dropwise [43] |
Frequently Asked Questions
Q1: Why does JC-1 staining sometimes show inconsistent red/green ratios between experiments?
A1: JC-1 ratio inconsistencies often stem from technical variations including:
- Dye concentration variations: JC-1 concentration critically affects J-aggregate formation. Consistently prepare fresh working solutions for each experiment [8].
- Temperature fluctuations: Maintain consistent 37°C incubation during staining, as temperature affects dye uptake and aggregation [8].
- Cellular confluence: Harvest cells at consistent confluence (70-80% recommended) as overcrowding affects mitochondrial function [41].
- Instrument calibration: Regularly calibrate flow cytometer with fluorescent beads and include within-experiment controls for normalization [43].
Q2: How can we distinguish genuine ΔΨm depolarization from dye redistribution artifacts caused by test compounds?
A2: Several approaches can help discriminate true depolarization from artifacts:
- Implement a plate-based positive control using CCCP (20 μM) or valinomycin to establish maximum depolarization reference [41] [8].
- Use alternative ΔΨm probes with different chemical structures (e.g., TMRM, TMRE) to confirm findings [45].
- Correlate ΔΨm measurements with other mitochondrial parameters such as reactive oxygen species production using DHR or DCFDA dyes [28].
- Perform kinetic measurements to distinguish rapid dye redistribution (artifact) from progressive depolarization (biological effect) [45].
Q3: What specific considerations are needed for high-throughput ΔΨm screening compared to conventional flow cytometry?
A3: HTS implementations require particular optimizations:
- Miniaturization: Adapt protocols to 384-well or 1536-well formats with reduced volumes (20-50 μL) while maintaining cell density (1×10^5 cells/mL) [42] [46].
- Automated fluidics: Implement plate-based samplers with integrated washing steps to minimize operator intervention [42] [46].
- Reduced staining time: Optimize incubation periods (can often be reduced to 15 minutes for JC-1) without compromising signal quality [42].
- Integrated analysis: Utilize automated gating algorithms and ratio calculations to process large datasets without manual intervention [42] [46].
Q4: How does fixation affect JC-1 staining and what alternatives exist for fixed samples?
A4: JC-1 is incompatible with standard fixation methods as it alters the dye's potential-dependent distribution. The MitoProbe JC-1 Assay Kit is explicitly designed for live cells only [8]. For experiments requiring fixation, consider these alternatives:
- Use fixable potentiometric dyes like TMRM or TMRE followed by mild formaldehyde fixation (1-2% for <15 minutes) [45] [44].
- Implement mitochondrial-targeted fluorescent proteins (e.g., mito-GFP) that tolerate fixation.
- Perform live-cell analyses first, then fix for subsequent intracellular staining of other targets [43].
Research Reagent Solutions
Table 2: Essential Reagents for High-Throughput ΔΨm Screening
| Reagent | Function | Application Notes |
|---|---|---|
| JC-1 Dye | Ratiometric ΔΨm indicator | Forms red J-aggregates at high potentials, green monomers at low potentials; Use at 2-5 μM for 15-30 min; Incompatible with fixation [8] |
| TMRM/TMRE | Single-wavelength ΔΨm probes | Suitable for kinetic studies; Lower toxicity than JC-1; Can be used in quenching or non-quenching mode; Compatible with mild fixation [45] |
| CCCP | Mitochondrial uncoupler | Positive control for depolarization; Use at 20 μM for 1 hour; Prepare fresh in DMSO for each experiment [41] [8] |
| Annexin V conjugates | Apoptosis detection | Binds externalized phosphatidylserine; Use with calcium-containing binding buffer; Can be combined with JC-1 staining [28] [8] |
| Propidium Iodide | Viability and DNA staining | Distinguishes live/dead cells; Penetrates compromised membranes; Use with RNase for cell cycle analysis [28] [43] |
| BrdU | Proliferation marker | Incorporated during DNA synthesis; Requires DNA denaturation and antibody detection; Correlates ΔΨm with cell cycle status [28] |
| MitoTracker Green | Mitochondrial mass indicator | Potential-independent staining; Useful for normalizing ΔΨm to mitochondrial content [45] |
| CellTrace Violet | Cell proliferation tracer | CFSE-like dye that dilutes with each cell division; Enables correlation of ΔΨm with proliferation capacity [28] |
Workflow and Signaling Pathway Diagrams
Diagram 1: High-Throughput ΔΨm Screening Workflow. This diagram illustrates the sequential steps for JC-1-based mitochondrial membrane potential screening in multiwell plate formats, including optional extensions for multiparametric analysis.
Diagram 2: JC-1 Response to Mitochondrial States and Potential Artifacts. This diagram illustrates the relationship between mitochondrial membrane potential, JC-1 fluorescence behavior, and potential sources of dye redistribution artifacts that can complicate data interpretation.
Frequently Asked Questions (FAQs)
FAQ 1: Why do I observe an increase in my ΔΨm dye signal, but a simultaneous decrease in ATP production? This seems counterintuitive.
This is a classic sign that your experimental treatment may be causing mitochondrial uncoupling or inhibiting the ATP synthase.
- Underlying Principle: The mitochondrial membrane potential (ΔΨm) is the electrical component of the proton motive force (PMF), which is used to drive ATP synthesis via ATP synthase [1] [47]. Normally, ATP production consumes the PMF, leading to a slight, regulated decrease in ΔΨm. If the ATP synthase (Complex V) is blocked (e.g., by oligomycin), protons cannot flow back into the matrix, leading to a hyperpolarization of ΔΨm and a complete halt of ATP production [1] [2].
- Troubleshooting Steps:
- Confirm ATP Synthase Function: Include a control with oligomycin (typically 1-10 µM). An expected result is a sharp decrease in ATP levels coupled with an increase in ΔΨm dye signal (hyperpolarization). If this doesn't happen, your model system or assay may be compromised.
- Check for Uncoupler Effects: Use the uncoupler FCCP (e.g., 0.5-2 µM) to collapse the ΔΨm. This should cause a rapid and complete loss of the potentiometric dye signal and abolish ATP production. A partial response suggests incomplete uncoupling or issues with dye equilibration.
- Validate Dye Specificity: Ensure that the observed hyperpolarization is not an artifact caused by changes in non-protonic charges, such as calcium fluxes, which can also affect cationic dye distribution [1].
FAQ 2: My ΔΨm measurements with different dyes (e.g., TMRM vs. a MitoTracker) give conflicting results. Which one should I trust?
The choice of dye is critical and depends on your experimental goal, as each probe has unique properties and potential artifacts [1] [30].
- Key Consideration: Fixability vs. Dynamic Measurement. MitoTracker dyes (e.g., CMXRos) are thiol-reactive and can be fixed for later imaging, but their retention is less dependent on acute changes in ΔΨm after fixation [48] [30]. In contrast, dyes like TMRM and TMRE are highly sensitive to acute, real-time changes in ΔΨm but are lost upon fixation or depolarization [1] [30].
- Troubleshooting Steps:
- Define Your Need: For live-cell, kinetic studies of ΔΨm, use potentiometric dyes like TMRM/TMRE or Rhod123 at low, non-quenching concentrations [1]. For endpoint analysis that requires fixation, a MitoTracker may be necessary.
- Be Aware of Performance Differences: Studies show that while multiple dyes can report on morphology, their sensitivity to ΔΨm depolarization varies significantly, with the order of sensitivity being TMRM >> MitoTracker Red CMXRos > MitoTracker Green FM [30]. MitoTracker Green FM can accumulate in a potential-independent manner in some cells, making it more a marker of mass than potential [48].
- Standardize and Validate: Always use the same dye and loading protocol within a single study. Include standard controls (FCCP, oligomycin) to confirm the dye is responding to ΔΨm changes as expected in your specific cell model.
FAQ 3: I am detecting high ROS in my cells, but my ΔΨm signal appears low/depolarized. Aren't high ROS levels usually linked to a hyperpolarized membrane?
This is a common misconception. The relationship between ΔΨm and ROS is complex and state-dependent [49] [50].
- Underlying Principle: While a highly hyperpolarized ΔΨm can increase electron leak and ROS production, a significantly depolarized ΔΨm is often associated with severe mitochondrial dysfunction. In this state, damaged electron transport chain (ETC) complexes can "leak" electrons at a high rate before the Q-site, leading to excessive ROS generation despite an overall low ΔΨm [49] [50].
- Troubleshooting Steps:
- Correlate with Metabolic State: Assess the bioenergetic state of your mitochondria. Use substrates and inhibitors to probe specific ETC sites. For instance, ROS can originate from Complex I (sites IF and IQ) or Complex III (site IIIQo), and the effect of ΔΨm on each site differs [50].
- Measure Multiple Parameters Simultaneously: If possible, use multi-parameter imaging or parallel assays to directly correlate ΔΨm and ROS (e.g., MitoSOX Red) in the same cell population over time [51]. This can reveal transient phenomena where ROS spikes precede depolarization.
- Check for Compensatory Mechanisms: The cell may attempt to mitigate ROS stress by activating uncoupling proteins (UCPs), which induce a mild proton leak to depolarize ΔΨm and reduce ROS production [47] [50]. The observed depolarization could be a consequence of this protective response.
Troubleshooting Guide: Dye Redistribution Artifacts
This guide addresses specific artifacts that can lead to misinterpretation of ΔΨm dye data.
| Observed Problem | Potential Artifact & Cause | Recommended Solution |
|---|---|---|
| High, Saturated Signal | Dye overloading and concentration-dependent quenching. High dye concentrations (>50-100 nM for TMRM) can cause aggregation and quenching, masking true ΔΨm changes [1]. | Use the lowest possible dye concentration that gives a clear signal. Perform a concentration gradient test to establish a non-quenching mode (~1-30 nM for TMRM) [1]. |
| Unexpected Signal Loss | Dye redistribution due to plasma membrane potential (Δψp) changes. Cationic ΔΨm dyes are also sensitive to the plasma membrane potential [1]. | Use a positive control (e.g., high K+ buffer) to assess the contribution of Δψp. Ensure experimental treatments do not directly affect plasma membrane permeability or ion gradients. |
| No Response to FCCP/Oligomycin | 1. Incorrect dye loading conditions.2. Loss of dye equilibration.3. Inactive reagents. | Follow standardized protocols for dye loading and incubation [52]. For TMRM in non-quenching mode, it is often best to image with the dye present in the bath [1]. Verify reagent activity and stock solution integrity. |
| Heterogeneous Signal Within Single Mitochondria | 1. True biological heterogeneity (cristae vs. inner boundary membrane).2. Imaging artifact or poor resolution. | Use super-resolution techniques (e.g., SIM, STED) to resolve sub-mitochondrial compartments [6]. For standard confocal microscopy, ensure optimal image acquisition settings and deconvolution if necessary. |
Standardized Experimental Protocols
Protocol: Concurrent Measurement of ΔΨm and ATP
This protocol outlines a method for correlating changes in ΔΨm with ATP production in live cells.
Principle: Tetramethylrhodamine methyl ester (TMRM) is used as a ratiometric ΔΨm probe, while a FRET-based ATP biosensor (e.g., ATeam) allows simultaneous quantification of mitochondrial ATP levels [6].
Workflow Diagram: Correlating ΔΨm with ATP
Materials:
- Cells: Primary neurons or appropriate cell line [52].
- Dye: TMRM (Tetramethylrhodamine methyl ester) [51] [6] [30].
- ATP Sensor: Cells transfected with a genetically-encoded mitochondrial ATP sensor (e.g., ATeam) [6].
- Assay Buffer: Physiological salt solution (e.g., Hanks' Balanced Salt Solution, HBSS) supplemented with glucose.
- Controls: Oligomycin (ATP synthase inhibitor, 1-10 µM), FCCP (uncoupler, 0.5-2 µM).
- Equipment: Live-cell fluorescence microscope with environmental control (37°C, 5% CO₂), appropriate filter sets for TMRM (Ex/Em ~548/573 nm) and the FRET sensor.
Procedure:
- Cell Preparation: Seed cells expressing the mitochondrial ATP sensor onto glass-bottom dishes or coverslips. Culture for the desired time (e.g., primary neurons for 7-14 days in vitro) [52].
- Dye Loading: On the day of the experiment, load cells with a low concentration of TMRM (e.g., 10-30 nM) in pre-warmed assay buffer for 30 minutes at 37°C in the dark [1] [52].
- Wash and Equilibration: Gently wash cells twice with pre-warmed assay buffer. For experiments in non-quenching mode, maintain a low concentration of TMRM (e.g., 5-10 nM) in the bath during imaging to prevent dye loss [1].
- Image Acquisition: Mount the dish on the microscope. Select fields of view with healthy, well-transfected cells. Acquire a 2-5 minute baseline of both TMRM and FRET signals.
- Treatment: Add your experimental compound or pharmacological controls (e.g., oligomycin, FCCP) without moving the field of view.
- Time-lapse Imaging: Continue imaging for the required duration (e.g., 30-60 minutes), capturing both channels at short intervals (e.g., 30-60 seconds).
- Data Analysis:
- ΔΨm: Calculate the ratio of TMRM fluorescence in mitochondria versus the cytosol. A decrease in ratio indicates depolarization; an increase indicates hyperpolarization.
- ATP: Calculate the FRET ratio of the ATP biosensor according to its specific instructions. A decrease indicates falling ATP levels.
- Plot both ratios over time to visualize their correlation.
Protocol: Correlative Analysis of ΔΨm and Mitochondrial ROS
This protocol describes how to measure ΔΨm and mitochondrial superoxide production in the same sample, either sequentially or, with a compatible setup, simultaneously.
Principle: TMRM is used to measure ΔΨm, and MitoSOX Red, a mitochondrial-targeted superoxide indicator, is used to detect ROS [51] [48].
Workflow Diagram: Correlating ΔΨm with ROS
Materials:
- Dyes: TMRM and MitoSOX Red mitochondrial superoxide indicator [51] [48].
- Controls: Antimycin A (Complex III inhibitor, induces ROS, 1-10 µM), Rotenone (Complex I inhibitor, induces ROS, 100-500 nM), FCCP (uncoupler, reduces ROS by mild depolarization, 0.5-2 µM) [49] [50].
- Equipment: Fluorescence microscope with filter sets for TMRM (Ex/Em ~548/573 nm) and MitoSOX Red (Ex/Em ~510/580 nm). Note: MitoSOX oxidation products can also be detected with a ~405 nm excitation [48].
Procedure:
- Cell Preparation: Seed cells as described in Protocol 3.1.
- ROS Dye Loading: Incubate cells with MitoSOX Red (1-5 µM in pre-warmed buffer) for 15 minutes at 37°C in the dark.
- Wash: Wash cells gently twice with warm buffer.
- ΔΨm Dye Loading: Subsequently, incubate cells with TMRM (10-30 nM) for 30 minutes at 37°C in the dark.
- Wash and Equilibration: Wash cells twice. If imaging in non-quenching mode, maintain a low TMRM concentration in the bath.
- Baseline Imaging: Acquire baseline images for both MitoSOX and TMRM. Due to potential spectral overlap, sequential imaging is recommended. If the microscope has good spectral separation, simultaneous imaging may be possible.
- Treatment and Monitoring: Apply the experimental treatment or controls. Continue time-lapse imaging to monitor changes in both parameters.
- Data Analysis:
- Quantify the mean fluorescence intensity of MitoSOX Red within mitochondrial regions defined by the TMRM signal. An increase indicates elevated superoxide production.
- Quantify TMRM fluorescence (ratio or intensity) as in Protocol 3.1.
- Correlate the time courses of ROS production and ΔΨm changes.
Research Reagent Solutions
This table provides a concise guide to key reagents used in the protocols above for correlating ΔΨm with functional assays.
| Reagent / Dye | Primary Function | Key Considerations & Experimental Tip |
|---|---|---|
| TMRM / TMRE | Cationic, potentiometric ΔΨm dye. Accumulates in the mitochondrial matrix in a potential-dependent manner [1] [51]. | Best for kinetic studies. Use low concentrations (1-30 nM) for non-quenching mode; image with dye in bath for pre-treatment paradigms. Lowest toxicity and mitochondrial binding among common dyes [1] [30]. |
| Rhodamine 123 | Cationic, potentiometric ΔΨm dye [1]. | Best for fast, acute quenching-mode studies. Often used at higher concentrations (1-10 µM) where depolarization causes fluorescence "unquenching" and a transient signal increase [1]. |
| JC-1 | Ratiometric ΔΨm dye. Forms green fluorescent monomers at low potentials and red fluorescent J-aggregates at high potentials [1]. | Best for flow cytometry or yes/no discrimination of polarization state (e.g., apoptosis). Very sensitive to dye concentration and loading conditions. Aggregate form can be sensitive to factors other than ΔΨm [1]. |
| MitoTracker Red CMXRos | Fixable mitochondrion-selective dye. Accumulates in active mitochondria and is retained after aldehyde fixation due to its thiol-reactive chloromethyl group [48] [30]. | Use for endpoint assays requiring immunostaining. Less sensitive to acute ΔΨm changes than TMRM. Not ideal for quantitative kinetic measurements of ΔΨm [30]. |
| MitoSOX Red | Fluorogenic dye selectively targeted to mitochondria that is oxidized by superoxide, producing bright red fluorescence [51] [48]. | Specific for mitochondrial superoxide. Can be used in live cells. Verify specificity with antioxidants (e.g., PEG-SOD). Be aware of potential photo-oxidation artifacts [48]. |
| Oligomycin | Inhibitor of F1Fo-ATP synthase (Complex V) [1] [2]. | Control for hyperpolarization. Use to test if ΔΨm is coupled to ATP synthesis. Expected result: ΔΨm increases, ATP production plummets. |
| FCCP | Protonophore uncoupler. Shuttles protons across the IMM, collapsing the proton motive force and ΔΨm [1] [52]. | Positive control for complete depolarization. Use to validate ΔΨm dye response. Titrate for optimal concentration (typically 0.5-2 µM). |
| Antimycin A | Inhibitor of Complex III (ubiquinol-cytochrome c reductase) [50]. | Control for ROS generation. Inhibiting Complex III at the Qo site promotes superoxide production from the ETC, useful for validating MitoSOX response [49] [50]. |
Signaling Pathway Diagrams
Diagram: Integrated Signaling of ΔΨm, ATP, and ROS
Identifying and Resolving Common Redistribution Artifacts in Treatment Studies
Recognizing Artifact Patterns During Uncoupler and Inhibitor Treatments
In the study of mitochondrial function, particularly during treatments with uncouplers and inhibitors, researchers frequently rely on fluorescent dyes to measure key parameters like the mitochondrial membrane potential (ΔΨm). A cornerstone of a broader thesis on this subject is that these measurements are susceptible to significant artifacts related to dye redistribution and other confounding factors. These artifacts can lead to the misinterpretation of a compound's mechanism of action, falsely attributing changes in fluorescence to shifts in ΔΨm when they are, in fact, caused by unrelated processes. This technical guide outlines common artifact patterns and provides methodologies for their identification and mitigation.
Troubleshooting Guides
Guide 1: Addressing Altered Fluorescence Independent of ΔΨm
Presenting Problem: A treatment with a novel compound causes a rapid decrease in the fluorescence intensity of a cationic ΔΨm-sensitive dye (e.g., TMRM, JC-1). The result is interpreted as mitochondrial depolarization, but the negative control (a known, potent uncoupler like FCCP or CCCP) does not produce a similar effect.
Artifact Pattern: The fluorescence loss is likely not due to a change in ΔΨm but to other factors. A primary suspect is the activation of multidrug resistance (MDR) transporters, such as P-glycoprotein (P-gp), in the plasma membrane. These ATP-dependent efflux pumps can recognize and actively export fluorescent dyes from the cell, preventing their accumulation in the mitochondria regardless of the actual ΔΨm [53].
Diagnostic and Resolution Workflow:
Detailed Protocol:
- Cell Model: Use P-gp-positive cells (e.g., L1210 R or T variants) and their P-gp-negative counterparts (e.g., L1210 S) for a controlled experiment [53].
- Staining: Load cells with your chosen ΔΨm dye (e.g., JC-1 at 2 μM for 15-30 minutes at 37°C).
- Inhibition: Apply a non-competitive, high-affinity P-gp inhibitor like Tariquidar (e.g., 1-2 hour pre-incubation). Note that weaker inhibitors like cyclosporine A or verapamil may not fully restore fluorescence [53].
- Measurement: Apply your test compound and measure fluorescence via flow cytometry or microscopy. The restoration of a normal fluorescence signal in the presence of Tariquidar confirms MDR-mediated artifact.
Guide 2: Validating True Uncoupling vs. Other Mechanisms
Presenting Problem: A compound increases cellular oxygen consumption rate (OCR) and decreases ΔΨm dye fluorescence, mimicking an uncoupler. However, the effect on ATP synthesis is inconsistent or the dose-response is atypical.
Artifact Pattern: The compound may not be a canonical protonophore but could be acting through a different mechanism that secondarily dissipates ΔΨm. Examples include ionophores that transport other ions (e.g., Ca²⁺), inhibitors of the electron transport chain (ETC), or inducers of the mitochondrial permeability transition pore (mPTP) [2] [54]. True uncouplers stimulate respiration and collapse ΔΨm while inhibiting ATP synthesis because they short-circuit the proton circuit.
Diagnostic and Resolution Workflow:
Detailed Protocol:
- Integrated Bioenergetic Assay: Perform a mitochondrial stress test to measure OCR and extracellular acidification rate (ECAR) in real-time using a Seahorse XF Analyzer or similar instrument [54].
- ATP Measurement: In parallel experiments, use a luciferase-based ATP assay to quantify cellular ATP levels after treatment with the compound. A true uncoupler will consume, not produce, ATP via reverse operation of the ATP synthase if ΔΨm is collapsed [2].
- Mechanism Testing:
- Ionophore Activity: Test if the effect is dependent on the presence of specific ions (e.g., Ca²⁺, K⁺).
- ETC Inhibition: Assess the compound's effect on respiration in permeabilized cells supplied with specific ETC complex substrates (e.g., pyruvate/malate for Complex I, succinate for Complex II) [54].
- mPTP Induction: Use cyclosporine A, an mPTP inhibitor, to see if it blocks the compound's effects.
Frequently Asked Questions (FAQs)
FAQ 1: Why does my positive control uncoupler (FCCP/CCCP) not cause a complete loss of JC-1 J-aggregate (red) fluorescence in my cell line?
- Answer: This is a common observation and does not necessarily indicate a failed experiment. The completeness of the ΔΨm collapse can vary. More importantly, JC-1 is a substrate for MDR pumps like P-gp. If your cell line highly expresses these transporters, they may be actively excluding the JC-1 dye, creating a baseline signal that appears "depolarized." Using a P-gp inhibitor like Tariquidar can help clarify this. Always include a validated uncoupler in your experimental design and interpret the magnitude of change relative to this control [53] [8].
FAQ 2: When measuring ΔΨm, what is a more reliable indicator of mitochondrial function?
- Answer: While ΔΨm is an important parameter, it is a measurement of potential, not flux. It reflects the proton motive force but not necessarily the rate of ATP production. A more sensitive and comprehensive assessment of mitochondrial oxidative phosphorylation is the direct measurement of oxygen consumption flux (OCR). It is recommended to use OCR measurements as a primary functional readout and to use ΔΨm as a complementary parameter to understand the mechanistic basis of any observed changes in flux [55].
FAQ 3: My lab uses both TMRM and Mitotracker Red (CMXRos). Can I use them interchangeably for morphofunctional analysis?
- Answer: Not exactly. While both dyes can be used to visualize mitochondria, their behavior is different. TMRM distribution is highly dynamic and strictly dependent on ΔΨm. Mitotracker Red CMXRos contains a thiol-reactive chloromethyl group that covalently binds to mitochondrial proteins, trapping it in the organelle for longer periods. This makes Mitotrackers better for fixed-cell imaging but less sensitive to acute, reversible changes in ΔΨm. For integrated analysis of ΔΨm and morphology in live cells, TMRM is generally superior, provided the ΔΨm is not severely and permanently depolarized [30].
Research Reagent Solutions
Table 1: Key Reagents for Investigating Mitochondrial Uncoupling and Avoiding Artifacts.
| Reagent / Assay | Function / Application | Key Considerations & Pitfalls |
|---|---|---|
| JC-1 Dye | Ratiometric ΔΨm indicator. Green monomers at low potential, red J-aggregates at high potential. Ideal for flow cytometry and imaging [8]. | Substrate for MDR pumps (e.g., P-gp). Use with inhibitor (Tariquidar) in resistant cell lines. Not fixable [53]. |
| TMRM / TMRE | Cationic, fluorescent ΔΨm probes. Used in quantitative, non-quenching mode for live-cell imaging. Signal decreases with depolarization [55] [30]. | Distribution is affected by both plasma membrane and mitochondrial potentials. Requires careful calibration for quantitative measurements [55]. |
| FCCP / CCCP | Proton ionophores (positive control uncouplers). Collapse ΔΨm and stimulate maximal OCR by equalizing proton gradient across the inner mitochondrial membrane [56] [57]. | Standard for validating assay sensitivity. High concentrations can be toxic. Prepare fresh stock solutions in DMSO or ethanol. |
| Oligomycin | ATP synthase (Complex V) inhibitor. Used in mitochondrial stress tests to probe ATP-linked respiration and proton leak [56] [54]. | Arrests ATP production, causing a drop in OCR and a transient hyperpolarization of ΔΨm. |
| Tariquidar | High-affinity, non-competitive P-glycoprotein (MDR1) inhibitor [53]. | Essential control for dye efflux artifacts, particularly with JC-1 and rhodamine-based dyes in cancer or transfected cell lines. |
| Seahorse XF Analyzer | Instrument platform for real-time measurement of OCR and ECAR in live cells. The gold standard for integrated bioenergetic flux analysis [54]. | Provides a functional profile (e.g., basal respiration, ATP production, spare capacity) that is more informative than ΔΨm alone. |
Experimental Protocols
Detailed Protocol: JC-1 Assay with MDR Inhibition for Flow Cytometry
This protocol is optimized to control for P-gp-mediated dye efflux artifacts [53] [8].
Materials:
- MitoProbe JC-1 Assay Kit (or equivalent: JC-1 dye, DMSO, CCCP)
- P-gp inhibitor: Tariquidar
- Cell suspension (e.g., Jurkat, L1210, or your cell line of interest)
- Flow cytometer with 488 nm excitation and filters for FITC (530 nm) and PE (585 nm)
Procedure:
- Pre-treatment (if testing MDR inhibition): Divide cells into two aliquots. Pre-incubate one aliquot with 1 µM Tariquidar for 60 minutes at 37°C. The other aliquot serves as the untreated control.
- Staining: Prepare a 2 µM working solution of JC-1 in pre-warmed culture medium. Resuspend all cell pellets in the JC-1 working solution. Incubate for 30-45 minutes at 37°C, 5% CO₂, protected from light.
- Washing and Resuspension: Centrifuge cells at low speed, aspirate the supernatant, and gently resuspend the cell pellet in warm PBS.
- Controls:
- Untreated, stained cells: Baseline ΔΨm.
- Cells + 10-50 µM CCCP (from kit): Fully depolarized control. Add CCCP 5-10 minutes before the end of the staining incubation.
- Cells + Tariquidar + test compound: To test for MDR artifact.
- Flow Cytometry Analysis:
- Analyze cells immediately using 488 nm excitation.
- Collect green fluorescence (JC-1 monomer) through a 530/30 nm bandpass filter (FITC channel).
- Collect red fluorescence (JC-1 aggregate) through a 585/42 nm bandpass filter (PE channel).
- Use a dot plot of Red (PE) vs. Green (FITC) fluorescence to gate the population.
- Calculate the Red/Green fluorescence ratio for each condition. A decrease in this ratio indicates mitochondrial depolarization, but only if the MDR control (Tariquidar) does not alter the ratio in the test compound group.
Detailed Protocol: Oxygen Consumption Rate (OCR) Measurement in Permeabilized Cells
This protocol allows for the direct investigation of effects on the electron transport chain and phosphorylation system [56] [54].
Materials:
- Oxytherm system (Hansatech) or Seahorse XF Analyzer (Agilent)
- Cells of interest (e.g., U2OS, NIH 3T3)
- Permeabilization agent: Digitonin (3-5 µM)
- Mitochondrial substrates: 5 mM Pyruvate + 2 mM Malate + 10 mM Glutamate (for Complex I); 10 mM Succinate (for Complex II, with rotenone)
- ADP (1 mM)
- Inhibitors: Oligomycin A (5 µM), test compound (e.g., DIDS), CCCP (50 µM), Sodium Azide (1 mM)
Procedure:
- Cell Preparation: Harvest and count cells. For the Oxytherm system, use 1 x 10⁶ cells per chamber in MAS buffer. For Seahorse, seed cells in a microplate the day before.
- Permeabilization: Add digitonin (final conc. 3 µM) to the cell suspension and incubate for 5-10 minutes on ice. Gently agitate. Optimal concentration should be determined empirically.
- Respirometry:
- Add permeabilized cells to the measurement chamber at 37°C.
- State 2: Add Complex I substrates (Pyruvate, Malate, Glutamate) to initiate basal, non-phosphorylating respiration.
- State 3: Add ADP (1 mM) to stimulate maximal phosphorylating respiration.
- Inhibition/Test: Add Oligomycin A (5 µM) to inhibit ATP synthase and measure State 4o (non-phosphorylating respiration due to proton leak).
- Uncoupling/Test: Add the test compound (e.g., 500 µM DIDS) or the standard uncoupler CCCP (50 µM) to induce maximal uncoupled respiration.
- Inhibition: Add sodium azide (1 mM) to inhibit Complex IV and measure residual non-mitochondrial respiration.
- Data Analysis: Calculate OCR (nmol O₂/min/10⁶ cells) at each state. Compare the effect of the test compound to CCCP. A true uncoupler will mimic CCCP by stimulating OCR in State 3 or after oligomycin.
Mitigating CCCP-Induced Depolarization Artifacts in JC-1 Assays
In mitochondrial membrane potential research, JC-1 dye redistribution artifacts present significant challenges for data interpretation, particularly during pharmacological interventions. The carbonil cyanide m-chlorophenyl hydrazone (CCCP) is a potent mitochondrial uncoupler that collapses the proton gradient by transporting protons across the inner mitochondrial membrane, thereby inducing depolarization [58]. However, CCCP-induced depolarization can produce technical artifacts that compromise experimental validity if not properly controlled. These artifacts stem from the fundamental property of JC-1 as a cationic dye that accumulates in mitochondria in a membrane potential-dependent manner, existing either as green-fluorescent monomers (at low membrane potentials) or red-fluorescent "J-aggregates" (at high membrane potentials) [8]. When CCCP collapses the mitochondrial membrane potential, the resulting dye redistribution can be misinterpreted without appropriate normalization and control experiments, potentially leading to false conclusions about mitochondrial function during treatment research. This technical guide addresses the most common artifact scenarios and provides validated troubleshooting approaches to ensure data reliability.
Troubleshooting Guides & FAQs
Fundamental Mechanisms & Artifact Identification
Q1: What are the primary mechanisms behind CCCP-induced JC-1 redistribution artifacts? CCCP acts as a protonophore that dissipates the hydrogen ion gradient across the inner mitochondrial membrane, effectively collapsing both the chemical (ΔpH) and electrical (ΔΨm) components of the proton motive force [1] [58]. This collapse triggers JC-1 redistribution artifacts through two primary mechanisms:
Potential-Dependent Relocalization: The driving force for JC-1 accumulation in the mitochondrial matrix is diminished, causing the dye to diffuse into the cytoplasm [8]. This leads to the dissociation of J-aggregates into monomers, resulting in a characteristic fluorescence shift from red to green that may not accurately reflect the kinetics of depolarization if imaging parameters are not optimized [59].
Spectral Interference from Compounds: As documented in studies using the GSK-3β inhibitor SB216763, some treatment compounds exhibit autofluorescence that overlaps with the green emission spectrum of JC-1 monomers (approximately 529 nm) [59]. This creates a false depolarization signal despite maintained membrane potential, requiring spectral deconvolution for accurate interpretation.
Q2: How can researchers distinguish true mitochondrial depolarization from CCCP-induced JC-1 artifacts? Valid discrimination requires a multi-parameter approach:
Control Experiments: Include both CCCP-treated positive controls (for complete depolarization) and oligomycin-treated negative controls (for hyperpolarization) in every experiment to establish baseline fluorescence ratios [58].
Spectral Validation: Conduct control experiments without JC-1 loading to identify compound-related autofluorescence that might overlap with JC-1 emission spectra [59].
Kinetic Monitoring: Artifacts often manifest as instantaneous fluorescence changes, while true biological depolarization typically follows more gradual kinetics that can be tracked in real-time using live-cell imaging systems [58].
Morphological Correlation: Correlate fluorescence changes with mitochondrial morphology assessments, as artifacts rarely correlate with structural changes in mitochondrial networks [6].
Technical Optimization & Experimental Design
Q3: What are the optimal JC-1 staining conditions to minimize redistribution artifacts? Proper staining protocol optimization is crucial for reducing technical artifacts:
Table 1: JC-1 Staining Optimization Parameters
| Parameter | Recommended Condition | Effect on Artifact Reduction |
|---|---|---|
| JC-1 Concentration | 2-10 μM for flow cytometry; 5 μg/mL for imaging [8] [59] | Prevents over-saturation and non-specific aggregation |
| Incubation Time | 15-30 minutes at 37°C [8] [60] | Ensures complete dye loading without cellular toxicity |
| Incubation Conditions | In cell culture incubator (37°C, 5% CO₂) [8] | Maintains physiological conditions during dye loading |
| Post-staining Washes | 1-2 gentle washes with pre-warmed buffer [8] | Removes excess dye without inducing depolarization |
| Time to Analysis | Within 30 minutes of staining completion [60] | Prevents dye redistribution due to prolonged storage |
Q4: How should adherent cells be prepared for JC-1 assays to minimize artifacts? For adherent cells, artifacts frequently arise from inappropriate cell preparation:
- Digestion Timing: Digest cells before JC-1 staining rather than after, as trypsinization following staining can artificially depolarize mitochondria [60].
- Alternative Approach: For microscopy-based assays, maintain cells in adherent state throughout staining and imaging to avoid digestion-induced stress.
- Handling Considerations: Avoid excessive centrifugation force (>300 × g) and prolonged processing times that can compromise membrane integrity and induce artifactual depolarization.
Data Interpretation & Validation Strategies
Q5: What validation experiments confirm that observed fluorescence changes reflect true mitochondrial depolarization? A robust validation framework incorporates multiple complementary approaches:
Pharmacological Validation: Use established mitochondrial modulators beyond CCCP, including oligomycin A (ATP synthase inhibitor inducing hyperpolarization) and rotenone/antimycin A (electron transport chain inhibitors inducing depolarization) to verify JC-1 response specificity [58].
Alternative Probe Correlation: Validate key findings with structurally distinct potentiometric dyes such as TMRM or TMRE that have different chemical properties and potential artifact profiles [1] [58].
Multiplexed Apoptosis Assessment: Combine JC-1 staining with annexin V/propidium iodide apoptosis assays [59] [8] or caspase activation markers to contextualize membrane potential changes within established cell death pathways.
Spectral Deconvolution: For compounds with suspected autofluorescence, implement mathematical spectral deconvolution based on reference spectra and least-squares minimization algorithms to isolate true JC-1 signals from interfering fluorescence [59].
Experimental Protocols for Artifact Mitigation
Spectral Deconvolution Protocol for Compound Interference
For experiments involving treatment compounds with potential autofluorescence, this protocol adapts the methodology successfully employed for SB216763 interference correction [59]:
Reference Spectrum Collection:
- Acquire fluorescence emission spectra (500-650 nm range with 470 nm excitation) of:
- Untreated cells without JC-1 (background autofluorescence)
- Cells with treatment compound alone without JC-1 (compound autofluorescence)
- Cells with JC-1 alone (reference JC-1 spectrum)
- Use identical instrument settings across all acquisitions
- Acquire fluorescence emission spectra (500-650 nm range with 470 nm excitation) of:
Mathematical Deconvolution:
- Apply algorithm for least-squares minimization to experimental measurements
- Utilize fluorophore reference spectra to produce corresponding unmixed spectra
- Calculate corrected green/red fluorescence ratios (540/595 nm) after deconvolution
Validation:
- Confirm deconvolution efficacy using control conditions with known membrane potential status
- Verify linearity of deconvoluted signals across a range of membrane potentials induced by CCCP and oligomycin
Quantitative JC-1 Assay with CCCP Controls
This optimized protocol minimizes artifacts while providing quantitative assessment of mitochondrial membrane potential:
Table 2: JC-1 Assay Reagent Preparation
| Reagent | Composition | Preparation Notes |
|---|---|---|
| JC-1 Stock Solution | 1-5 mg/mL in DMSO [8] [61] | Aliquot and store at -20°C protected from light; avoid freeze-thaw cycles |
| JC-1 Working Solution | 2-10 μM in serum-free culture medium [8] | Prepare fresh for each experiment; do not store working solution |
| CCCP Control Solution | 10-50 μM in DMSO or culture medium [58] [60] | Titrate concentration for specific cell type to ensure complete depolarization |
| Oligomycin A Control | 1-10 μM in DMSO [58] | Use as hyperpolarization control to establish dynamic range |
| Assay Buffer | Serum-free culture medium or PBS | Pre-warm to 37°C before use |
Procedure:
- Cell Preparation: Harvest cells gently using non-enzymatic dissociation methods when possible. Count and adjust cell density to 0.5-1 × 10⁶ cells/mL in serum-free medium.
- Staining:
- Add JC-1 working solution to cell suspension (1:1 volume ratio)
- Incubate 15-30 minutes at 37°C in the dark
- Centrifuge gently (300 × g for 5 minutes) and remove supernatant
- Resuspend in pre-warmed assay buffer
- Control Treatments:
- For CCCP-treated controls: Add CCCP to final concentration (typically 10-50 μM) during the last 5-10 minutes of JC-1 staining
- Include vehicle control (DMSO at same dilution as CCCP)
- Analysis:
- For flow cytometry: Use 488 nm excitation with 530/30 nm (green) and 585/42 nm (red) emission filters [8]
- For fluorescence microscopy: Use standard FITC/TRITC filter sets with consistent exposure times across conditions [8]
- Calculate red/green fluorescence ratio for each condition normalized to vehicle control
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for JC-1 Artifact Mitigation
| Reagent/Tool | Function in Artifact Mitigation | Application Notes |
|---|---|---|
| JC-1 Dye | Ratiometric mitochondrial membrane potential indicator | Preferred over single-wavelength dyes due to internal calibration capability [8] [61] |
| TMRM/TMRE | Low-binding alternative potentiometric dyes | Validate key findings; less prone to aggregation artifacts [1] [58] |
| CCCP | Protonophore for depolarization controls | Titrate concentration for complete depolarization without cellular toxicity [58] [60] |
| Oligomycin A | ATP synthase inhibitor for hyperpolarization controls | Establishes assay dynamic range and validates dye responsiveness [58] |
| FCCP | Alternative mitochondrial uncoupler | Can substitute for CCCP with potentially different artifact profile [58] |
| MitoTracker Green FM | Mitochondrial mass reference dye | Helps normalize for mitochondrial content independent of membrane potential [6] |
| Annexin V/Propidium Iodide | Apoptosis detection reagents | Contextualizes membrane potential changes within cell death pathways [59] [8] |
| Spectral Imaging System | Fluorescence detection with spectral resolution | Enables deconvolution of overlapping emission spectra [59] |
Conceptual Diagrams
Artifact Mitigation Workflow
Troubleshooting Guide: Common Artifacts in Mitochondrial Membrane Potential Assays
This guide addresses frequent challenges researchers face when using fluorescent dyes to measure mitochondrial membrane potential (ΔΨm) in the presence of experimental drugs or treatments. Proper interpretation of these assays is crucial as they are key indicators of cell health and pivotal in apoptosis studies [1] [8].
Table 1: Troubleshooting Common ΔΨm Dye Artifacts
| Problem | Possible Causes | Recommended Solutions | Key Controls & Validation |
|---|---|---|---|
| Lack of Assay Window [62] | Incorrect instrument setup (filters, gain); incorrect dye concentration; complete ΔΨm collapse. | Verify instrument configuration and filter settings; optimize dye concentration using a titration; validate with a positive control (e.g., FCCP). | Include FCCP/CCCP (uncoupler) and Oligomycin (ATP synthase inhibitor) controls in every experiment [1]. |
| Inconsistent Results Between Labs [62] | Differences in compound stock solution preparation; cell culture conditions; dye loading protocols. | Standardize compound solubilization and storage; use identical cell passages and seeding densities; document detailed dye loading procedures. | Use a standardized internal control compound in all experiments. |
| Unexpected Hyperpolarization [1] | Drug-induced changes in intracellular ion homeostasis (e.g., Ca²⁺ release); inhibition of ATP synthase. | Measure mitochondrial Ca²⁺ levels; assess overall cellular ATP production; use parallel assays to confirm results. | Perform a dose-response curve for the test compound; use ion chelators (e.g., BAPTA-AM) to test for Ca²⁺-dependent effects. |
| No Signal or Weak Staining | Dye degradation; loss of ΔΨm; incorrect dye loading parameters; drug-induced dye efflux or sequestration. | Prepare fresh dye stocks; confirm cell viability; optimize loading time and temperature; use a quenching mode protocol [1]. | Test dye performance on untreated, healthy cells; use a viability stain to confirm plasma membrane integrity. |
| High Non-Specific Background | Drug or dye partitioning into cellular membranes [63] [64]; overloading of dye. | Titrate dye to the lowest effective concentration; switch to a less lipophilic dye (e.g., TMRM over DiOC₆(3)); include wash steps after loading [1]. | Run a no-dye control to measure cellular autofluorescence; use a no-drug control to establish a baseline. |
Frequently Asked Questions (FAQs)
Q1: My drug treatment causes an unexpected increase in ΔΨm (hyperpolarization). Does this mean the drug is improving mitochondrial health? Not necessarily. Hyperpolarization can be a sign of stress and may be unrelated to the proton gradient. For example, it can be caused by the release of non-protonic ions like Ca²⁺ from mitochondrial or ER stores. To confirm, you should perform parallel measurements of mitochondrial pH and calcium levels [1].
Q2: Why do I get different IC₅₀ values for the same compound in different cell types or between biochemical and cell-based assays? Differences in free drug concentration are a primary cause. Lipophilic compounds can partition into cellular membranes, a major mechanism of drug sequestration. This membrane partitioning reduces the free concentration available to interact with the intended target, leading to an underprediction of potency (higher apparent IC₅₀). This effect is more pronounced in systems with higher membrane content [63] [64].
Q3: For JC-1 staining, my red/green ratio is low even in control cells. What could be wrong? A low ratio indicates a lack of J-aggregate formation, which can be due to several factors:
- Dye Concentration: JC-1 is highly sensitive to concentration. Use a concentration that allows for aggregate formation in healthy, polarized mitochondria (typically >0.1 µM) [8].
- Loading Time: The dye may require longer loading times than commonly used to reach equilibrium and form aggregates [1].
- Instrument Setup: Ensure you are using the correct optical filters (e.g., FITC/TRITC or FITC/PE) to properly separate the monomer (green) and aggregate (red) signals [8].
Q4: When should I use TMRM in non-quenching mode versus quenching mode?
- Non-quenching Mode (~1-30 nM): Best for measuring pre-existing ΔΨm and for slow, acute studies. The dye is used at low concentrations and fluorescence intensity is directly proportional to ΔΨm. The dye can be kept in the bath during imaging [1].
- Quenching Mode (>50-100 nM): Used to monitor acute changes after dye loading and washout. At high concentrations, the dye aggregates and quenches itself. Depolarization causes dye release and a transient increase in fluorescence (unquenching). This mode is best for fast, dynamic studies [1].
Experimental Protocols for Validating Dye Behavior
Protocol 1: Establishing a Baseline and Controls for ΔΨm Measurement with TMRM
This protocol is essential for confirming that your TMRM signal accurately reflects changes in ΔΨm [1] [30].
Research Reagent Solutions:
| Reagent | Function | Key Consideration |
|---|---|---|
| TMRM | Cationic, fluorescent ΔΨm indicator. | Use low concentrations (nM range) for non-quenching mode to minimize toxicity and artifact [1]. |
| FCCP/CCCP | Protonophore uncoupler; dissipates ΔΨm. | Positive control for depolarization. Prepare fresh in DMSO [8]. |
| Oligomycin | ATP synthase inhibitor; causes hyperpolarization. | Positive control for hyperpolarization by inhibiting proton flow back into the matrix [1]. |
| Hanks' Balanced Salt Solution (HBSS) | Physiological buffer for live-cell imaging. | Should contain Ca²⁺/Mg²⁺ and be supplemented with glucose for energy. |
Methodology:
- Cell Preparation: Plate cells in an appropriate imaging dish and culture until they reach 60-80% confluency.
- Dye Loading: Load cells with a low concentration of TMRM (e.g., 20 nM) in pre-warmed HBSS for 30 minutes at 37°C, 5% CO₂. For non-quenching mode, the dye can be maintained in the bath during imaging.
- Baseline Acquisition: Acquire images on a fluorescence microscope (Ex/Em ~548/575 nm) to establish a baseline TMRM intensity.
- Application of Controls:
- Add FCCP/CCCP (e.g., 1-10 µM) and observe a rapid decrease in TMRM fluorescence, confirming depolarization.
- In a separate experiment, add Oligomycin (e.g., 1-5 µM) and observe an increase in TMRM fluorescence, confirming hyperpolarization.
- Data Analysis: Normalize the fluorescence intensity to the baseline. A valid assay will show a clear response to both controls.
The workflow below outlines this critical validation process.
Protocol 2: Differentiating ΔΨm from ΔpHm Using Ratiometric JC-1
This protocol leverages the ratiometric nature of JC-1 to provide a more robust measurement of membrane potential, which is less sensitive to artifacts from changes in mitochondrial mass or dye loading [1] [8].
Research Reagent Solutions:
| Reagent | Function | Key Consideration |
|---|---|---|
| JC-1 Dye | Ratiometric ΔΨm indicator (monomer vs. J-aggregates). | Form aggregates at high potentials (red); remains monomeric at low potentials (green). Sensitive to concentration [1]. |
| FCCP/CCCP | Protonophore uncoupler; positive control. | Used to induce depolarization and collapse the red/green ratio [8]. |
| Dimethyl Sulfoxide (DMSO) | Solvent for JC-1 and many drugs. | Keep concentration low (e.g., <0.1%) to avoid solvent toxicity. |
| Phosphate Buffered Saline (PBS) | Buffer for washing and dye dilution. |
Methodology:
- Dye Solution: Prepare a 2-10 µM JC-1 working solution in serum-free media or PBS from a DMSO stock.
- Staining: Incubate cells with the JC-1 working solution for 15-30 minutes at 37°C, 5% CO₂, protected from light.
- Washing: Gently wash cells twice with pre-warmed PBS to remove excess dye.
- Imaging: Acquire images using two channels:
- Green Channel: FITC filter set (Ex/Em ~514/529 nm) for JC-1 monomers.
- Red Channel: TRITC/PE filter set (Ex/Em ~514/590 nm) for JC-1 J-aggregates.
- Image Analysis: Calculate the ratio of red (aggregate) to green (monomer) fluorescence intensity for each mitochondrion or cell. A decrease in this ratio indicates mitochondrial depolarization.
- Validation: Treat cells with FCCP/CCCP to confirm a collapse of the red/green ratio.
The diagram below illustrates the fundamental principle of the JC-1 assay.
The Scientist's Toolkit: Key Reagents for Mitochondrial Research
Table 2: Essential Reagents for Mitochondrial Membrane Potential Studies
| Reagent Category | Specific Examples | Function & Application | Critical Notes |
|---|---|---|---|
| Cationic ΔΨm Dyes | TMRM, TMRE, Rhod123, JC-1, DiOC₆(3) | Accumulate in mitochondria in a potential-dependent manner; used to monitor ΔΨm in live cells [1]. | TMRM has low binding/toxicity; JC-1 is ratiometric; DiOC₆(3) requires very low conc. for ΔΨm specificity [1]. |
| ΔΨm Disruptors (Controls) | FCCP, CCCP (uncouplers); Oligomycin (ATP synthase inhibitor) | Essential positive controls to validate dye response (FCCP depolarizes, Oligomycin hyperpolarizes) [1] [8]. | Always include in experimental design. Prepare fresh stock solutions in DMSO. |
| Morphology Reference Dyes | MitoTracker Green FM (MG), MitoTracker Deep Red FM (MDR) | Label mitochondria largely independent of ΔΨm (after loading); used as a reference for morphology [6] [30]. | MG signal is largely ΔΨm-independent only after loading; not suitable for tracking dynamic ΔΨm changes [30]. |
| Ion Chelators | BAPTA-AM (calcium chelator) | Used to investigate the role of Ca²⁺ fluxes in observed ΔΨm changes, as Ca²⁺ can alter the potential without changing ΔpHm [1]. | Helps dissect the contribution of non-protonic charges to the measured ΔΨm signal. |
Optimization Strategies for Combination Therapies and Long-Term Exposure Studies
In the context of treatment research, particularly during long-term exposure studies and combination therapy evaluations, accurate measurement of mitochondrial membrane potential (ΔΨm) is crucial. However, researchers frequently encounter dye redistribution artifacts that can compromise data interpretation. These artifacts arise from complex interactions between experimental treatments, dye properties, and mitochondrial ultrastructure. This technical support center provides targeted guidance to identify, troubleshoot, and prevent these common technical challenges, ensuring reliable assessment of mitochondrial function in complex experimental paradigms.
Understanding Dye Redistribution Artifacts
What are dye redistribution artifacts and why do they matter in combination therapy research?
Dye redistribution artifacts occur when fluorescent probes used to measure mitochondrial membrane potential exhibit altered localization patterns that do not accurately reflect true biological changes in ΔΨm. These artifacts are particularly problematic in combination therapy and long-term exposure studies because:
- They mimic pathological changes: Artifactual dye redistribution can be misinterpreted as early apoptosis or mitochondrial dysfunction, leading to false conclusions about treatment efficacy or toxicity [65].
- They complicate mechanism of action studies: When evaluating multiple therapeutic agents simultaneously, dye artifacts can obscure the actual mitochondrial responses to treatment, making it difficult to attribute effects to specific compounds [6] [30].
- They affect high-content screening: Automated morphology analysis depends on consistent dye behavior, which artifacts disrupt, potentially invalidating screening results for drug discovery [30].
How does mitochondrial ultrastructure influence dye redistribution?
The inner mitochondrial membrane is divided into two functionally distinct compartments: the cristae membrane (CM) and inner boundary membrane (IBM), separated by cristae junctions (CJ) [6]. This compartmentalization creates different membrane potential gradients (ΔΨC and ΔΨIBM) that significantly impact dye behavior:
- Cristae junctions act as barriers: The CJ regulates ion movement between compartments, creating distinct electrical environments that affect dye localization [6].
- Compartment-specific potential differences: The CM typically maintains a higher (more negative) membrane potential compared to the IBM due to concentrated proton pump activity [6].
- Treatment-induced alterations: Therapeutic interventions can modify CJ permeability, leading to dye redistribution that may not reflect overall mitochondrial health [6].
Frequently Asked Questions (FAQs)
Q1: Why do I observe different mitochondrial morphology patterns when using TMRM versus MitoTrackers in long-term studies?
A1: These dyes have fundamental differences in mechanism and sensitivity that produce varying results:
- TMRM functions as a potential-sensitive redistribution dye, accumulating in mitochondrial compartments based on membrane potential and readily exiting during depolarization [30].
- MitoTrackers (e.g., CMXRos, MG) bind covalently to mitochondrial proteins, providing more stable morphology assessment but less sensitivity to rapid potential changes [30].
- Comparative performance: In primary human fibroblasts, all probes enable morphology quantification but yield numerically different results. TMRM shows highest sensitivity to FCCP-induced depolarization, while MitoTracker Green demonstrates lowest sensitivity [30].
Q2: How does TMRM concentration affect interpretation of membrane potential gradients in combination therapy screening?
A2: TMRM concentration critically influences observed spatial distribution patterns:
- Low concentrations (1.35-5.4 nM): Preferentially accumulate in cristae membranes, highlighting regions with higher (more negative) potential [6].
- High concentrations (13.5-81 nM): Saturate cristae compartments, with excess dye distributing to inner boundary membranes, potentially obscuring true potential gradients [6].
- Experimental implication: Using inappropriate concentrations can mask treatment-induced changes in compartment-specific membrane potentials, leading to incorrect conclusions about drug effects [6].
Q3: What fixation methods best preserve mitochondrial morphology and dye distribution for endpoint analysis in long-term studies?
A3: Fixation choice significantly impacts morphology preservation:
- Paraformaldehyde (PFA) alone: May disrupt mitochondrial morphology and antigenicity [66].
- PFA-Glutaraldehyde combination (3% PFA/1.5% GA): Better preserves mitochondrial network structure and protein antigenicity while maintaining fluorescent signals [66].
- Critical consideration: JC-1 and similar potential-sensitive dyes require live-cell imaging; fixation eliminates membrane potential and causes dye redistribution [65].
Q4: How can I distinguish true early apoptosis from dye artifacts in combination treatment experiments?
A4: Implement these control strategies:
- Use multiple detection methods: Correlate JC-1 results (red-to-green shift) with other apoptosis markers [65].
- Include appropriate controls: CCCP (carbonyl cyanide m-chlorophenyl hydrazone) serves as a positive control for complete depolarization [65].
- Validate with complementary assays: Combine potential-sensitive dyes with caspase activation assays or cytochrome c release measurements [30] [65].
Troubleshooting Guides
Common Dye Redistribution Artifacts and Solutions
Table 1: Troubleshooting Dye Redistribution Artifacts
| Problem | Possible Causes | Solutions | Prevention Tips |
|---|---|---|---|
| High background fluorescence | Non-specific dye binding; incomplete washing; dye concentration too high | Use background suppressors (e.g., BackDrop Background Suppressor); optimize washing protocols; titrate dye concentration [3] | Validate washing efficiency; include no-dye controls; perform concentration curves |
| Unexpected dye redistribution in control cells | Dye overloading; improper loading temperature; plasma membrane potential changes | Ensure proper loading temperature (37°C); use lower dye concentrations; validate with potentiometric controls [6] [30] | Standardize loading protocols across experiments; include CCCP depolarization controls |
| Loss of signal during long-term imaging | Photobleaching; dye leakage; treatment-induced exporter activation | Use antioxidant mounting media; minimize light exposure; consider protein-binding dyes (Mitotracker) for fixed-timepoint studies [30] | Optimize imaging intervals; validate dye stability in pilot studies |
| Inconsistent morphology quantification between dyes | Differential sensitivity to ΔΨm; distinct binding mechanisms; varied depolarization sensitivity [30] | Standardize analysis parameters; validate with multiple probes; select dyes based on specific experimental questions [30] | Establish probe-specific reference values; use consistent imaging platforms |
Combination Therapy-Specific Challenges
Table 2: Troubleshooting Combination Therapy Artifacts
| Challenge | Impact on ΔΨm Assessment | Resolution Strategies |
|---|---|---|
| Delayed onset of drug effects | Early timepoints may miss progressive ΔΨm changes | Extend monitoring duration; implement frequent sampling; use real-time continuous readouts [67] |
| Additive or synergistic toxicity | Complex ΔΨm response patterns difficult to attribute | Include single-agent controls; implement time-staggered dosing; correlate with viability assays [67] |
| Pharmacokinetic interactions | Altered drug exposure affects ΔΨm dynamics | Monitor compound interactions; measure actual intracellular concentrations; adjust dosing schedules [67] |
| Competing protective and toxic effects | Contradictory ΔΨm responses | Analyze subcellular compartments separately; use high-resolution imaging; correlate with functional endpoints [6] |
Experimental Protocols for Artifact Prevention
Validating Dye Performance in Combination Therapy Studies
Purpose: Establish reliable ΔΨm measurement conditions before initiating combination treatment experiments.
Materials:
- TMRM (1.35-81 nM) or JC-1 (recommended concentration) [6] [65]
- MitoTracker Green FM (500 nM) as morphology reference [6]
- CCCP (10-50 μM) as depolarization control [65]
- Appropriate cell culture model
Procedure:
- Dose titration: Perform concentration series for each dye to identify optimal working concentrations.
- Loading optimization: Incubate cells with dyes for 30 minutes at 37°C [30] [66].
- Depolarization control: Treat samples with CCCP (10 μM, 20 minutes) to validate depolarization response [65].
- Co-localization assessment: For multi-dye experiments, verify spectral separation and absence of dye interactions.
- Time-course validation: Establish dye stability over planned experimental duration.
Protocol for Spatial Membrane Potential Gradient Analysis
Purpose: Quantify compartment-specific ΔΨm changes during treatment exposure.
Materials:
- TMRM (13.5 nM recommended for gradient detection) [6]
- MitoTracker Green FM (500 nM) [6]
- Super-resolution capable microscope (e.g., SIM, STED) [6]
Procedure:
- Cell preparation: Plate cells on imaging-optimized surfaces.
- Dye loading: Co-load with MTG (500 nM) and TMRM (13.5 nM) for 30 minutes at 37°C [6].
- Image acquisition: Acquire super-resolution images using structured illumination microscopy [6].
- IBM association index calculation:
- Use MTG channel to define mitochondrial boundaries
- Create IBM and CM regions through automated shrinking/widening of borders
- Calculate ratio: IBM Association Index = FIBM/FCM [6]
- ΔFWHM analysis:
- Generate cross-section intensity profiles for MTG and TMRM
- Calculate difference in full width at half maximum (ΔFWHM) [6]
Research Reagent Solutions
Table 3: Essential Reagents for Mitochondrial Membrane Potential Studies
| Reagent | Primary Function | Key Considerations | Example Applications |
|---|---|---|---|
| TMRM | Potential-sensitive redistribution dye | Concentration-dependent compartmentalization; reversible binding [6] [30] | Spatial gradient analysis; real-time potential monitoring |
| JC-1 | Ratiometric potential sensor | Monomer (green) aggregate (red) shift with depolarization [65] | Early apoptosis detection; quantitative depolarization assessment |
| MitoTracker Green FM | Morphology reference dye; potential-insensitive after binding [6] | Protein-binding; retained after fixation [6] [30] | Morphology quantification; reference for spatial analysis |
| MitoTracker Red CMXRos | Potential-sensitive protein-binding dye [30] | Retained after fixation; moderate depolarization sensitivity [30] | Fixed-endpoint studies with potential correlation |
| CCCP | Protonophore uncoupler | Complete depolarization positive control [65] | Assay validation; maximum depolarization control |
| PFA-GA Fixative | Morphology preservation | 3% PFA/1.5% GA combination optimal for mitochondrial structure [66] | Endpoint analysis with morphology preservation |
Signaling Pathways and Experimental Workflows
Mitochondrial Membrane Potential Regulation and Compartmentalization
Mitochondrial Membrane Potential Regulation
Experimental Workflow for Combination Therapy Assessment
Combination Therapy Assessment Workflow
Advanced Technical Considerations
Integrating Morphofunctional Analysis
For comprehensive assessment during long-term exposure studies, integrate membrane potential measurements with morphological parameters:
- Simultaneous TMRM and MitoTracker staining: Enables correlation of potential changes with structural alterations [6] [30].
- Automated morphology quantification: Parameters include mitochondrial area, aspect ratio, form factor, and network complexity [30].
- Dynamic monitoring: Track individual mitochondria over time to distinguish transient artifacts from sustained pathological changes [30].
Calcium-Mediated Dye Redistribution
Calcium signaling significantly influences dye behavior through multiple mechanisms:
- MICU1 regulation of cristae junctions: Calcium-dependent MICU1 oligomerization controls CJ permeability, affecting compartment-specific dye distribution [6].
- TCA cycle activation: Mitochondrial calcium uptake stimulates metabolic activity, potentially hyperpolarizing cristae membranes and altering dye gradients [6].
- Fission coupling: Calcium signals precede mitochondrial fission, which itself alters dye distribution patterns [6].
Statistical Considerations for Combination Therapy Studies
- Sample size requirements: Anticipate effect size reductions due to artifact variability.
- Multiple comparison corrections: Adjust for correlated measurements across timepoints and treatment conditions.
- Concentration-response modeling: Account for non-linear dye behavior across potential ranges.
- Replicate strategy: Include biological and technical replicates to distinguish artifacts from true biological effects.
Frequently Asked Questions (FAQs) and Troubleshooting Guides
FAQ 1: Why do I get inconsistent TMRM fluorescence readings between different cell types in the same experiment?
Answer: Inconsistent staining is frequently caused by variable expression of efflux pumps like P-glycoprotein (P-gp), which actively extrudes cationic dyes like TMRM and TMRE. This is particularly prevalent in immune cells, such as invariant Natural Killer T (iNKT) cells, which express high P-gp levels [68].
- Troubleshooting Steps:
- Confirm P-gp Interference: Treat a sample with a P-gp inhibitor (e.g., 1μM PSC833) for 10 minutes prior to dye loading. A significant increase in fluorescence signal suggests P-gp activity is skewing your results [68].
- Use Complementary Methods: Validate your findings with dye-independent measurements, such as quantifying mitochondrial DNA (mtDNA) copy number or using proteomics to assess mitochondrial protein levels [68].
- Dye Selection: If P-gp interference is confirmed, consider using alternative dyes or ensure the inhibitor is included in the staining protocol for consistent results.
FAQ 2: My TMRM signal is weak or absent, even in healthy control cells. What could be wrong?
Answer: A weak signal can stem from several issues related to dye handling, cell health, or instrument settings.
- Troubleshooting Steps:
- Check Dye Integrity and Concentration: Ensure the dye stock is fresh and properly stored. Titrate the dye concentration; low concentrations (e.g., 1.35-5.4 nM) are optimal for detecting hyperpolarization as they preferentially accumulate in cristae, while high concentrations (>40 nM) can saturate the signal [6].
- Verify Mitochondrial Depolarization: Confirm cells are healthy and untreated controls are functioning. Test with a depolarizing agent like FCCP; a significant drop in TMRM signal confirms the dye is responsive to membrane potential [30].
- Review Imaging/Flow Cytometry Settings: Ensure lasers, filters, and detector settings are correctly configured for TMRM. Compare with a known positive control.
FAQ 3: How can I distinguish genuine changes in ΔΨm from dye redistribution artifacts caused by my experimental treatment?
Answer: Dye redistribution is a major artifact, especially during treatments that affect cristae junction permeability or overall ion homeostasis.
- Troubleshooting Steps:
- Use a ΔΨm-Insensitive Reference Stain: Co-stain with a potential-insensitive dye like MitoTracker Green FM (MTG) to control for changes in mitochondrial mass and morphology [6] [30].
- Analyze Spatial Distribution: Use high-resolution microscopy to check for abnormal dye distribution. In healthy cells, low concentrations of TMRM should localize to the cristae. Redistribution to the inner boundary membrane can indicate hyperpolarization or altered cristae junction permeability [6].
- Employ a Potentiometric Control: Always include a control well treated with FCCP to define the baseline for a fully depolarized state. The difference between the FCCP-treated signal and the control signal represents the valid potentiometric component of your measurement.
Experimental Protocols for Key Validation Experiments
Protocol 1: Validating Dye Specificity and P-gp Interference
Purpose: To determine if P-glycoprotein activity is confounding ΔΨm measurements, especially when comparing different cell populations [68].
Workflow:
Materials:
- Cell suspension
- P-gp inhibitor (e.g., PSC833)
- Vehicle control (e.g., DMSO)
- TMRM or TMRE dye
- Viability dye (e.g., 7-AAD)
- Flow cytometer or microscope
Procedure:
- Prepare a single-cell suspension and stain with a viability dye.
- Split the sample into two equal portions.
- Pre-treat one portion with 1μM PSC833 for 10 minutes at 37°C. Treat the other portion with vehicle only.
- Add TMRM (e.g., 5-13.5 nM) to both portions and incubate for 15-30 minutes at 37°C.
- Analyze by flow cytometry or microscopy without washing.
- Interpretation: A rightward shift (increased fluorescence) in the PSC833-treated sample compared to the vehicle control indicates significant P-gp-mediated dye efflux. Data from such cell types without inhibitor treatment is unreliable.
Protocol 2: Establishing a QC Range Using Potentiometric Controls
Purpose: To define acceptance criteria for ΔΨm experiments by quantifying the signal range between fully energized and fully depolarized mitochondria.
Workflow:
Materials:
- Cell culture plate (e.g., 96-well)
- Potentiometric control compounds: FCCP (e.g., 1-10 μM) or CCCP
- ΔΨm dye (e.g., TMRM)
- Fluorescence plate reader or flow cytometer
Procedure:
- Plate cells at a consistent density in at least 12 replicate wells.
- On the day of the experiment, assign wells to three groups:
- Positive Control (High ΔΨm): Untreated cells.
- Negative Control (Depolarized): Cells treated with FCCP/CCCP for 15-30 minutes prior to reading.
- Test Samples: Cells with your experimental treatment.
- Load all wells with an optimal concentration of TMRM and incubate.
- Measure fluorescence intensity.
- QC Metric Calculation: Calculate the Signal-to-Background Ratio and Z'-factor to assess assay robustness.
- Signal-to-Background: Mean(Positive Control) / Mean(Negative Control). A ratio of >3 is generally desirable.
- Z'-factor: ( 1 - \frac{3 \times (SD{positive} + SD{negative})}{|Mean{positive} - Mean{negative}|} ). An assay with Z' > 0.5 is considered excellent for screening.
Key Research Reagent Solutions
Table 1: Essential Reagents for ΔΨm Measurement and QC.
| Reagent | Function/Brief Explanation | Key Considerations for QC |
|---|---|---|
| TMRM/TMRE | Cationic, potentiometric dye; accumulates in mitochondria proportional to ΔΨm. | Concentration is critical; use low nM range (e.g., 2-20 nM) to avoid saturation and enable detection of hyperpolarization [6] [30]. |
| MitoTracker Green FM (MTG) | ΔΨm-insensitive dye; binds to mitochondrial proteins, useful as a mass/morphology reference [6] [30]. | Use to distinguish ΔΨm changes from changes in mitochondrial mass or number. Not a standalone measure of mass if P-gp is expressed [68]. |
| FCCP/CCCP | Protonophore; uncouples oxidative phosphorylation by dissipating the proton gradient, serving as a negative control for full depolarization. | Use fresh stocks and titrate for each cell type (typical range 1-10 μM). Validates that the dye signal is potential-dependent. |
| PSC833 | Potent and specific P-glycoprotein inhibitor. | Use (e.g., 1 μM) to confirm or prevent P-gp-mediated dye efflux, which can cause severe underestimation of ΔΨm [68]. |
| Rotenone/Antimycin A | Inhibitors of Electron Transport Chain (ETC) Complex I and III, respectively. | Used to inhibit proton pump activity and validate that ΔΨm changes are dependent on ETC function [6]. |
Table 2: Quantitative Data on TMRM Distribution and Concentration Effects. Based on super-resolution microscopy (SIM) data from Scientific Reports [6].
| TMRM Concentration | Primary Localization | IBM Association Index (HeLa) | ΔFWHM | Interpretation for Experiment Design |
|---|---|---|---|---|
| Low (1.35 - 5.4 nM) | Cristae Membranes (CM) | Low | High | Optimal for detecting hyperpolarization (e.g., after Ca²⁺ stimulation). |
| High (40.5 - 81 nM) | Inner Boundary Membrane (IBM) | High | Low | Cristae become saturated. Less sensitive to dynamic changes; can mask hyperpolarization. |
Validation Strategies and Next-Generation Solutions for ΔΨm Imaging
Orthogonal Validation Using Functional Assays and Metabolic Probes
FAQs on Orthogonal Validation and Common Experimental Challenges
Q1: What is orthogonal validation, and why is it critical in mitochondrial research? Orthogonal validation is the practice of verifying experimental results using two or more independent, non-overlapping methods. In the context of mitochondrial research, it is crucial for confirming that observed changes in membrane potential (ΔΨm) are genuine and not due to artifacts from dye redistribution, non-protonic ion fluxes, or other confounding factors. Using independent methods controls for bias and results in more conclusive evidence of specificity [69]. For instance, a change in TMRM fluorescence should be corroborated by a functional assay, such as ATP production measurement, to ensure the signal reflects a true biological event [6].
Q2: My TMRM signal decreases after treatment. Does this always indicate mitochondrial depolarization? Not necessarily. A decrease in TMRM fluorescence can be caused by several factors other than true depolarization. Key artifacts to rule out include:
- Plasma Membrane Potential (ΔΨp) Changes: A loss of ΔΨp can reduce dye uptake into the cell, indirectly lowering mitochondrial signal [1].
- Dye Saturation or Quenching: Using TMRM at high concentrations (>50-100 nM) can lead to aggregation and quenching, where fluorescence decreases without a change in ΔΨm [1] [30].
- Non-Protonic Charges: Cationic dyes like TMRM are sensitive to other ions, particularly Ca²⁺. An influx of calcium can cause hyperpolarization that masks a concurrent loss of proton gradient [1].
- Altered Mitochondrial Mass or Morphology: A treatment that fragments mitochondria or reduces their network size can appear as a loss of signal [30].
Q3: What are the best-practice controls for experiments using TMRM? To ensure meaningful interpretation of TMRM data, include these essential controls [1]:
- FCCP/CCCP Uncoupler Control: Apply a protonophore (e.g., 1-10 µM FCCP) at the experiment's end to fully collapse ΔΨm. This confirms the dye is responding to the membrane potential and sets a baseline for depolarization.
- Oligomycin Control: Using an ATP synthase inhibitor (e.g., 1-5 µM Oligomycin) can hyperpolarize mitochondria by preventing proton re-entry, validating the dye's response to hyperpolarization.
- Dye Concentration Controls: Use TMRM at the lowest possible concentration that provides a robust signal (typically 1-30 nM for non-quenching mode) to avoid artifacts from saturation, quenching, or inhibition of the electron transport chain [1] [30].
- Correlation with Functional Assays: Always pair dye measurements with an orthogonal functional readout, such as ATP levels or oxygen consumption rate (OCR) [6].
Q4: How does orthogonal validation apply to the use of chemical probes beyond dyes? The same principles govern the use of chemical probes (e.g., kinase inhibitors). The "Rule of Two" is a community best practice recommending that every study should use [70]:
- At least two orthogonal chemical probes (different chemical structures targeting the same protein).
- A paired target-inactive control compound (a structurally similar but inactive molecule).
- Probes used within their recommended concentration range. A systematic review found that only 4% of publications adhered to these standards, highlighting a major source of non-reproducible results [70].
Troubleshooting Guide for Dye Redistribution Artifacts
| Problem | Potential Cause | Orthogonal Validation Strategy |
|---|---|---|
| Signal Drop Not Due to Depolarization | Loss of plasma membrane potential (ΔΨp); Dye saturation/quenching; Changes in mitochondrial mass [1] [30]. | Measure ΔΨp with a plasma membrane-specific dye (e.g., DiBAC₄(³)); Use TMRM in non-quenching mode (low concentration); Correlate with mitochondrial mass markers (e.g., Mitotracker Green FM under depolarized conditions) [30]. |
| Hyperpolarization Artifact | Dye response to non-protonic cation influx (e.g., Ca²⁺) [1]. | Correlate with a direct measure of mitochondrial pH (e.g., mt-SNARF) to dissociate ΔΨm from ΔpHm; Use Ca²⁺ chelators (BAPTA-AM) to isolate the protonic component [1]. |
| Unspecific Probe Effects | Off-target effects of a chemical probe used to induce a biological state [70]. | Employ the "Rule of Two": use a second probe with a different chemical structure and a matched target-inactive control compound [70]. |
| Confounding Metabolic Variation | Intrinsic physiological variation (e.g., diurnal rhythms) obscuring treatment effects [71]. | Use data filtering methods like Orthogonal Signal Correction (OSC) to remove confounding variation before statistical analysis [71]. |
Experimental Protocols for Key Orthogonal Assays
Protocol 1: Correlative Measurement of ΔΨm and ATP Production This protocol is adapted from multi-parameter microscopy studies [6].
Objective: To simultaneously monitor changes in mitochondrial membrane potential and ATP levels in live cells, ensuring that depolarization events correlate with a functional energy output defect.
Materials:
- Cell Line: Primary human skin fibroblasts or EA.hy926 cells [6] [30].
- Dyes: TMRM (1.35 - 40.5 nM for SIM, 20-30 nM for epifluorescence) and a FRET-based mitochondrial ATP biosensor (e.g., mtAT1.03) [6].
- Controls: FCCP (1-10 µM), Oligomycin (1-5 µM), Histamine (100 µM for Ca²⁺ induction) [6].
- Equipment: Structured Illumination Microscope (SIM) or high-resolution epifluorescence microscope with environmental control.
Method:
- Cell Preparation: Plate cells on glass-bottom dishes 24-48 hours before imaging to achieve 60-80% confluency.
- Dye Loading: Incubate cells with TMRM and the FRET-based ATP sensor in imaging buffer for 20-30 minutes at 37°C. Use the lowest effective TMRM concentration to avoid quenching [6] [30].
- Image Acquisition: Acquire time-lapse images of both TMRM and FRET channels before and after treatment.
- For TMRM, use SIM to resolve spatial membrane potential gradients between the inner boundary membrane (IBM) and cristae membrane (CM). Calculate the IBM association index or ΔFWHM to quantify relative changes in ΔΨc [6].
- For the ATP sensor, calculate the FRET ratio (emission donor/emission acceptor).
- Data Analysis: Plot TMRM distribution parameters and the FRET ratio over time. A true depolarization should show a coordinated decrease in the TMRM IBM association index (indicating cristae depolarization) and a drop in the FRET ratio (indicating falling ATP levels) [6].
Protocol 2: Validating a Chemical Probe with Orthogonal Controls This protocol is based on guidelines for the use of chemical probes in cell-based research [70].
Objective: To confirm that a phenotypic effect of a chemical probe is due to on-target inhibition and not an off-target artifact.
Materials:
- Chemical Probe: e.g., AZD2014 (mTOR inhibitor), THZ1 (CDK7/12/13 inhibitor) [70].
- Orthogonal Probe: A second inhibitor against the same target with a different chemical structure (e.g., for mTOR: Torin1 vs. AZD2014) [70].
- Inactive Control: A structurally matched, target-inactive compound (e.g., for UNC0638, use UNC0737) [70].
- Assay Reagents: Cell viability assay (e.g., MTT, CellTiter-Glo) or a target-specific functional readout (e.g., Western blot for phosphorylation status).
Method:
- Dose-Response: Treat cells with a range of probe concentrations (e.g., 1 nM - 10 µM) for 24-72 hours. Crucially, ensure the highest concentration used is within the probe's recommended range (often below 1 µM) to maintain selectivity [70].
- Inactive Control: In parallel, treat cells with the same concentrations of the target-inactive control compound. The absence of the phenotype with this control supports on-target activity.
- Orthogonal Probe Validation: Repeat the dose-response experiment with the second, structurally distinct orthogonal probe. The observation of a similar phenotypic effect strengthens the conclusion.
- Data Interpretation: A high-confidence result is achieved when the phenotype is observed with the primary probe at its selective concentration, is not seen with the inactive control, and is recapitulated by the orthogonal probe.
Quantitative Data on Best-Practice Probe Usage
The table below summarizes findings from a systematic review of 662 publications, highlighting the suboptimal use of chemical probes in biomedical research [70].
| Validation Criteria | Description | Percentage of Publications Adhering to Practice |
|---|---|---|
| Optimal Concentration | Using the chemical probe within its recommended concentration range (typically <1 µM for on-target effect). | 4% |
| Use of Inactive Control | Including a structurally matched, target-inactive control compound in the experimental design. | 4% |
| Use of Orthogonal Probes | Employing a second, structurally independent chemical probe to target the same protein. | 4% |
| All Three Criteria | Simultaneously using the probe at the recommended concentration, an inactive control, and an orthogonal probe. | 4% |
The Scientist's Toolkit: Essential Research Reagents
| Reagent | Function & Application in Orthogonal Validation |
|---|---|
| TMRM / TMRE | Cationic, fluorescent dye used to measure ΔΨm in non-quenching (low nM) or quenching (high nM) modes. Preferred for low mitochondrial binding and minimal ETC inhibition [1] [30]. |
| FCCP / CCCP | Proton ionophores that uncouple mitochondrial respiration by dissipating the proton gradient. Serves as a critical positive control for complete depolarization [1] [30]. |
| Oligomycin | ATP synthase inhibitor. Used as a control to induce hyperpolarization of ΔΨm by blocking proton flow back into the matrix [1]. |
| Matched Inactive Control Compound | A structurally similar analog of a chemical probe that lacks activity against the primary target. Essential for controlling for off-target effects and vehicle-related phenotypes [70]. |
| Orthogonal Chemical Probe | A second chemical tool with a different chemical structure that inhibits the same target as the primary probe. Used to confirm on-target biology [70]. |
| MitoTracker Green FM (MG) | Mitochondrial mass marker that accumulates independently of membrane potential (after fixation). Used to normalize ΔΨm signals or assess morphology, though its signal can be potential-dependent in live cells [6] [30]. |
| mt-SNARF | Ratiometric, pH-sensitive dye targeted to the mitochondria. Used to measure ΔpHm orthogonally to ΔΨm, helping to dissociate the two components of the proton motive force [1]. |
Experimental Workflow and Signaling Pathways
The following diagram illustrates the integrated workflow for orthogonally validating mitochondrial membrane potential changes and the associated signaling relationships that can confound interpretation.
Comparative Analysis of Cationic vs. Neutral Dye Performance in Diseased Cells
Accurate assessment of mitochondrial function is fundamental to advancing our understanding of cellular health, disease mechanisms, and therapeutic development. Mitochondria, as central regulators of cellular metabolism and apoptosis, represent promising targets for both diagnostic imaging and therapeutic intervention [72]. The investigation of mitochondrial morphology, dynamic distribution, and membrane potential provides critical support for understanding mitochondrial pathology and associated diseases [72]. Fluorescent microscopy offers powerful in-situ and real-time visualization of mitochondrial dynamics with molecular specificity, complementing ultrastructural methods [72]. However, the choice between cationic and neutral dyes introduces significant methodological considerations that profoundly impact data interpretation, particularly in diseased cell models with compromised membrane potentials.
The redistribution artifacts of cationic dyes in treated or diseased cells represent a substantial challenge in treatment research. These artifacts stem from the fundamental reliance of cationic dyes on the mitochondrial membrane potential (ΔΨm) for their accumulation, which becomes problematic when studying disease states or treatments that inherently alter this potential. This technical review provides a comprehensive comparative analysis and troubleshooting framework to guide researchers in selecting appropriate dyes and mitigating artifacts in their experimental systems.
Fundamental Mechanisms: How Dyes Accumulate in Mitochondria
Cationic Dye Accumulation Mechanism
Cationic dyes primarily rely on electrostatic interactions for mitochondrial localization. The proton-pumping activity of the electron transport chain generates a significant electric gradient across the inner mitochondrial membrane (ΔΨm ≈ -150 to -180 mV), creating a negatively charged environment within the mitochondrial matrix [72]. This potential drives the accumulation of lipophilic cations through the Nernst equation principle. Representative examples include derivatives of triphenylphosphonium (TPP+), quaternary ammonium salts, and pyridinium salts [72]. The accumulation occurs because the positively charged molecules are electrophoretically driven into the mitochondrial matrix in response to the negative potential inside. While this mechanism provides excellent signal-to-noise ratio in healthy cells, it becomes problematic in diseased or treated cells where ΔΨm may be compromised, leading to potential-insensitive dye redistribution and inaccurate measurements.
Neutral Dye Accumulation Mechanism
Neutral dyes utilize fundamentally different targeting mechanisms that do not depend solely on membrane potential. These dyes achieve mitochondrial localization primarily through intense hydrophobic interactions with the inner mitochondrial membrane lipid bilayer [72]. Neutral or weakly cationic probes with large hydrophobic domains can integrate into the membrane structure without requiring a strong electrostatic driving force. For instance, fluorescent probes bearing acetoxymethyl esters and long alkyl chains efficiently target mitochondria through this mechanism [72]. Some neutral probes may also achieve localization through covalent conjugation to mitochondrial macromolecules, forming stable covalent bonds that eliminate ΔΨm dependence entirely [72]. This potential-independent accumulation makes neutral dyes particularly valuable for studies involving cells with depolarized membranes.
Quantitative Performance Comparison in Diseased Cells
Table 1: Comparative Performance Characteristics of Cationic vs. Neutral Mitochondrial Dyes
| Parameter | Cationic Dyes | Neutral Dyes | Experimental Implications |
|---|---|---|---|
| ΔΨm Dependence | High (electrophoretic accumulation) [72] | Low (hydrophobic partitioning) [72] | Neutral dyes maintain localization in depolarized diseased cells |
| Targeting Mechanism | Electrostatic targeting via mitochondrial membrane potential [72] | Hydrophobic interactions with IMM lipid bilayer [72] | Neutral dyes less affected by ΔΨm fluctuations in pathology |
| Accumulation in Depolarized Conditions | Severely reduced or absent [72] [5] | Maintained [72] | Cationic dyes fail in diseased cells with compromised ΔΨm |
| Signal Stability in Treatment Studies | Unstable during ΔΨm-altering treatments [28] | Stable regardless of ΔΨm changes [72] | Neutral dyes provide consistent imaging during longitudinal studies |
| Specificity in Diseased Cells | Compromised due to reduced ΔΨm [72] | Maintained regardless of metabolic state [72] | Neutral dyes offer reliable targeting across disease models |
| Cytotoxicity Potential | Higher due to charge interactions [72] | Lower with neutral skeletons [72] | Neutral dyes preferred for long-term live-cell imaging |
Table 2: Artifact Profiles and Technical Limitations in Disease Research
| Artifact Type | Cationic Dyes | Neutral Dyes | Impact on Data Interpretation |
|---|---|---|---|
| Redistribution Artifacts | Significant during treatment-induced ΔΨm loss [72] | Minimal [72] | False-negative results with cationic dyes in treatment studies |
| Membrane Potential Sensitivity | High (slow-response probes) [3] | Limited to none [72] | Cationic dyes better for monitoring transient potential changes |
| Phototoxic Effects | Variable depending on structure | Similar variable potential | Affects long-term viability in live-cell imaging |
| Measurement Specificity | Reflects ΔΨm status rather than mass [5] | Reflects mitochondrial mass/location [5] | Fundamental difference in biological parameter measured |
| Dye Leakage | Potential-dependent [5] | Minimal due to covalent binding or hydrophobic retention [72] | Signal loss issues with cationic dyes during extended imaging |
Experimental Protocols for Validating Dye Performance
Protocol: Validation of Dye Performance in Depolarized Cells
Purpose: To compare the retention and localization accuracy of cationic versus neutral dyes in experimentally depolarized cells, simulating diseased states with compromised mitochondrial membrane potential.
Reagents:
- Cationic dye (e.g., TMRM, JC-1) [6]
- Neutral dye (e.g., BTNDP, CytoPainter) [72] [5]
- Depolarizing agent (e.g., CCCP, carbonyl cyanide m-chlorophenyl hydrazone)
- Cell culture medium appropriate for cell line
- Phosphate buffered saline (PBS)
- Paraformaldehyde (if fixed imaging is required)
Procedure:
- Culture cells in appropriate medium on glass-bottom dishes for 24-48 hours until 70-80% confluent.
- Divide cells into three treatment groups: untreated control, acute depolarization, and chronic depolarization.
- For dye loading, incubate cells with the recommended concentration of cationic or neutral dye in culture medium for 30 minutes at 37°C [72].
- Replace dye-containing medium with fresh warm medium and incubate for additional 30 minutes to allow for complete esterase activation and intracellular distribution.
- For depolarization groups, add CCCP (10-50 μM final concentration) either during dye loading (chronic) or after dye loading and washing (acute).
- Image using appropriate microscopy systems. For simultaneous membrane potential assessment, include a cationic dye control that reflects ΔΨm status.
- For fixed-cell studies, use paraformaldehyde fixation (avoid methanol) to preserve dye localization [5].
Validation Metrics:
- Quantitative analysis of mitochondrial localization using Pearson's correlation coefficient with structural markers
- Measurement of signal retention over time in depolarized conditions
- Assessment of dye leakage rates following depolarization
- Comparison of signal-to-noise ratio in healthy versus depolarized cells
Protocol: Multiparameter Assessment of Dye-Induced Artifacts
Purpose: To systematically evaluate potential artifacts introduced by dye compounds themselves, including effects on cell mechanics and function.
Reagents:
- Test dyes (cationic and neutral comparators)
- Cell tracing dyes (CMFDA, CMTMR, Calcein Green AM, CFDA-SE) [73]
- AFM setup with microsphere-modified probes [73]
- Live-cell imaging system
Procedure:
- Culture cells as described in Protocol 4.1.
- Incubate cells with dyes following standard uptake procedures [73].
- For mechanical property assessment, use atomic force microscopy (AFM) with microsphere-modified probes to measure single-cell compression and adhesion [73].
- Record force-deformation profiles for minimum of 30 cells per condition.
- Extract Young's moduli of cellular membrane and cytoskeleton using Hertzian mechanics and elastic membrane theory [73].
- Parallel samples should be assessed for viability markers (annexin V/PI) [28] and proliferation rates (BrdU incorporation) [28] to correlate mechanical changes with functional impacts.
- Include DMSO-only controls to account for solvent effects [73].
Analysis:
- Compare stiffness changes relative to untreated controls
- Quantify adhesion alterations to probes
- Correlate mechanical changes with functional impacts on proliferation and viability
- Establish concentration thresholds for significant artifact induction
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Mitochondrial Dye Studies
| Reagent Category | Specific Examples | Function/Application | Key Considerations |
|---|---|---|---|
| Cationic Dyes | TMRM, JC-1, Rhodamine derivatives [72] [6] | Monitoring mitochondrial membrane potential dynamics | Susceptible to redistribution in depolarized cells; ideal for ΔΨm kinetics |
| Neutral Dyes | BTNDP, CytoPainter Red/Green [72] [5] | Mitochondrial morphology and mass assessment; ΔΨm-independent imaging | Maintain localization in diseased cells; better for fixed samples |
| Depolarizing Agents | CCCP, FCCP, Rotenone, Antimycin A [6] | Experimental validation of ΔΨm-independent dye performance | Concentration optimization required; time-dependent effects |
| Viability Assessment | Annexin V, Propidium Iodide, BrdU [28] | Multiparameter analysis of cell health during dye validation | Critical for assessing dye toxicity and functional impacts |
| Mechanical Property Tools | AFM with microsphere probes [73] | Quantification of dye-induced changes in cell stiffness and adhesion | Reveals artifacts not detected by conventional viability assays |
| Background Suppressors | BackDrop Background Suppressor (R37603) [3] | Reducing extracellular background fluorescence | Particularly useful for neuronal cells and high-sensitivity detection |
Troubleshooting Guide: Frequently Asked Questions
Q: My cationic dye fails to label mitochondria in my diseased cell model. What alternatives exist?
A: This common issue arises from compromised mitochondrial membrane potential in diseased cells. Transition to neutral dyes such as BTNDP or structural dyes like CytoPainter series that utilize ΔΨm-independent targeting mechanisms [72] [5]. These dyes achieve mitochondrial localization through hydrophobic interactions with the inner mitochondrial membrane lipid bilayer rather than electrostatic accumulation [72]. Validation experiments should include depolarization controls (e.g., CCCP treatment) to confirm ΔΨm-independent retention.
Q: How do I determine if observed fluorescence changes reflect genuine biological changes versus dye redistribution artifacts?
A: Implement a multiparametric validation approach:
- Use a combination of cationic and neutral dyes in parallel experiments [72] [5]
- Include structural markers (e.g., TOMM20, COX IV antibodies) as localization references [5]
- Measure independent parameters of mitochondrial function (e.g., ATP production, oxygen consumption) to correlate with imaging data [6]
- Perform time-course experiments to distinguish rapid redistribution from gradual biological changes
Q: What specific experimental conditions most exacerbate cationic dye redistribution artifacts?
A: Several conditions profoundly affect cationic dye performance:
- Treatment with electron transport chain inhibitors (rotenone, antimycin A) [6]
- Apoptosis induction leading to mitochondrial outer membrane permeabilization
- Metabolic perturbations that uncouple oxidative phosphorylation
- Disease models with inherent mitochondrial dysfunction (neurodegeneration, cancer) [72]
- Long-term imaging where phototoxicity may compromise ΔΨm
Q: Can dye selection affect measurements beyond mere localization, such as cellular mechanical properties?
A: Yes, evidence indicates that some fluorescent dyes significantly alter cell mechanics. Studies demonstrate that cell tracing dyes can increase living cell stiffness 3-6 times and cell-to-probe adhesion up to 7 times [73]. These effects can be more significant than those induced by certain toxins. When conducting mechanobiology studies alongside mitochondrial imaging, include appropriate controls to account for these effects, and consider using minimal dye concentrations validated for your specific application.
Visualizing Experimental Decision Pathways
The comparative analysis of cationic versus neutral dye performance reveals critical considerations for mitochondrial research in diseased cells. Cationic dyes, while excellent reporters of membrane potential dynamics in healthy systems, suffer from significant redistribution artifacts in disease contexts with compromised ΔΨm. Neutral dyes offer a robust alternative through their potential-independent targeting mechanisms, maintaining reliable localization regardless of pathological status. The strategic selection between these dye classes should be guided by the specific research question, with cationic dyes preferred for functional assessment of ΔΨm dynamics in controlled systems, and neutral dyes superior for morphological studies and investigations involving substantial mitochondrial dysfunction. As research increasingly focuses on diseased cellular states, understanding and mitigating dye redistribution artifacts becomes essential for generating biologically meaningful data and advancing our understanding of mitochondrial pathology.
Troubleshooting Guides
Guide 1: Addressing Probe Accumulation Failures in Depolarized Mitochondria
Problem: My mitochondrial probe fails to accumulate in my cell model, which is known to have a depleted mitochondrial membrane potential (ΔΨm).
Potential Cause 1: Use of a ΔΨm-dependent probe.
- Solution: Switch to a ΔΨm-independent probe, such as the neutral benzothiazole-based probe BTNDP. Unlike cationic probes like its analog BTVMP or traditional dyes (e.g., TMRM), BTNDP accumulates in mitochondria via a hydrophobic interaction mechanism that does not rely on the electrical gradient [72].
- Verification Protocol: Treat a sample of your cells with a mitochondrial uncoupler like CCCP to depolarize the membranes. If the probe signal is lost, it is ΔΨm-dependent. A ΔΨm-independent probe will maintain its localization.
Potential Cause 2: Poor membrane permeability of the probe.
- Solution: Ensure the probe has adequate lipophilicity. The neutral BTNDP probe demonstrates efficient cell membrane crossing, a feature that can be compromised by highly charged molecular structures [72].
- Verification Protocol: Compare the performance of your probe with a well-characterized neutral probe in a live-cell imaging experiment. Check for uniform cytosolic distribution prior to mitochondrial accumulation.
Guide 2: Managing Fluorescence Signal Issues During SIM Imaging
Problem: The fluorescence signal is weak or bleaches too quickly during Structured Illumination Microscopy (SIM) imaging of mitochondria.
Potential Cause 1: Probe is not suitable for super-resolution imaging.
- Solution: Select a probe with high photon output and photostability. The benzothiazole fluorophore is a good candidate. For SIM, probes should have a high fluorescence quantum yield and stable optical properties to support the multiple image acquisitions required for reconstruction [74].
- Verification Protocol: Confirm the probe's quantum yield and photostability data from the literature or perform a simple photobleaching test under a confocal microscope before attempting super-resolution imaging.
Potential Cause 2: Incorrect dye concentration or imaging parameters.
- Solution: Optimize dye concentration and imaging settings. For membrane potential-sensitive dyes like TMRM, use low concentrations (e.g., 1.35-5.4 nM) to visualize spatial potential gradients across cristae membranes without saturating the signal [6].
- Verification Protocol: Perform a concentration series to find the ideal balance between signal intensity and resolution. For TMRM, low concentrations will show a punctate (cristae) pattern, while high concentrations show a more uniform mitochondrial signal [6].
Frequently Asked Questions (FAQs)
FAQ 1: What is the main advantage of using a ΔΨm-independent mitochondrial probe like BTNDP in drug development research?
Traditional cationic probes (e.g., TPP+, Rhodamine derivatives) rely on the negative potential across the inner mitochondrial membrane for accumulation. In diseased cells, such as cancer or neurodegenerative models, the ΔΨm is often compromised, leading to a loss of probe signal and false-negative results [72] [75]. ΔΨm-independent probes, such as the neutral benzothiazole-based BTNDP, accumulate via hydrophobic interactions, ensuring consistent mitochondrial labeling regardless of the metabolic or pathological state of the cell. This provides more accurate and reliable data for evaluating drug effects on mitochondrial morphology and function [72].
FAQ 2: How can I distinguish between different targeting mechanisms when characterizing a new mitochondrial probe?
You can perform a simple pharmacological test using a mitochondrial uncoupler:
- ΔΨm-Dependent Targeting: Probe accumulation is diminished or lost upon treatment with an uncoupler like CCCP.
- ΔΨm-Independent Targeting: Probe accumulation is maintained after CCCP treatment, indicating a mechanism based on hydrophobic interactions or covalent binding [72]. Systematically comparing a neutral probe (BTNDP) with its cationic analog (BTVMP) under depolarized conditions is an effective strategy to confirm the targeting mechanism [72].
FAQ 3: My research involves tracking biothiols like Cysteine. Are there benzothiazole-based probes for this purpose?
Yes, benzothiazole derivatives are highly versatile. For example, the probe DNBS-NHBBT uses a 2,4-dinitrobenzenesulfonyl (DNBS) group that is cleaved by thiols, releasing the highly fluorescent benzothiazole amine (NH2BBT) and turning on the signal [76]. Another probe, HBT-T, utilizes an acrylate group that reacts with Cysteine via a Michael addition and cyclization, leading to a fluorescence turn-on response with high selectivity over other biothiols like homocysteine and glutathione [77]. These probes are useful for monitoring redox homeostasis in the context of drug treatments.
FAQ 4: What are the key parameters to measure when setting up a multiparametric flow cytometry experiment to study mitochondrial function and cell death?
A robust integrated protocol can assess up to eight parameters from a single sample. Key stainings and their purposes are summarized in the table below [28].
Table: Essential Reagents for Multiparametric Flow Cytometry Analysis of Cell State
| Parameter | Reagent | Function |
|---|---|---|
| Proliferation | CellTrace Violet | Tracks cell division by dye dilution in daughter cells. |
| Cell Cycle | Bromodeoxyuridine (BrdU) / Propidium Iodide (PI) | Identifies cells in S-phase (BrdU+) and quantifies DNA content for G1/G2/M phases (PI). |
| Apoptosis | Annexin V / PI | Distinguishes live (Annexin V-/PI-), early apoptotic (Annexin V+/PI-), and late apoptotic/necrotic cells (Annexin V+/PI+). |
| Mitochondrial Membrane Potential | JC-1 | Differentiates between polarized mitochondria (red J-aggregates) and depolarized mitochondria (green monomers). |
FAQ 5: Why is the mitochondrial membrane potential not uniform within a single mitochondrion?
Advanced super-resolution microscopy techniques like SIM have revealed that the inner mitochondrial membrane is divided into two main compartments with different membrane potentials: the cristae membrane (CM, more negative) and the inner boundary membrane (IBM, less negative) [6]. The narrow cristae junction acts as a barrier, separating these electrical potentials. This gradient is physiologically important; for instance, mitochondrial calcium uptake can hyperpolarize the cristae membranes specifically, boosting ATP production, while the cristae junction can act as an "overflow valve" to protect mitochondrial integrity [6].
Data Presentation
Table: Quantitative Comparison of Benzothiazole-Based Fluorescent Probes
| Probe Name | Target / Analytic | Key Mechanism | Detection Limit / Performance | Primary Application |
|---|---|---|---|---|
| BTNDP [72] | Mitochondria (ΔΨm-independent) | Neutral; hydrophobic interaction | Maintains staining under CCCP treatment | Mitochondrial imaging and Photodynamic Therapy (PDT) in diseased cells. |
| DNBS-NHBBT [76] | H₂S & Biothiols | DNBS cleavage | Quantum yield of product (NH₂BBT) > 80% | Detection of reactive sulfur species. |
| HBT-T [77] | Cysteine (Cys) | ESIPT & ICT; acrylate cleavage | 1.92 μM; detection in 15 seconds | Selective tracking of endogenous/exogenous Cys in cells and zebrafish. |
| Unnamed (CORM-3 probe) [78] | CORM-3 (CO donor) | "Turn-on" NIR response | LOD = 0.034 μM | Visualization of CORM-3 in living cells. |
Experimental Protocols
Protocol 1: Validating ΔΨm-Independent Mitochondrial Targeting
Purpose: To confirm that a novel probe accumulates in mitochondria independently of the membrane potential.
Reagents:
- Novel mitochondrial probe (e.g., BTNDP) [72]
- Control ΔΨm-dependent dye (e.g., TMRM, BTVMP) [72]
- Mitochondrial uncoupler (e.g., CCCP, 10-20 μM)
- Appropriate cell culture medium and cell line (e.g., 4T1 cells)
Methodology:
- Cell Preparation: Seed cells in a multi-well imaging plate and culture until ~70% confluent.
- Dye Loading:
- Divide cells into two groups.
- Load both groups with the novel probe according to the manufacturer's instructions.
- Incubate for an appropriate time (e.g., 30 min at 37°C).
- Uncoupler Treatment:
- For the test group, add CCCP to the medium and incubate for 15-30 minutes.
- The control group receives vehicle only.
- Image Acquisition: Image the cells using a fluorescence microscope with appropriate settings.
- Analysis: Compare the mitochondrial staining pattern and intensity between the CCCP-treated and control groups. A ΔΨm-independent probe will show retained, bright punctate staining, while a dependent probe's signal will be drastically reduced or diffuse.
Protocol 2: Assessing Anti-Tumor Efficacy via Mitochondria-Targeted Photodynamic Therapy (PDT)
Purpose: To evaluate the therapeutic potential of a photosensitizing probe like BTNDP.
Reagents:
- Photosensitizer (e.g., BTNDP) [72]
- Cell culture materials (DMEM, FBS, antibiotics, 96-well plates)
- Light source for irradiation (e.g., white light at 28 mW/cm²)
Methodology:
- Cell Seeding: Seed cells (e.g., 4T1) in 96-well plates at a density of 8 × 10⁴ cells/mL and culture for 12 hours [72].
- Probe Incubation: Add different concentrations of the probe (BTNDP) to the wells and incubate for 12 hours at 37°C.
- Light Irradiation: Replace the medium with fresh, probe-free medium. Irradiate the plates with white light for a set time (e.g., 10 min). Include control wells that are not irradiated.
- Viability Assay: After further incubation (e.g., 12 hours), assess cell viability using a standard assay (e.g., MTT, CCK-8).
- Data Analysis: Calculate the percentage of cell death in irradiated vs. non-irradiated groups to determine the phototoxic efficacy of the probe.
The Scientist's Toolkit
Table: Key Research Reagent Solutions for Mitochondrial and Redox Biology
| Item | Function / Explanation | Example from Context |
|---|---|---|
| Neutral Benzothiazole Probe | A ΔΨm-independent dye for reliable mitochondrial labeling in diseased or depolarized cells. | BTNDP [72] |
| Cationic Mitochondrial Dye | A ΔΨm-dependent control dye for comparative studies and validation of depolarization. | TMRM [6], BTVMP [72] |
| Chemical Uncoupler | A tool to dissipate the proton gradient and collapse ΔΨm, used to test probe dependency. | CCCP (Carbonyl cyanide m-chlorophenyl hydrazone) [72] |
| Thiol-Reactive Probe | A fluorescent tool for detecting and quantifying biothiols like Cysteine or H₂S in live cells. | DNBS-NHBBT [76], HBT-T [77] |
| Integrated Staining Kit | A combination of dyes for multiparametric analysis of cell death, proliferation, and mitochondrial health via flow cytometry. | BrdU, PI, Annexin V, JC-1, CellTrace Violet [28] |
Signaling Pathways and Experimental Workflows
Diagram: Mechanism Overcoming Redistribution Artifacts. This workflow contrasts the failure of traditional potential-dependent probes with the reliable labeling provided by novel benzothiazole-based, potential-independent probes in depolarized mitochondria.
Diagram: Probe Validation Workflow. A step-by-step experimental protocol to determine if a novel mitochondrial probe's accumulation is dependent on or independent of the mitochondrial membrane potential.
Troubleshooting Guides & FAQs
FAQ: Addressing Common Experimental Issues
1. My fluorescent dye signal decreases during time-lapse imaging. Is this always indicative of mitochondrial depolarization?
Not necessarily. Photobleaching from prolonged light exposure or dye leakage from the cell can also cause a signal decrease. To confirm depolarization, include a positive control (e.g., 20 µM CCCP, a protonophore that collapses ΔΨm) in your experiment. A genuine depolarization event will be replicated by the CCCP control, whereas photobleaching will occur uniformly across all samples. Furthermore, ensure you are using the correct imaging mode for your dye; for example, TMRM used in non-quenching mode requires the dye to remain in the bathing solution during imaging to prevent artifactual signal loss from dye efflux [1].
2. When using JC-1, I observe green fluorescence but little to no red fluorescence. What does this mean?
A high green (monomer) to red (J-aggregate) fluorescence ratio is indicative of mitochondrial depolarization. However, this can also occur due to technical issues. First, confirm that the JC-1 concentration and loading time are optimal, as aggregate formation is highly sensitive to dye concentration [1] [41]. Second, validate your results with a positive control like CCCP. Finally, ensure your flow cytometer or microscope filters are correctly set to detect both fluorescence emissions (e.g., ~530 nm for monomers and ~590 nm for aggregates) without bleed-through [41].
3. I see a strong fluorescent signal in my positive control (CCCP/FCCP-treated) cells. Is my experiment failing?
A persistent strong signal after treatment with an uncoupler like CCCP suggests that the dye behavior may not be solely dependent on ΔΨm. Some dyes, particularly the Mitotracker family (e.g., Mitotracker Red CMXRos), contain a thiol-reactive chloromethyl group that covalently binds to mitochondrial proteins. Once bound, their retention is less sensitive to changes in membrane potential [79] [30]. For dynamic assays of ΔΨm, use potentiometric probes like TMRM or TMRE, which exhibit Nernstian distribution and rapidly redistribute upon depolarization [1] [30].
4. How can I be sure that my dye is specifically labeling mitochondria and not other cellular structures?
Dye specificity is a major concern. Cationic dyes are attracted to any negatively charged membrane potential. While healthy mitochondria have the highest potential, other organelles like the endoplasmic reticulum and Golgi apparatus also possess membrane potentials that can weakly attract these dyes [79]. To confirm mitochondrial specificity, perform a co-localization experiment using a second, independent mitochondrial marker, such as a fluorescent protein targeted to the mitochondrial matrix (e.g., mito-GFP) [79]. A high degree of co-localization supports specific mitochondrial labeling.
5. I am getting conflicting results between microscopy and flow cytometry for the same treatment. Why?
This is a common challenge due to the different principles of each technique. Microscopy can resolve subcellular localization and heterogeneity within a single cell, while flow cytometry provides a population-average measurement. A treatment that induces depolarization in only a subset of mitochondria within a cell might be detectable by microscopy but diluted in the overall signal from flow cytometry [6]. Ensure that the dye concentration and loading protocols are optimized for each specific platform, as parameters like TMRM concentration can drastically affect its distribution between mitochondrial sub-compartments [6].
Troubleshooting Guide: Dye Redistribution Artifacts
Problem: In a co-culture experiment, I detect transfer of a mitochondrial dye from donor to recipient cells, but a genetically encoded mitochondrial marker (e.g., mito-GFP) does not transfer. What is happening?
This is a classic signature of dye redistribution artifact, not genuine horizontal mitochondrial transfer (HMT).
- Root Cause: The fluorescent dye itself is transferring between cells, not the entire organelle. Mitotracker and TMRE dyes are lipophilic and can be exchanged between cells via direct contact (e.g., tunneling nanotubes) or through the extracellular medium, independent of whole-mitochondria transfer [79]. The covalently bound Mitotracker dyes may also label other thiol-containing proteins in non-mitochondrial membranes [79].
- Solution:
- Use a Protein-Based Marker: Always corroborate dye-based findings with a genetically encoded, mitochondrially targeted fluorescent protein (e.g., COX8a or TOM20 fused to GFP). The transfer of this protein signal is a more reliable indicator of actual HMT [79].
- Conduct Essential Controls: Include a control with mitochondria-deficient (ρ0) cells. If dye signal is still transferred from these cells, it conclusively demonstrates non-specific dye movement [79].
- Validate with Functional Assays: Couple your imaging with a functional assay, such as measuring the restoration of oxidative phosphorylation in recipient cells that lack mitochondrial function.
Table: Key Differences Between Dye Redistribution and Genuine Mitochondrial Transfer
| Feature | Dye Redistribution Artifact | Genuine Mitochondrial Transfer |
|---|---|---|
| Primary Indicator | Transfer of small molecule dye | Transfer of entire organelle |
| Mito-GFP Transfer | No | Yes |
| Source (ρ0 cells) | Possible | Impossible |
| Functional Rescue | No | Yes (can restore OXPHOS) |
| Inhibition by Cytochalasin D | Often unaffected [79] | Typically inhibited (blocks TNTs) |
Experimental Protocols for Validation
Protocol 1: Validating Dye Specificity and ΔΨm-Dependence by Flow Cytometry
This protocol uses JC-1 and flow cytometry to distinguish specific ΔΨm-dependent staining from non-specific artifacts [41].
Research Reagent Solutions:
- JC-1 Staining Solution: Prepare a working solution containing JC-1 dye, ultrapure water, and staining buffer as per manufacturer's instructions. Warm to 37°C before use [41].
- Carbonyl cyanide m-chlorophenyl hydrazone (CCCP): A potent uncoupler used as a positive control for depolarization. Prepare a 20 µM solution in DMSO [41].
- Ice-cold Staining Buffer: For washing cells to remove unincorporated dye.
Methodology:
- Cell Preparation and Treatment: Harvest and pellet your cells by centrifugation (500 x g for 3 min). Treat an aliquot of cells with 20 µM CCCP for 1 hour as a positive control [41].
- Dye Loading: Resuspend the cell pellets in 0.5 mL of complete medium and 0.5 mL of JC-1 working solution. Incubate for 20 minutes at 37°C in the dark [41].
- Washing: Pellet the cells again and wash twice with 2 mL of ice-cold staining buffer to remove excess dye [41].
- Flow Cytometry Analysis: Resuspend cells in staining buffer and analyze immediately on a flow cytometer.
- Create a dot plot with Forward Scatter (FSC) vs. Side Scatter (SSC) to gate the cell population.
- Create a second dot plot with FITC (530 ± 30 nm, JC-1 monomer) on the X-axis and PE (575 ± 26 nm, JC-1 aggregate) on the Y-axis.
- For healthy, polarized cells, the population will shift to the upper right (high PE, low FITC). Depolarized cells (CCCP-treated) will shift to the lower left (high FITC, low PE). Calculate the MMP as the ratio of red (PE) to green (FITC) fluorescence intensity [41].
JC-1 Flow Cytometry Workflow
Protocol 2: High-Content Microscopy Assay for ΔΨm Using TMRM
This protocol is suitable for high-throughput analysis of ΔΨm in 2D and 3D models using TMRM, a dye with minimal mitochondrial binding and low toxicity [80] [81] [30].
Research Reagent Solutions:
- Tetramethylrhodamine methyl ester (TMRM): A cationic, potentiometric probe. Use low concentrations (e.g., 1–30 nM for non-quenching mode; 50-100 nM for quenching mode) to avoid artifacts and inhibition of the electron transport chain [1] [6].
- MitoTracker Green FM (MTG): A ΔΨm-independent mitochondrial mass marker. Use for morphological reference and normalization [6].
- Oligomycin: An ATP synthase inhibitor that causes hyperpolarization. Use as a control.
- FCCP: A protonophore uncoupler that causes depolarization. Use as a positive control.
Methodology:
- Cell Seeding and Staining: Seed cells in an appropriate multi-well plate for imaging. Prior to treatment, load cells with a low concentration of TMRM (e.g., 10-50 nM) and, if desired, MTG (500 nM) in culture medium for 30 minutes at 37°C [81] [6].
- No-Wash Assay: For a simplified high-throughput screen, the TMRM-containing medium can be replaced with fresh medium without dye, or the assay can be run without washing if the signal-to-noise ratio is acceptable [81] [82]. For precise quantification, washing is recommended.
- Image Acquisition: Acquire images using a high-content or confocal microscope. For TMRM in non-quenching mode, maintain a low concentration of TMRM in the bath during imaging to prevent dye loss [1].
- Image Analysis:
- Use MTG signal to create a mask of mitochondrial morphology.
- Quantify the TMRM fluorescence intensity within the mitochondrial mask.
- Normalize the TMRM intensity to the MTG intensity to account for changes in mitochondrial mass.
- Validate the assay by treating control wells with FCCP (depolarization) and oligomycin (hyperpolarization).
Table: Comparison of Common Mitochondrial Membrane Potential Dyes
| Probe | Best Used For | Key Strengths | Key Limitations & Considerations |
|---|---|---|---|
| TMRM/TMRE | Acute studies; non-quenching mode; high-content microscopy [1] [80]. | Low mitochondrial binding; minimal ETC inhibition; reversible distribution [1] [30]. | Signal is highly concentration-dependent; requires careful optimization [6]. |
| JC-1 | "Yes/No" discrimination of polarization (e.g., apoptosis); flow cytometry [1] [41]. | Ratiometric (Red/Green); internal calibration; sensitive to subtle changes. | Aggregate formation is sensitive to concentration, pH, and ROS; not ideal for kinetics [1]. |
| Rhodamine 123 | Fast acute studies in quenching mode [1]. | Slow permeation allows easier observation of unquenching. | Poor sensitivity in some modes; can have varying fluorescence emissions in a single cell [1] [41]. |
| Mitotracker (e.g., CMXRos) | Fixed-cell imaging; long-term tracking [79] [30]. | Covalent binding allows fixation and permeabilization. | Retention is not fully potential-dependent after binding; can label non-mitochondrial structures [79] [30]. |
The Scientist's Toolkit: Research Reagent Solutions
Table: Essential Reagents for Mitochondrial Membrane Potential Assays
| Item | Function | Example Usage |
|---|---|---|
| TMRM | Potentiometric probe for dynamic ΔΨm measurement. | High-content imaging in non-quenching mode (10-50 nM) [1] [6]. |
| JC-1 | Ratiometric, J-aggregate forming probe for population screening. | Flow cytometry-based apoptosis and depolarization assays [41]. |
| CCCP / FCCP | Protonophore uncouplers; positive controls for depolarization. | Collapse ΔΨm to validate dye response (e.g., 10-20 µM) [41] [30]. |
| Oligomycin | ATP synthase inhibitor; control for hyperpolarization. | Inhibits proton flow through ATP synthase, increasing ΔΨm [83]. |
| MitoTracker Green FM | ΔΨm-independent mitochondrial stain. | Used as a morphological reference to normalize potentiometric dye signal [6]. |
| Mito-GFP (COX8a/TOM20) | Genetically encoded mitochondrial marker. | Validates mitochondrial specificity and rules out dye artifacts in transfer studies [79]. |
Dye Behavior and Common Artifacts
Accurate measurement of the mitochondrial membrane potential (ΔΨm) is fundamental for assessing cellular health, metabolic activity, and drug mechanisms. However, researchers frequently encounter artifacts and inaccuracies stemming from dye redistribution, improper experimental controls, and methodological inconsistencies. This technical support center addresses these challenges by providing standardized protocols, troubleshooting guides, and frequently asked questions to establish reliable reference measurements for your research.
The electrochemical proton gradient across the inner mitochondrial membrane consists of both a membrane potential (ΔΨm) and a pH gradient (ΔpHm). Typically, ΔΨm values range between 150-180 mV, accounting for the majority of the total proton motive force of 180-220 mV [1]. Cationic fluorescent dyes like TMRM, TMRE, Rhod123, and JC-1 accumulate in the mitochondrial matrix in proportion to ΔΨm, but their behavior must be carefully controlled and interpreted to avoid artifacts [1]. This guide synthesizes best practices from current literature to help researchers overcome common pitfalls in ΔΨm measurement.
Research Reagent Solutions: ΔΨm Detection Dyes
Table 1: Common ΔΨm-sensitive fluorescent dyes and their applications
| Probe Name | Primary Applications | Excitation/Emission | Key Strengths | Important Limitations |
|---|---|---|---|---|
| TMRM/TMRE | Slow resolving acute studies; measuring pre-existing ΔΨm (non-quenching mode) [1] | ~548/~573 nm [1] | Lowest mitochondrial binding and electron transport chain (ETC) inhibition; suitable for acute or chronic studies [1] | Fast equilibration makes them less suited to some quenching studies [1] |
| Rhod123 | Fast resolving acute studies (quenching mode) [1] | ~507/~529 nm [1] | Slow permeation means quenching/unquenching changes are easier to detect [1] | Slightly more ETC inhibition and mitochondrial binding than TMRM [1] |
| JC-1 | "Yes/No" discrimination of polarization state (e.g., apoptosis studies) [1] [28] | Monomer: ~514/~529 nm; J-aggregate: ~585/~590 nm [1] | Dual-color, ratiometric assessment of ΔΨm via monomer/aggregate forms [1] | Very sensitive to concentration; aggregate form sensitive to factors other than ΔΨm [1] |
| DiOC6(3) | Flow cytometry studies [1] | ~484/~501 nm [1] | Widely employed for ΔΨm assessment in flow cytometry [1] | Requires very low concentrations (<1 nM) to accurately monitor ΔΨm rather than plasma membrane potential (Δψp) [1] |
Experimental Protocols for Reliable ΔΨm Measurement
Standardized Protocol for Live-Cell ΔΨm Imaging with TMRM
This protocol, adapted from the CeBioND consortium guidelines, provides a robust method for measuring ΔΨm in primary neuronal cultures and other cell types [52].
Reagents and Equipment:
- TMRM (or TMRE) dye solution
- Extracellular buffer (e.g., Krebs-Ringer Buffer)
- Pharmacological uncouplers (FCCP, 1-2 µM) and inhibitors (oligomycin, 1-2 µg/mL)
- Live-cell imaging system with temperature and CO₂ control
- Appropriate filter sets for TMRM (excitation/emission: ~548/~573 nm)
Procedure:
- Cell Preparation: Plate cells on appropriate imaging dishes at optimal density. For primary neurons, use days in vitro (DIV) 10-14 to ensure mature neuronal networks [52].
- Dye Loading: Incubate cells with 1-30 nM TMRM in extracellular buffer for 30 minutes at 37°C. Use the lowest possible concentration that provides adequate signal-to-noise ratio [1].
- Wash and Equilibration: Remove dye-containing solution and wash twice with fresh buffer. Equilibrate for 10 minutes before imaging to allow dye stabilization.
- Image Acquisition: Acquire images using appropriate microscopy settings. For quantitative comparisons, maintain consistent laser power, gain, and exposure times across experiments.
- Validation with Controls:
- Data Analysis: Quantify fluorescence intensity per cell or mitochondrial region of interest. Normalize to baseline or control conditions as appropriate.
Critical Considerations:
- For non-quenching mode, use low TMRM concentrations (1-30 nM) and maintain dye in bath during imaging [1]
- Avoid fixation when using potential-sensitive dyes as they require mitochondrial activity [5]
- Include parallel assays to monitor cell viability and mitochondrial morphology
Flow Cytometry Protocol for Multiparametric ΔΨm Assessment
This integrated workflow allows simultaneous assessment of ΔΨm alongside cell death, proliferation, and cell cycle parameters [28].
Reagents:
- JC-1 dye solution
- Annexin V binding buffer and conjugate
- Propidium iodide (PI)
- CellTrace Violet or similar proliferation dye
- Bromodeoxyuridine (BrdU)
Procedure:
- Cell Staining: Harvest approximately 0.5 million cells per condition and stain with JC-1 according to manufacturer's instructions.
- Multiparametric Staining: Combine with Annexin V/PI for apoptosis detection, BrdU/PI for cell cycle analysis, or CellTrace Violet for proliferation tracking [28].
- Flow Cytometry Analysis: Acquire data using appropriate laser and filter settings for each fluorophore.
- Data Interpretation:
- For JC-1, calculate the ratio of aggregate (red) to monomer (green) fluorescence
- Higher ratios indicate more polarized mitochondria
- Depolarization manifests as decreased red/green ratio [28]
Technical Notes:
- JC-1 is particularly suited for apoptosis studies where a clear "polarized/depolarized" discrimination is needed [1]
- Ensure proper compensation between fluorescence channels to avoid bleed-through artifacts [84]
- This multiparametric approach requires careful titration of all dyes to avoid spectral overlap
Troubleshooting Guides and FAQs
Common ΔΨm Measurement Artifacts and Solutions
Table 2: Troubleshooting common ΔΨm measurement issues
| Problem | Potential Causes | Solutions | Preventive Measures |
|---|---|---|---|
| Unexpected ΔΨm increases | Dye redistribution artifacts; non-protonic charge influences (e.g., Ca²⁺ fluxes) [1] | Measure mitochondrial pH and Ca²⁺ in parallel; validate with multiple dyes | Include controls for ionic disturbances; use complementary assays |
| Poor signal-to-noise ratio | Suboptimal dye concentration; inappropriate imaging settings; photobleaching | Titrate dye concentration; optimize imaging parameters; use antioxidant-containing media | Perform dye titration curve for each cell type; use lowest possible illumination |
| Spectral bleed-through | Fluorophore emission spectra overlap; filter misconfiguration [84] | Sequential scanning; adjust detector slits; choose fluorophores with well-separated spectra | Select dye combinations with minimal spectral overlap; verify with single-label controls |
| Dye leakage from mitochondria | ΔΨm collapse during experiment; improper dye retention | Use fixable structural dyes for fixed samples; image live cells before fixation [5] | Include viability markers; minimize experimental duration |
| Inconsistent results between assays | Methodological differences; cellular heterogeneity; different ΔΨm components measured | Standardize protocols across laboratories; use multiple complementary methods [52] | Follow established consortium guidelines; report detailed methods |
Frequently Asked Questions
Q: Why do I observe mitochondrial hyperpolarization in response to cellular stress when I expected depolarization? A: Unexpected hyperpolarization can occur due to non-protonic charges influencing ΔΨm. For example, Ca²⁺ dumping from mitochondrial and ER stores can cause hyperpolarization that masks the expected depolarization from proton gradient changes. Always measure complementary parameters like mitochondrial pH and Ca²⁺ to interpret these results correctly [1].
Q: How do I determine whether changes in fluorescence intensity represent ΔΨm changes or variations in mitochondrial mass? A: Use a two-dye strategy with a potential-insensitive structural dye (e.g., Mitotracker Green in non-quenching mode) to visualize all mitochondria regardless of function, alongside your ΔΨm-sensitive dye. This approach controls for mitochondrial mass and distribution changes [5].
Q: What is the optimal TMRM concentration for my experiment? A: The optimal concentration depends on your measurement mode. For non-quenching mode (recommended for most steady-state measurements), use 1-30 nM. For quenching mode, use higher concentrations (>50-100 nM). Always use the lowest concentration that provides adequate signal-to-noise ratio for your specific cell type [1].
Q: Can I use these dyes in fixed cells? A: Most potential-sensitive dyes do not work reliably in fixed cells because fixation destroys mitochondrial activity. If you must fix cells, use fixable structural dyes or antibody-based mitochondrial markers (e.g., COX IV, TOMM20) that covalently bind to mitochondrial proteins [5].
Q: How do I validate that my ΔΨm measurements accurately reflect true bioenergetic changes? A: Always include pharmacological controls: FCCP (or other uncouplers) should collapse ΔΨm, serving as a depolarization control, while oligomycin should cause hyperpolarization by inhibiting ATP synthase. These controls verify that your measurements reflect genuine bioenergetic changes [1] [52].
Advanced Technical Considerations
Addressing Spatial Heterogeneity in ΔΨm Measurements
Recent super-resolution microscopy studies reveal that the inner mitochondrial membrane maintains different electrical potentials across its sub-compartments. The cristae membrane (CM) typically shows a higher (more negative) membrane potential (ΔΨC) compared to the inner boundary membrane (ΔΨIBM) [6]. This spatial gradient has functional implications:
- Cristae junctions act as barriers separating these compartments
- Calcium signaling can preferentially hyperpolarize the cristae membranes
- ATP production correlates with these membrane potential gradients [6]
When interpreting bulk ΔΨm measurements, consider that population averages may mask this important spatial heterogeneity. Advanced imaging techniques can resolve these sub-mitochondrial domains, providing more nuanced insights into mitochondrial function.
Correlation with Other Bioenergetic Parameters
For comprehensive assessment, correlate ΔΨm measurements with other bioenergetic parameters:
- Oxygen consumption rate (OCR) using Seahorse or similar platforms
- Mitochondrial NAD(P)H autofluorescence [52]
- ATP production using FRET-based sensors [6]
- Mitochondrial calcium using targeted indicators
This multi-parameter approach provides cross-validation and a more complete picture of mitochondrial function, helping to distinguish true bioenergetic changes from measurement artifacts.
Visual Protocols and Workflows
Integrated Workflow for ΔΨm Quality Control
Artifact Identification and Resolution Pathway
By implementing these standardized protocols, troubleshooting guides, and quality control measures, researchers can establish reliable ΔΨm reference measurements that minimize artifacts and enhance the reproducibility of their findings. Remember that consistent benchmarking against established gold standards is essential for generating meaningful, interpretable data in mitochondrial research.
Conclusion
Mitochondrial membrane potential dye redistribution represents a significant experimental challenge that requires systematic approaches from mechanistic understanding to practical validation. The integration of optimized staining protocols, advanced imaging techniques, and orthogonal validation methods provides a robust framework for artifact mitigation. Future directions should focus on developing ΔΨm-insensitive probes like neutral benzothiazole derivatives for depolarized systems, implementing standardized quality controls across platforms, and establishing comprehensive guidelines for specific treatment contexts. These advances will enhance reliability in fundamental research and accelerate translation in drug discovery, particularly for cancer therapies, neurodegenerative diseases, and metabolic disorders where accurate ΔΨm assessment is critical for understanding therapeutic efficacy and mechanisms.
References
- 1. Mitochondrial membrane potential probes and the proton ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3115691/]
- 2. Mitochondrial membrane potential - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5792320/]
- 3. JC-9 Dye (Mitochondrial Membrane Potential Probe) - FAQs [https://www.thermofisher.com/store/v3/products/faqs/D22421]
- 4. LDS 698: A highly sensitive probe for tracking ... [https://www.sciencedirect.com/science/article/abs/pii/S0006291X25011568]
- 5. What to consider before choosing a stain for mitochondria [https://www.abcam.com/en-us/stories/articles/choosing-a-mitochondrial-stain]
- 6. Implications of mitochondrial membrane potential gradients ... [https://www.nature.com/articles/s41598-024-65595-z]
- 7. Mitochondrial Membrane Potential - an overview [https://www.sciencedirect.com/topics/medicine-and-dentistry/mitochondrial-membrane-potential]
- 8. JC-1 Dye for Mitochondrial Membrane Potential [https://www.thermofisher.com/us/en/home/life-science/cell-analysis/cell-viability-and-regulation/apoptosis/mitochondria-function/jc-1-dye-mitochondrial-membrane-potential.html]
- 9. JC-1 Experiment Common Questions and Solutions [https://www.elabscience.com/resources/cell-function-technical-topics/1845?srsltid=AfmBOorsK4nOOc19qzRI4MRV0n_aA6YP52YjHGNRuto_x033vFyCqjlR]
- 10. Simultaneous Measurement of Individual Mitochondrial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4018156/]
- 11. Is the Mitochondrial Membrane Potential (∆Ψ) Correctly ... [https://www.mdpi.com/1422-0067/23/1/482]
- 12. TMRE & TMRM | Explore Membrane Potentials [https://www.potentiometricprobes.com/tmre-tmrm?srsltid=AfmBOoogiqpEmyJcQsYc-0bs6UnxRHSH3xR1FlU4u_G-UhPH9QBBN301]
- 13. What is the mechanism of staining for potential- ... [https://biotium.com/faqs/what-is-the-mechanism-of-staining-for-potential-independent-mitochondrial-dyes/]
- 14. Interference and Artifacts in High-content Screening - NCBI - NIH [https://www.ncbi.nlm.nih.gov/books/NBK615088/]
- 15. Membrane dynamics are slowed for Alexa594-labeled ... [https://www.sciencedirect.com/science/article/pii/S2667160321000259]
- 16. Mitochondrial Compartmentalization: Emerging Themes in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11008732/]
- 17. as a fundamental switchboard for mitochondrial ion... Cristae junction [https://pubmed.ncbi.nlm.nih.gov/34915234/]
- 18. Identity, structure, and function of the mitochondrial ... [https://www.nature.com/articles/s41418-023-01187-0]
- 19. The Mitochondrial Permeability Transition Pore-Current ... [https://pubmed.ncbi.nlm.nih.gov/37174672/]
- 20. Inner mitochondrial membrane compartmentalization [https://www.sciencedirect.com/science/article/pii/S135727252030011X]
- 21. An automated differential nuclear staining assay for accurate... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8310432/]
- 22. Visualizing Mitophagy with Fluorescent Dyes for Mitochondria and... [https://www.jove.com/t/64647/visualizing-mitophagy-with-fluorescent-dyes-for-mitochondria]
- 23. CRISPR activation and interference screens decode ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9307090/]
- 24. Tetramethylrhodamine, Methyl Ester, Perchlorate (TMRM) [https://www.thermofisher.com/store/v3/products/faqs/T668]
- 25. High-Throughput Microscopy Analysis of Mitochondrial ... [https://www.mdpi.com/2073-4409/12/7/1089]
- 26. Quantitative measurement of mitochondrial membrane ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3448152/]
- 27. Comparative Analysis of the Different Dyes' Potential to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7060866/]
- 28. From proliferation to mitochondrial membrane potential ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11933298/]
- 29. Best Image Award 2025 Winners Announced [https://wms-site.com/alert-on-mitochondria/1352-best-image-award-2025-winners-announced]
- 30. Performance of TMRM and Mitotrackers in mitochondrial ... [https://www.sciencedirect.com/science/article/pii/S0005272823000737]
- 31. High-Throughput Microscopy Analysis of Mitochondrial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10093082/]
- 32. Analysis of the Mitochondrial Using the Cationic... Membrane Potential [https://bio-protocol.org/en/bpdetail?id=3128&type=0]
- 33. Spontaneous Changes in Mitochondrial Membrane Potential ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6762873/]
- 34. JC-1 Dye for Mitochondrial Membrane Potential [https://www.thermofisher.com/tw/zt/home/life-science/cell-analysis/cell-viability-and-regulation/apoptosis/mitochondria-function/jc-1-dye-mitochondrial-membrane-potential.html]
- 35. Probes for Mitochondria—Section 12.2 [https://www.thermofisher.com/us/en/home/references/molecular-probes-the-handbook/probes-for-organelles/probes-for-mitochondria.html]
- 36. Methods for imaging mammalian mitochondrial morphology [https://pmc.ncbi.nlm.nih.gov/articles/PMC6322684/]
- 37. Exploring Mitochondrial in Potential Dyes Research [https://physicscore.com/articles/mitochondrial-potential-dyes-research/]
- 38. Intra- and intercellular distribution of mitochondrial probes and... [https://pubmed.ncbi.nlm.nih.gov/11463164/]
- 39. Resolution in super-resolution microscopy – facts, artifacts ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12148039/]
- 40. A novel super-resolution STED microscopy analysis ... [https://www.sciencedirect.com/science/article/pii/S0167488925000059]
- 41. A Flow Cytometry-based Assay for Measuring Mitochondrial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6126459/]
- 42. A Fully Automated High-Throughput Flow Cytometry ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6055113/]
- 43. Flow Cytometry Troubleshooting Guide [https://www.cellsignal.com/learn-and-support/troubleshooting/flow-cytometry-troubleshooting-guide?srsltid=AfmBOoqQjokgqDDCjTL0mX48ZYWbXruslr-DNoVFxvpUVVIWhUNucXTF]
- 44. Troubleshooting Flow Cytometry Issues [https://www.bosterbio.com/protocol-and-troubleshooting/flow-cytometry-troubleshooting?srsltid=AfmBOoqBHTj_bDsUHRQdKcg3KvZU6iBiS0OzUUv9By71QGPFyQp4H5yO]
- 45. Flow-cytometric monitoring of mitochondrial depolarisation [https://www.sciencedirect.com/science/article/pii/S1011134404001526]
- 46. Expert view: Challenges in flow cytometry [https://www.drugtargetreview.com/article/35131/expert-view-challenges-in-flow-cytometry/]
- 47. Mitochondrial membrane potential and compartmentalized ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12454667/]
- 48. Probes for Mitochondria—Section 12.2 [https://www.thermofisher.com/za/en/home/references/molecular-probes-the-handbook/probes-for-organelles/probes-for-mitochondria.html]
- 49. Relation between mitochondrial membrane potential and ... [https://pubmed.ncbi.nlm.nih.gov/22057568/]
- 50. Mitochondrial electron transport chain, ROS generation ... [https://www.spandidos-publications.com/10.3892/ijmm.2019.4188]
- 51. Analysis of mitochondrial membrane potential, ROS, and ... [https://www.sciencedirect.com/science/article/pii/S1016847825000627]
- 52. Guidelines on experimental methods to assess mitochondrial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5864235/]
- 53. Detection of the Mitochondrial Membrane Potential by ... [https://pubmed.ncbi.nlm.nih.gov/29986516/]
- 54. Guidelines on experimental methods to assess ... [https://www.nature.com/articles/s41418-017-0020-4]
- 55. How and when to measure mitochondrial inner membrane ... [https://www.sciencedirect.com/science/article/abs/pii/S0006349524001760]
- 56. VDAC1 inhibitor DIDS uncouples respiration in isolated ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12556276/]
- 57. Dodecyl and octyl esters of fluorescein as protonophores ... [https://www.sciencedirect.com/science/article/pii/S0005272813001679]
- 58. Mitochondrial Membrane Potential Assay [https://www.sartorius.com/en/applications/life-science-research/live-cell-assays/mitochondrial-membrane-potential]
- 59. Monitoring mitochondrial depolarization with JC-1 and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3695757/]
- 60. JC-1 Experiment Common Questions and Solutions [https://www.elabscience.com/resources/cell-function-technical-topics/1845?srsltid=AfmBOook4k_S2W2nM8c8ftY8Ey3Ux9Fb4PclrJ6xSfIte4kOl8VPhfgO]
- 61. JC-1: Advancing Mitochondrial Membrane Potential Assays i... [https://mito-mturquoise2.com/index.php?g=Wap&m=Article&a=detail&id=10713]
- 62. Drug Discovery Assays Support—Troubleshooting [https://www.thermofisher.com/us/en/home/technical-resources/technical-reference-library/drug-discovery-development-support-center/drug-discovery-assays-support/drug-discovery-assays-support-troubleshooting.html]
- 63. Nonspecific Protein Binding versus Membrane Partitioning: ... [https://www.sciencedirect.com/science/article/abs/pii/S0090955624227719]
- 64. nonspecific protein binding versus membrane partitioning ... [https://pubmed.ncbi.nlm.nih.gov/22711748/]
- 65. JC-1 Experiment Common Questions and Solutions [https://www.elabscience.com/resources/cell-function-technical-topics/1845?srsltid=AfmBOop40EPEZhKkVCK1S_hYYvIxuqkhbMDdHmN-lLv_UeZHp3WbYtw-]
- 66. The Combination of Paraformaldehyde and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8151741/]
- 67. Study Designs for Evaluation of Combination Treatment [https://pmc.ncbi.nlm.nih.gov/articles/PMC8869609/]
- 68. P-glycoprotein expression skews mitochondrial dye ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12213397/]
- 69. Antibody Validation Essentials: Orthogonal Strategy - CST Blog [https://blog.cellsignal.com/hallmarks-of-validation-orthogonal-strategy]
- 70. Systematic literature review reveals suboptimal use of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10239480/]
- 71. use of orthogonal signal correction and PLS-DA [https://www.sciencedirect.com/science/article/pii/S0014579302034762]
- 72. Potential-insensitive AIE-active fluorescent molecule for ... [https://www.sciencedirect.com/science/article/abs/pii/S0143720825007569]
- 73. Cell tracing dyes significantly change single cell mechanics [https://pmc.ncbi.nlm.nih.gov/articles/PMC2698996/]
- 74. Advancements and perspectives on organelle-targeted ... [https://pubs.rsc.org/en/content/articlehtml/2025/sc/d5sc04640h]
- 75. Cyanine dyes in the mitochondria-targeting photodynamic ... [https://www.nature.com/articles/s42004-024-01256-6]
- 76. A new strongly fluorescent benzothiazole-based derivative ... [https://www.sciencedirect.com/science/article/abs/pii/S0143720825000890]
- 77. A benzothiazole derivative-based turn-on fluorescent ... [https://www.sciencedirect.com/science/article/abs/pii/S1386142525012004]
- 78. A benzothiazole-based near-infrared fluorescent probe for ... [https://pubmed.ncbi.nlm.nih.gov/41270614/]
- 79. Exploring the limitations of mitochondrial dye as a genuine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10917768/]
- 80. High-Throughput Microscopy Analysis of Mitochondrial ... [https://pubmed.ncbi.nlm.nih.gov/37048162/]
- 81. Development of a high throughput screening assay for ... [https://pubmed.ncbi.nlm.nih.gov/12230893/]
- 82. Development of a No-Wash Assay for Mitochondrial ... [https://www.sciencedirect.com/science/article/pii/S2472555222079837]
- 83. Measuring mitochondrial membrane potential | The EMBO ... [https://link.springer.com/article/10.1038/s44318-025-00632-9]
- 84. Spectral Bleed-Through Artifacts in Confocal Microscopy [https://evidentscientific.com/en/microscope-resource/knowledge-hub/techniques/confocal/bleedthrough]