The Protonmotive Force Decoded: Unraveling the Critical Interplay Between Mitochondrial Membrane Potential and pH Gradient
This article provides a comprehensive analysis of the mitochondrial protonmotive force (PMF), the essential bioenergetic gradient composed of the mitochondrial membrane potential (ΔΨm) and the pH gradient (ΔpH).
The Protonmotive Force Decoded: Unraveling the Critical Interplay Between Mitochondrial Membrane Potential and pH Gradient
Abstract
This article provides a comprehensive analysis of the mitochondrial protonmotive force (PMF), the essential bioenergetic gradient composed of the mitochondrial membrane potential (ΔΨm) and the pH gradient (ΔpH). Tailored for researchers, scientists, and drug development professionals, we dissect the fundamental principles, state-of-the-art measurement methodologies, common pitfalls in experimental practice, and advanced validation techniques. By synthesizing foundational knowledge with recent breakthroughs—including the discovery of spatially distinct membrane potentials within mitochondrial sub-compartments—this review serves as a critical resource for advancing studies in metabolism, neurodegeneration, and cardiovascular disease, and for informing the development of targeted therapeutic strategies.
The Energetic Core: Defining the Protonmotive Force and Its Components
The chemiosmotic theory, formulated by Peter Mitchell in 1961, represents a foundational pillar of modern bioenergetics [1]. This revolutionary theory provided the first comprehensive explanation for how cells convert energy from one form to another, specifically how energy derived from electron transfer through the electron transport chain (ETC) is harnessed to synthesize adenosine triphosphate (ATP) [1]. At its core, the theory proposes that energy from substrate oxidation is used to pump protons across an impermeable membrane, creating an electrochemical gradient known as the protonmotive force (PMF) [2]. This force subsequently drives ATP synthesis through a specialized molecular machine—the FoF1-ATP synthase [1]. The theory's basic postulates include: (1) an electron transport chain that provides energy for H+ transfer across the membrane; (2) ATP synthase that synthesizes ATP through H+ translocation; and (3) impermeability of the membrane to ionic species including protons to maintain the gradient [1]. For decades, this delocalized coupling model has served as the central paradigm for understanding energy conversion in mitochondria, chloroplasts, and bacteria, though recent advances have prompted significant refinements to the original framework.
Fundamental Principles and Components
The Protonmotive Force and Its Components
The protonmotive force (PMF) represents the central energy intermediate in the chemiosmotic theory. It is an electrochemical gradient consisting of two primary components: the electrical potential (ΔΨ) resulting from charge separation across the membrane, and the chemical potential (ΔpH) arising from differences in proton concentration [2] [3]. Under physiological conditions, the mitochondrial membrane potential (ΔΨm) typically ranges between -160 mV to -180 mV (negative inside), while the pH gradient (ΔpH) is approximately 0.4 units (alkaline inside) [3]. The electrical component contributes approximately 75-80% of the total PMF, while the chemical component contributes the remaining 20-25% [3]. This relationship exists because the membrane potential generates a driving force equivalent to a 1000-fold difference in proton concentration across the membrane, whereas the ΔpH of 0.4 units corresponds to only a 2.5-fold difference in proton concentration [3]. The total PMF is expressed in millivolts and can be calculated using the formula: PMF = ΔΨ - 60ΔpH at 37°C, where both components are additive in driving proton回流 through ATP synthase [2].
Table 1: Components of the Protonmotive Force in Mitochondria Under Physiological Conditions
| Component | Description | Typical Magnitude | Contribution to PMF |
|---|---|---|---|
| ΔΨ (Electrical Gradient) | Voltage difference across inner mitochondrial membrane | -160 to -180 mV | ~75-80% |
| ΔpH (Chemical Gradient) | Proton concentration gradient across membrane | ~0.4 pH units | ~20-25% |
| Total PMF | Combined electrochemical proton gradient | ~200 mV | 100% |
Key Molecular Complexes
The generation and utilization of the PMF involves several sophisticated molecular machines embedded in the inner mitochondrial membrane. Complex I (NADH:ubiquinone oxidoreductase), Complex III (cytochrome bc1 complex), and Complex IV (cytochrome c oxidase) function as proton pumps that couple electron transfer to vectorial proton translocation from the matrix to the intermembrane space [1] [3]. Complex IV (cytochrome c oxidase), for instance, receives electrons generated during cellular respiration and uses them to reduce molecular oxygen to water, while simultaneously translocating protons across the membrane [4]. Recent studies on cytochrome c oxidase have revealed that proton uptake is not affected by small pH gradients (<0.7 pH units) but decays exponentially for larger gradients, providing quantitative insights into the enzyme's function against electrochemical gradients [4].
The FoF1-ATP synthase (Complex V) is the central enzyme that harnesses the PMF for ATP production [1]. This remarkable molecular motor consists of two functionally distinct domains: the membrane-embedded Fo portion that facilitates proton translocation, and the catalytic F1 portion that synthesizes ATP [1] [2]. The direction of rotation of ATP synthase determines its function: clockwise rotation drives ATP synthesis at the expense of PMF, while counterclockwise rotation hydrolyzes ATP to generate ΔΨm [2]. To prevent wasteful ATP hydrolysis when ΔΨm is low, mitochondria express an inhibitory factor 1 (IF1) that hinders counter-clockwise rotation by interacting with the F1 portion [2].
Contemporary Challenges and Updates to the Theory
Controversies and Limitations of the Classical Model
Despite its widespread acceptance and explanatory power, the chemiosmotic theory has faced persistent controversies and challenges since its inception [1]. A significant limitation concerns the poor correlation observed between bulk-phase measurements of membrane potential and ATP synthesis yield [1]. This discrepancy has led researchers to question whether delocalized proton gradients across the entire membrane adequately explain the efficiency of energy coupling in oxidative phosphorylation [1]. Additionally, the physical impossibility of free proton osmosis presents a fundamental challenge to the classical model, as protons are quantum particles that quickly bind to water molecules forming hydronium ions (H3O+) rather than existing as free ions [1]. Any free protons in the membrane would be rapidly drained by the aqueous phase, releasing solvation energy to the detriment of the membrane [1]. Moreover, free protons exhibit destructive force on biological membranes, necessitating specialized proton transporters like the potential-dependent proton pump Hv1 that are specifically designed to prevent this damaging effect [1].
Emerging Evidence for Localized Coupling and Proton Currents
Recent structural and biophysical evidence has prompted substantial updates to the original chemiosmotic framework. Advanced technologies using fluorescence indicators to track proton movements have revealed that proton translocation is frequently lateral rather than transversal with respect to the coupling membrane [1]. This suggests that protons may never actually reside in the bulk aqueous phase but instead accumulate on the membrane surface or move through specialized channels within the membrane itself [1]. A groundbreaking study using green fluorescent protein as a pH indicator inserted into respiratory complex III and the Fo moiety of ATP synthase in HeLa cells provided direct evidence for localized coupling rather than delocalized chemiosmosis [1]. This localized coupling model suggests that proton transfer occurs through restricted pathways that directly connect proton pumps to ATP synthase, potentially explaining the observed efficiency of energy transduction that exceeds predictions based on bulk-phase proton gradients.
The concept of proton currents, drawing on the proton migration mechanism described two centuries ago by Theodor von Grotthuss, is now gaining traction as a possible explanation for efficient proton movement without dissipation into the bulk phase [1]. In this model, proton charge diffuses through hydrogen-bonded networks within the membrane or along its surface, avoiding the energetic cost of releasing individual protons into solution [1]. Heberle et al. reported findings that reconcile delocalized and localized models by demonstrating "proton migration along the membrane surface and retarded surface to bulk transfer" [1]. This mechanism allows for both localized energy transduction and communication across the membrane surface.
Table 2: Classical vs. Updated Chemiosmotic Theory Concepts
| Aspect | Classical Chemiosmotic Theory | Updated/Modern Perspective |
|---|---|---|
| Proton Movement | Transversal across bulk aqueous phase | Lateral, along membrane surface or through structured pathways |
| Coupling Mechanism | Delocalized bulk-phase gradients | Localized coupling between closely associated complexes |
| Proton Physical Form | Free protons or hydronium ions | Proton currents/charge diffusion via Grotthuss mechanism |
| Energy Transmission | Through bulk electrochemical gradient | Through structured pathways with limited bulk-phase exchange |
| Membrane Properties | Impermeable barrier to ions | Active participant with proton capacitor properties |
Methodologies for Investigating Chemiosmotic Parameters
Measuring Mitochondrial Membrane Potential
Accurate measurement of mitochondrial membrane potential (ΔΨm) is crucial for studying chemiosmotic processes. The JC-1 fluorescent dye represents a widely used approach for assessing ΔΨm [5]. This cationic dye accumulates within mitochondria in a membrane potential-dependent manner, exhibiting a fluorescence shift from green (~529 nm) at low concentrations to red (~590 nm) as it forms aggregates at higher concentrations in energized mitochondria [5]. The experimental protocol involves administering JC-1 directly to cell culture media at a final concentration of 10 μM followed by incubation for 10 minutes at 37°C in the dark [5]. After washing with PBS, fluorescence is measured at 550/615 nm (excitation/emission) for red aggregates and 489/535 nm for green monomers [5]. The ratio of red to green fluorescence provides a quantitative measure of ΔΨm that is independent of mitochondrial number [5]. Alternative potentiometric dyes include tetramethylrhodamine methyl ester (TMRM), which is used according to similar principles [6].
Advanced imaging techniques now enable researchers to study mitochondrial membrane potential with unprecedented spatial and temporal resolution. Super-resolution microscopy techniques such as STED (stimulated emission depletion) and STORM (stochastic optical reconstruction microscopy) can visualize individual cristae and monitor dynamic changes in membrane potential across sub-mitochondrial compartments [7]. These approaches have revealed that MMP is not uniform across a single mitochondrion, challenging the traditional view of homogeneous energy distribution [3]. For tissue-level and organ-level imaging, techniques like MRI offer non-invasive methods for investigating mitochondrial dysfunction in disease states [7].
Monitoring pH Gradients and Proton Dynamics
Real-time imaging of mitochondrial matrix pH provides crucial insights into proton gradient dynamics and their regulation. A recent innovative study employed a fluorescent pH probe targeted to the mitochondrial matrix to investigate how H+ fluxes across the inner mitochondrial membrane are regulated by the ADP/ATP carrier (AAC) and ATP synthase [8]. The experimental approach revealed that activation of AAC-dependent H+ transport by the mitochondrial uncoupler BAM15 causes matrix acidification followed by a re-alkalization phase mediated by reversed activity of ATP synthase [8]. Similar re-alkalization and ATP synthase reversal occurred after acidification caused by inhibition of the electron transport chain [8]. This methodology demonstrated a functional interaction between AAC and ATP synthase in controlling H+ fluxes, suggesting these proteins work in concert to maintain proton homeostasis.
For studying proton uptake kinetics in specific respiratory complexes, single-proteoliposome assays provide high-resolution data. This approach has been applied to cytochrome c oxidase to follow proton uptake of individual enzymes operating against well-defined pH gradients [4]. Measurements reveal that proton uptake is not affected by small pH gradients (<0.7 pH units) but decays exponentially for larger gradients [4]. Furthermore, a linear dependence of substrate concentration on proton uptake rate is observed over more than three orders of magnitude [4]. These surprisingly simple scaling laws provide quantitative constraints for models of proton coupling in the respiratory chain.
Regulatory Mechanisms and Functional Interactions
Coordination Between ADP/ATP Carrier and ATP Synthase
Recent research has revealed a sophisticated functional interaction between the ADP/ATP carrier (AAC) and ATP synthase in regulating proton distribution across the inner mitochondrial membrane [8]. The AAC performs the electrogenic exchange of ATP4− for ADP3−, resulting in a net charge transfer that depends on ΔΨm [2]. Real-time pH imaging demonstrates that activation of AAC-dependent H+ transport induces matrix acidification followed by a re-alkalization phase mediated by reversed activity of ATP synthase [8]. This coupling suggests that H+ influx via AAC stimulates ATP synthase to operate in reverse, effectively functioning as an ATP hydrolase to pump protons back out of the matrix [8]. This regulatory interaction helps maintain appropriate H+ distribution across the membrane and may prevent excessive acidification that could damage mitochondrial components.
The discovery that strong protonophoric activity independent of AAC suppresses both the re-alkalization phase and the reverse action of ATP synthase indicates the need for strict control of H+ flux through the inner mitochondrial membrane [8]. This coordinated regulation between AAC and ATP synthase represents a sophisticated feedback mechanism that goes beyond the original chemiosmotic theory, suggesting that specific protein-protein interactions and localized signaling help optimize energy transduction efficiency under varying physiological conditions.
Diagram 1: Regulatory interactions between AAC and ATP synthase in maintaining proton homeostasis. The diagram shows how matrix acidification resulting from AAC activity can trigger reverse operation of ATP synthase to re-alkalize the matrix.
Mitochondrial Membrane Potential in Quality Control and Signaling
Beyond its canonical role in ATP synthesis, mitochondrial membrane potential serves as a critical regulator of mitochondrial quality control and cellular signaling [3]. Reduced MMP acts as a clear signal for mitophagy, the selective elimination of dysfunctional mitochondria [3]. The process begins when diminished MMP leads to accumulation of PINK1 on the mitochondrial surface, which recruits Parkin and LC3 to mark the organelle for degradation [3]. Mitochondrial fission and fusion events dynamically remodel the network, with MMP of daughter mitochondria determining their fate—fragments with higher MMP typically re-fuse with the network, while those with lower MMP are targeted for destruction [3].
MMP also regulates protein import into mitochondria, as most mitochondrial proteins synthesized in the cytosol carry positively charged targeting signals that are pulled into the matrix by the electrical driving force of MMP [3]. This import dependence on potential may reflect local differences in mitochondrial function and composition, potentially helping to sort mitochondrial fragments toward either biogenesis or degradation pathways [3]. Additionally, emerging evidence indicates that MMP facilitates metabolic specialization by influencing the activity of metabolic enzymes such as pyrroline-5-carboxylate synthase (P5CS), which forms filamentous assemblies under elevated MMP to drive reductive biosynthesis [3]. This dynamic partitioning enables the emergence of specialized mitochondrial subpopulations tailored to specific metabolic demands, particularly evident in cancer cells where augmented substrate production supports rapid proliferation [3].
Experimental Models and Research Applications
Minimal Protocell Systems for Studying Primitive Chemiosmosis
Simplified experimental systems have provided remarkable insights into the evolutionary origins and fundamental requirements of chemiosmotic energy coupling. Recent research demonstrates that fatty acid membranes—potential precursors to modern phospholipid bilayers—can maintain sufficient proton gradients to drive ATP synthesis by ATP synthase under the steep pH and temperature gradients observed in hydrothermal vent systems [9]. This finding challenges the traditional view that complex phospholipid membranes are absolutely required for energy transduction and suggests that ancestral ATP synthase could harness naturally formed geochemical proton gradients before the evolution of specialized proton pumps and modern membrane biogenesis machinery [9]. The experimental approach involves embedding ATP synthase in fatty acid vesicle membranes and demonstrating light-driven ATP production, providing a functional model for intermediate stages in the evolution of chemiosmosis during protocellular stages [9].
The Scientist's Toolkit: Key Research Reagents and Methods
Table 3: Essential Research Reagents and Methods for Investigating Chemiosmotic Parameters
| Reagent/Method | Application | Key Features | Experimental Considerations |
|---|---|---|---|
| JC-1 Dye | Measurement of mitochondrial membrane potential | Dual-emission potential-sensitive dye; forms aggregates at high MMP | Ratio metric measurement (red/green) independent of mitochondrial density [5] |
| Tetramethylrhodamine Methyl Ester (TMRM) | Quantitative assessment of ΔΨm | Cationic potentiometric dye; distribution follows Nernst equation | Requires careful calibration; suitable for both fluorescence imaging and flow cytometry [6] |
| Single-Proteoliposome Assay | Study proton uptake kinetics of respiratory complexes | Enables observation of individual enzyme molecules | Reveals proton uptake unaffected by <0.7 pH unit gradients [4] |
| Targeted pH Probes | Real-time monitoring of mitochondrial matrix pH | Genetically encoded or chemically targeted fluorescent proteins | Enables observation of matrix pH dynamics and protein interactions [8] |
| Fatty Acid Vesicles | Study membrane biophysics and primitive chemiosmosis | Models early evolutionary membranes | Demonstrates ATP synthesis maintenance without complex phospholipids [9] |
| Super-resolution Microscopy (STED/STORM) | Sub-mitochondrial localization and dynamics | Resolution beyond diffraction limit (~20-100 nm) | Visualizes individual cristae, protein distributions [7] |
Diagram 2: Comprehensive experimental workflow for investigating chemiosmotic parameters, integrating multiple methodological approaches from membrane potential measurements to structural analysis.
The chemiosmotic theory continues to serve as the foundational framework for understanding biological energy conversion, though significant refinements have emerged from recent research. Evidence for localized coupling, proton currents, and functional protein complexes that optimize energy transduction has supplemented the original delocalized chemiosmosis model [1]. The sophisticated regulatory interactions between the ADP/ATP carrier and ATP synthase demonstrate a level of control beyond simple bulk-phase proton gradients [8]. Meanwhile, the recognition that mitochondrial membrane potential functions not only in ATP synthesis but also in quality control, protein import, metabolic specialization, and cellular signaling has expanded our understanding of its physiological roles [3].
Future research directions will likely focus on resolving the precise physical mechanisms of proton movement along membranes and through protein complexes, potentially incorporating quantum effects that influence proton transfer [1]. The application of advanced structural techniques such as cryo-electron microscopy and super-resolution fluorescence imaging will provide atomic-level insights into the conformational changes that couple proton movement to ATP synthesis [7] [10]. Additionally, investigating how mitochondrial membrane potential is compartmentalized within individual organelles and how this heterogeneity regulates mitochondrial function represents a promising frontier [3]. As our understanding of these fundamental processes deepens, so too will our ability to intervene therapeutically in the numerous diseases associated with mitochondrial dysfunction, from neurodegenerative disorders to cancer and metabolic syndromes [3]. The continued evolution of the chemiosmotic theory underscores its enduring value as a scientific framework that adapts to incorporate new evidence while maintaining its explanatory power for one of biology's most essential processes.
The protonmotive force (pmF) is the central energy transducer in oxidative phosphorylation, coupling the energy from nutrient oxidation to the production of ATP [11] [12]. According to Peter Mitchell's chemiosmotic theory, universally accepted today, the electron transport chain (ETC) acts as a redox-driven proton pump, generating an electrochemical proton gradient across the mitochondrial inner membrane [11] [12]. This gradient, the pmF, consists of two primary components: the electrical potential difference (ΔΨm), resulting from charge separation, and the chemical proton gradient (ΔpH), resulting from a difference in proton concentration [11] [13]. The total pmF, expressed in millivolts (mV), is described by the equation: Δp = ΔΨ - ZΔpH, where Z is a constant (~59 mV/pH unit at 37°C) that converts the pH difference into millivolts [13] [12]. While ΔΨm and ΔpH are thermodynamically interconvertible parts of the same protonic potential, they play distinct and often specialized roles in mitochondrial bioenergetics, ion transport, and cellular signaling. This whitepaper deconstructs the generation, regulation, and functional specialization of these two components within the context of modern mitochondrial research, providing a framework for understanding their complex interplay in health and disease.
Quantitative Composition and Thermodynamic Relationship
The contributions of ΔΨm and ΔpH to the total protonmotive force are not fixed but vary depending on the tissue, metabolic state, and external conditions. However, a clear quantitative pattern emerges from experimental data.
Table 1: Quantitative Contributions of ΔΨm and ΔpH to the Total Protonmotive Force
| System/Condition | ΔΨm Contribution | ΔpH Contribution | Total pmF (Δp) | Key Measurement Method | Citation |
|---|---|---|---|---|---|
| General Animal Mitochondria | ~80% (∼160-180 mV) | ~20% (ΔpH ~0.5 units) | ∼200 mV | TPP+ electrode, fluorescent dyes | [11] [14] |
| HeLa Cells (at 37°C) | Dominant | ΔpHm ∼0.45 units | Not specified | mitochondrially-targeted SypHer | [15] |
| E. coli (at pH 7.5) | ∼150-200 mV | Near zero (ΔpH ~0.2-0.3) | ∼200 mV | Ion-selective electrodes | [13] |
The balance between these two components is dynamically regulated. The mitochondrial matrix is alkaline (pH ~7.8-8.0) compared to the more acidic intermembrane space (pH ~6.9-7.0) [14] [15]. The low H+-buffering power of the mitochondrial matrix (∼5 mM) compared to the cytosol (∼20 mM) makes matrix pH and ΔpH more susceptible to fluctuations during metabolic shifts, such as cytosolic calcium elevations [15]. The following diagram illustrates the fundamental structure of the pmF and its components.
Diagram 1: The two-component structure of the protonmotive force.
Generation and Functional Specialization of ΔΨm and ΔpH
Distinct Roles in Energy Transduction and Transport
The primary function of the total pmF is to drive ATP synthesis via the F1Fo-ATP synthase as protons flow back into the matrix [2] [11]. However, ΔΨm and ΔpH have developed distinct functional specializations beyond this core role, acting as selective drivers for different cellular processes.
ΔΨm as a Driver for Electrogenic Transport: The electrical field of the ΔΨm (negative inside) provides the dominant driving force for the transport of cations, such as Ca2+, Fe2+, and K+, into the mitochondrial matrix [2] [16]. This is critical for regulating metabolism, biogenesis of Fe-S clusters, and ion homeostasis [2]. Furthermore, ΔΨm is essential for the electrogenic exchange of cytosolic ATP4− for mitochondrial ADP3− by the adenine nucleotide translocator (ANT), a process that consumes one net negative charge per exchange cycle [2] [14].
ΔpH as a Driver for Electroneutral Exchange: The chemical gradient of ΔpH is the primary driver for the transport of electroneutral species and metabolites. Phosphate (Pi) is imported into the matrix in symport with a H+ (or in exchange for OH−), a process powered directly by ΔpH [15]. Similarly, the export of lactate and pyruvate can occur via H+-coupled symporters. This specialization ensures efficient substrate availability for oxidative metabolism.
Differential Roles in Quality Control and Signaling
The functional divergence of the two pmF components extends to mitochondrial quality control and retrograde signaling to the nucleus.
ΔΨm in Mitochondrial Quality Control: A sustained loss of ΔΨm is a well-recognized signal for targeting dysfunctional mitochondria for elimination via mitophagy [2] [16]. Conversely, recent research indicates that chronic mitochondrial hyperpolarization (elevated ΔΨm) can also have profound effects, including remodeling of the nuclear epigenome. Hyperpolarized mitochondria have been linked to nuclear DNA hypermethylation and altered gene expression, surprisingly mediated by phospholipid remodeling rather than redox or metabolic changes [17].
ΔΨm as a Retrograde Signal: Beyond quality control, ΔΨm acts as a key retrograde signal to the nucleus. A decrease in ΔΨm, experimentally induced by uncouplers like BAM15 or CCCP, triggers a delay in the G1-to-S phase transition of the cell cycle in both yeast and mammalian cells [18]. This demonstrates that ΔΨm is a proximal signal for mito-cellular communication, regulating cell cycle progression independent of ATP levels or reactive oxygen species (ROS) [18].
Methodologies for Experimental Dissection
Disentangling the individual contributions of ΔΨm and ΔpH to biological processes requires specific pharmacological tools and measurement techniques. The following table outlines key reagents used in this research.
Table 2: Research Reagent Solutions for Dissecting ΔΨm and ΔpH
| Reagent / Tool | Primary Target / Function | Experimental Effect on pmF Components | Key Application |
|---|---|---|---|
| Nigericin | K+/H+ exchanger ionophore | Decreases ΔpH; Increases ΔΨm (compensatory) | To isolate effects of ΔΨm by collapsing ΔpH [19] |
| Valinomycin | K+ ionophore | Decreases ΔΨm; Increases ΔpH (compensatory) | To isolate effects of ΔpH by collapsing ΔΨm [19] |
| Oligomycin | ATP synthase (Complex V) inhibitor | Increases ΔΨm (in coupled mitochondria) | To inhibit pmF consumption, study reverse mode ATPase [14] |
| Protonophores (FCCP, CCCP, BAM15) | H+ ionophores | Dissipates both ΔΨm and ΔpH | To uncouple OXPHOS, study maximum respiration [14] [18] |
| TMRE / TMRM | Fluorescent cationic dyes | Accumulates in matrix dependent on ΔΨm | Quantitative measurement of ΔΨm in cells and isolated mitochondria [17] [18] |
| SypHer / BCECF | Genetically encoded / chemical pH indicators | Fluorescence sensitive to local pH | Ratiometric measurement of matrix pH (pHin) and ΔpH [15] [19] |
The experimental workflow for manipulating and measuring these components often involves a combination of these reagents to isolate specific effects. A pivotal application is determining the dominant driver behind processes like reactive oxygen species (ROS) production. For instance, to test whether succinate-driven reverse electron transport (RET)-induced ROS is more dependent on ΔΨm or ΔpH, researchers can use nigericin to decrease ΔpH while increasing ΔΨm, and valinomycin to decrease ΔΨm while increasing ΔpH [19]. Studies using this approach in guinea pig brain and heart mitochondria have concluded that ΔΨm is the dominant factor controlling RET-driven ROS production, with absolute pH values having a greater influence than ΔpH itself [19]. The following diagram summarizes a logical workflow for such an investigation.
Diagram 2: A logical workflow for determining the dominant pmF component in a biological process.
Pathophysiological Consequences and Therapeutic Implications
Sustained deviations in the normal homeostasis of ΔΨm and ΔpH are linked to pathology. While mitochondrial depolarization is a well-known trigger for cell death, chronic hyperpolarization is increasingly recognized as a deleterious signal.
Hyperpolarization and Disease: Elevated resting ΔΨm has been documented in pathologies such as pulmonary hypertension, glioblastoma, and ovarian cancer [17]. This hyperpolarization can facilitate excessive mitochondrial calcium uptake and promote ROS production, creating a permissive environment for tumorigenesis and cellular dysfunction [17] [19]. The discovery that hyperpolarization can induce nuclear DNA hypermethylation through phospholipid remodeling provides a novel mechanistic link between mitochondrial bioenergetics and epigenetic regulation in disease [17].
Therapeutic Targeting: The sensitivity of RET-driven ROS production to minor decreases in ΔΨm offers a therapeutic strategy. A 10% decrease in ΔΨm can lead to a 90% reduction in succinate-driven ROS production [19]. This suggests that mild, controlled uncoupling or modulation of ion channels that slightly dissipate ΔΨm could be beneficial in conditions like ischemia-reperfusion injury, where RET is a major source of damaging oxidative stress [19].
The Scientist's Toolkit: Key Methodological Considerations
Accurate measurement of ΔΨm and ΔpH is technically challenging and requires careful experimental design to avoid common artifacts.
Principles of ΔΨm Measurement: Fluorescent cationic dyes like TMRE and TMRM are widely used, but their signals must be interpreted with caution. A key principle is that ΔΨm has a low sensitivity and specificity for reporting changes in OXPHOS activity in coupled mitochondria [14]. This is because the ETC can compensate for increased pmF consumption (e.g., during elevated ATP demand) by increasing electron flow, thereby maintaining a stable ΔΨm. Therefore, a constant ΔΨm does not preclude significant changes in mitochondrial respiration and ATP turnover. Complementary measurements of oxygen consumption rate are essential for a complete bioenergetic profile [14].
Measuring ΔpH and Matrix pH: The direct dynamic measurement of ΔpH in living cells has become possible with tools like the genetically encoded, mitochondrially-targeted pH sensor SypHer [15]. These measurements revealed that the mitochondrial matrix pH is around 7.6 with a ΔpHm of approximately 0.45 in HeLa cells at 37°C, which is lower than earlier estimates [15]. Such tools are crucial for understanding how cytosolic pH shifts, for instance during calcium signaling, are transmitted to and buffered by mitochondria [15].
The protonmotive force is not a monolithic entity but a composite of two dynamically regulated components, ΔΨm and ΔpH. While thermodynamically equivalent, they have evolved distinct functional specializations: ΔΨm serves as the primary driver for electrogenic cation transport and a key retrograde signal, whereas ΔpH facilitates the electroneutral exchange of critical metabolites and buffers the mitochondrial matrix. Their balanced regulation is essential for cellular health, with both hyperpolarization and depolarization of ΔΨm linked to pathological outcomes. Future research, leveraging increasingly sophisticated ionophores, fluorescent probes, and genetic models, will continue to deconstruct the nuanced roles of ΔΨm and ΔpH, offering novel therapeutic avenues for a range of diseases rooted in mitochondrial dysfunction.
The proton motive force (PMF) is the cornerstone of oxidative phosphorylation, serving as the primary energy reservoir that drives adenosine triphosphate (ATP) synthesis in mitochondria. This electrochemical gradient across the inner mitochondrial membrane (IMM) consists of two distinct components: a chemical potential from the proton concentration gradient (ΔpH) and an electrical potential from the charge separation across the membrane (ΔΨm). Within the context of ongoing mitochondrial research, a fundamental question persists: why does ΔΨm constitute the dominant share of the total PMF? This whitepaper provides a comprehensive technical analysis of the quantitative contributions of ΔΨm and ΔpH to the total PMF, synthesizing current biochemical principles, experimental data, and methodological approaches relevant to researchers and drug development professionals investigating mitochondrial bioenergetics. We examine the underlying biophysical reasons for this disparity in contribution, detail cutting-edge measurement techniques, and explore the functional implications for cellular health and disease, providing a foundational resource for thesis-driven research in this domain.
The Biochemical Composition of the Proton Motive Force
The PMF (Δp) is quantitatively defined by the equation: Δp = ΔΨm - (2.303RT/F)(ΔpH)
Where ΔΨm is the mitochondrial membrane potential in millivolts (mV), ΔpH is the transmembrane pH gradient, R is the gas constant, T is the absolute temperature, and F is the Faraday constant. The term 2.303RT/F converts the pH gradient into millivolts, approximating to 60 mV at 37°C. Thus, the equation simplifies to: Δp = ΔΨm - 60(ΔpH) [20]
The negative sign indicates that the matrix is negative and alkaline relative to the intermembrane space. The total PMF is a sum of the electrical (ΔΨm) and chemical (ZΔpH, where Z ≈ 60) components. Extensive research confirms that under physiological conditions, the electrical component, ΔΨm, constitutes the majority of the total PMF. In resting coupled mitochondria, the ΔΨm is typically measured between -120 and -180 mV, while the ΔpH is equivalent to approximately -0.5 to -1.0 pH units (contributing roughly -30 to -60 mV) [2] [20]. This translates to ΔΨm contributing approximately 70-80% of the total proton motive force [2] [14].
Table 1: Quantitative Breakdown of PMF Components in a Model Cell Type (e.g., Cultured Rat Cortical Neurons)
| Parameter | Value | Contribution to Total PMF (Δp) | Measurement Method |
|---|---|---|---|
| Mitochondrial Membrane Potential (ΔΨm) | -139 mV | ~80% | TMRM fluorescence, calibrated |
| pH Gradient (ΔpH) | ~0.9 units | ~20% | SNARF-1 ratioed fluorescence |
| Total Proton Motive Force (Δp) | ~ -193 mV | 100% | Calculated: ΔΨm - 60(ΔpH) |
The dominance of ΔΨm is not merely a static observation but has profound functional implications. The large electrical gradient is a more efficient energy transducer for the ATP synthase and provides the major driving force for the electrophoretic transport of ions and metabolites across the IMM, including the critical exchange of ATP(^{4-}) for ADP(^{3-}) by the adenine nucleotide translocase (ANT) [2].
Experimental Quantification of PMF Components
Simultaneous Measurement of ΔΨm and ΔpH
A definitive protocol for quantifying both components of the PMF in intact cells involves co-loading with potentiometric and pH-sensitive fluorescent probes, followed by confocal microscopy. This method allows for the direct and simultaneous measurement of ΔΨm and ΔpH within individual mitochondria of living cells [20].
Detailed Experimental Protocol:
- Cell Culture and Loading: Cells (e.g., adult rabbit cardiac myocytes) are cultured on appropriate dishes. To load the dyes, cells are incubated with 5 μM SNARF-1-AM (a pH-sensitive dye) for 45 minutes at 37°C in culture medium. Subsequently, a potentiometric dye such as Tetramethylrhodamine Methyl Ester (TMRM) is added at a low, non-quenching concentration (e.g., 10-50 nM) for an additional 30 minutes.
- Image Acquisition: Loaded cells are washed and imaged in a suitable buffer (e.g., Krebs-Ringer-HEPES buffer) on a confocal microscope stage. TMRM is excited, and its emission is collected to reflect ΔΨm. For SNARF-1, ratioed fluorescence emissions (e.g., 584 nm vs. >620 nm) upon excitation at 568 nm are used to estimate pH.
- Calibration: The SNARF-1 signal is calibrated against an in situ pH calibration using buffers of known pH and ionophores (e.g., nigericin and high K+) to clamp intracellular pH. The TMRM signal is calibrated using a separate biophysical model that accounts for plasma membrane potential, mitochondrial volume, and dye binding to convert fluorescence intensities into absolute millivolt values [21].
- Data Analysis: The values for ΔΨm (in mV) and ΔpH (in pH units) are extracted from the fluorescence signals. The total PMF (in mV) is then calculated using the standard equation: Δp = ΔΨm - 60(ΔpH). An example measurement in cardiac myocytes yielded a ΔΨm of -100 mV and a ΔpH of 0.9 units, resulting in a total Δp of at least -140 mV [20].
This workflow and the relationship between the measured parameters are summarized in the diagram below.
Diagram 1: Workflow for simultaneous ΔΨm and ΔpH measurement.
Advanced Spatial Analysis of Membrane Potential
Recent super-resolution microscopy techniques, such as Structured Illumination Microscopy (SIM), have revealed that the ΔΨm is not uniform across a single mitochondrion. The inner mitochondrial membrane is divided into two compartments: the cristae membrane (CM) and the inner boundary membrane (IBM), separated by the crista junction (CJ). These compartments can sustain different membrane potentials (ΔΨC and ΔΨIBM), with the CM often being more hyperpolarized [22].
Protocol for Spatial Membrane Potential Gradient (SMPG) Analysis:
- Staining: Cells are co-stained with MitoTracker Green FM (MTG, 500 nM), which accumulates in the IMM and serves as a morphological reference, and a low concentration of TMRM (1.35-2.7 nM) to avoid saturation and allow for compartment-specific accumulation.
- SIM Imaging: Simultaneous dual-channel SIM imaging is performed.
- Quantitative Analysis: Two methods are employed:
- IBM Association Index: An automated threshold defines mitochondrial boundaries from the MTG channel. The fluorescence intensity of TMRM in the IBM region is compared to that in the CM region.
- ΔFWHM Method: The full width at half maximum (FWHM) of the cross-section intensity profiles of MTG and TMRM are compared. A larger difference (ΔFWHM) indicates greater TMRM accumulation in the cristae [22].
This technique has shown that stimuli like Ca(^{2+}) influx can hyperpolarize the cristae specifically, demonstrating a new layer of regulation in mitochondrial bioenergetics where the dominant component of the PMF can be further localized and intensified [22].
Functional Implications of ΔΨm Dominance
Bioenergetic and Signaling Superiority
The dominance of ΔΨm is not a biochemical accident but is critical for key mitochondrial functions.
- ATP Synthesis and Export: The F(1)F(O) ATP synthase uses the energy of proton flow down the electrical gradient to power the mechanical rotation that synthesizes ATP. Furthermore, the electrogenic exchange of cytosolic ADP(^{3-}) for matrix ATP(^{4-}) by the ANT is directly driven by ΔΨm, consuming ~1 charge per exchange and ensuring a continuous supply of ADP for oxidative phosphorylation [2] [14].
- Ion and Metabolite Transport: ΔΨm is the primary driving force for the mitochondrial import of positively charged ions, most notably Ca(^{2+}) and Fe(^{2+}) [2]. Mitochondrial calcium handling is crucial for regulating metabolism and cellular signaling, while iron import is essential for the biogenesis of iron-sulfur clusters, which are vital cofactors for numerous enzymes.
- Quality Control and Mitophagy: A sustained loss of ΔΨm is a key trigger for the selective elimination of damaged mitochondria via mitophagy. The PINK1/Parkin pathway is activated on mitochondria with a collapsed ΔΨm, marking them for degradation. This makes ΔΨm a central readout of mitochondrial health and a critical parameter in neurodegenerative disease research [2].
ΔΨm as a Therapeutic Vulnerability
The high ΔΨm in certain pathological cell types can be exploited for therapeutic purposes. In clonal hematopoiesis driven by Dnmt3a mutations, hematopoietic stem and progenitor cells (HSPCs) exhibit elevated ΔΨm and increased dependence on oxidative phosphorylation. This creates a therapeutic vulnerability that can be targeted with lipophilic cations like triphenylphosphonium (TPP(^+))-conjugated molecules (e.g., MitoQ). These compounds accumulate preferentially in mitochondria with high ΔΨm, where they can inhibit the electron transport chain or induce apoptosis, selectively ablating the mutant HSPCs while sparing wild-type cells [23].
Table 2: Key Research Reagents for PMF and Mitochondrial Function Analysis
| Reagent / Tool | Primary Function | Application in PMF Research |
|---|---|---|
| TMRM / TMRE | Potentiometric fluorescent dye | Quantitative and spatial measurement of ΔΨm in live cells [21] [22]. |
| SNARF-1 | Ratiometric pH-sensitive dye | Measurement of mitochondrial matrix pH for ΔpH calculation [20]. |
| Oligomycin | ATP synthase inhibitor | Used to assess coupling status; inhibits ΔΨm consumption, causing hyperpolarization [14]. |
| FCCP/CCCP | Protonophore / Uncoupler | Collapses the PMF by equalizing proton distribution; used to measure maximal respiration and probe ETC capacity. |
| Rotenone & Antimycin A | Complex I & III inhibitors | Inhibit electron transport and ΔΨm generation; used to dissect ETC function [22]. |
| JC-1 | ΔΨm-sensitive dye forming J-aggregates | Flow cytometric assessment of mitochondrial depolarization (shift from red to green fluorescence) [24]. |
| MitoTracker Probes | IMM-labeling dyes (e.g., MTG) | Used as morphological references, often in conjunction with potentiometric dyes like TMRM [22]. |
The central role of ΔΨm in these core functions and its relationship to other key mitochondrial processes are illustrated below.
Diagram 2: Functional dependencies on the proton motive force.
The preeminence of the mitochondrial membrane potential as the dominant component of the proton motive force is a well-established quantitative principle in bioenergetics, with ΔΨm contributing 70-80% of the total Δp under physiological conditions. This dominance is rooted in fundamental biophysical necessities: it provides the most efficient form of energy for ATP synthesis and export, drives the electrophoretic transport of essential cations, and serves as a key metric of organellar health. Contemporary research, leveraging advanced techniques like super-resolution microscopy and multi-parameter correlation analysis, continues to refine our understanding, revealing unexpected complexities such as spatial gradients of ΔΨm within single mitochondria. For researchers and drug developers, accurately measuring and interpreting ΔΨm is paramount. The methodologies and reagents detailed herein provide a toolkit for probing this critical parameter, whose dysregulation represents a hallmark of metabolic diseases, neurodegenerative disorders, and cancer, while also offering a unique target for therapeutic intervention.
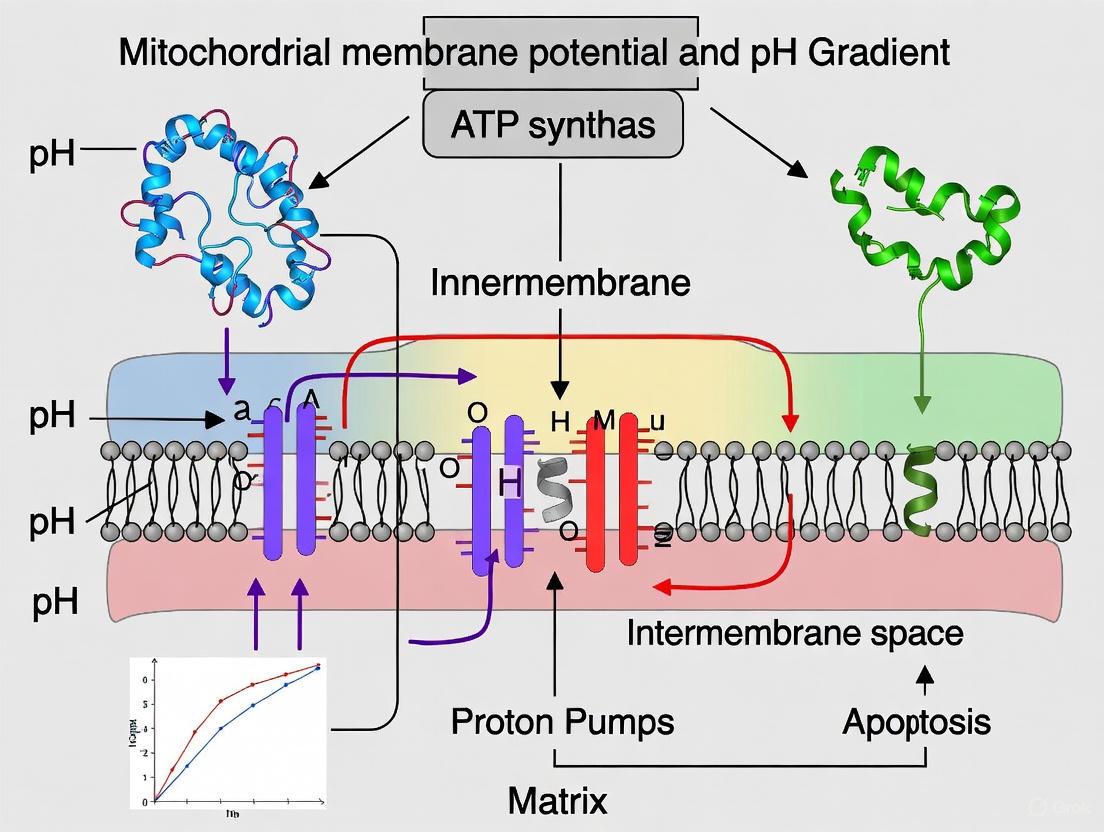
The mitochondrial proton circuit is the fundamental framework for energy conversion in eukaryotic cells. This process, described by the chemiosmotic theory, involves the generation of a protonmotive force (PMF) across the inner mitochondrial membrane by the electron transport chain (ETC) and its subsequent utilization by ATP synthase to phosphorylate ADP [25] [26]. The PMF consists of two components: a large electrical gradient, the mitochondrial membrane potential (ΔΨm), and a smaller chemical pH gradient (ΔpH) [3] [26]. Under physiological conditions, the ΔΨm of approximately -180 mV is the dominant force, equivalent to a 1000-fold difference in proton concentration, while the ΔpH of about 0.4 units contributes roughly a quarter of the total PMF [3]. This whitepaper provides a technical overview of the proton circuit's core mechanisms, quantitative dynamics, and key experimental methodologies, contextualized within ongoing research into membrane potential and pH gradient regulation.
Core Mechanism of the Proton Circuit
Generation of the Protonmotive Force by the Electron Transport Chain
The proton circuit begins with the establishment of the PMF by the ETC. The ETC consists of four protein complexes (I-IV) embedded in the inner mitochondrial membrane. Through a series of oxidation-reduction reactions, energy is released from electron donors like NADH and FADH2 and used to actively pump protons (H+) from the mitochondrial matrix into the intermembrane space.
- Complex I (NADH:ubiquinone oxidoreductase) catalyzes the transfer of electrons from NADH to ubiquinone, coupled with the translocation of 4 protons across the membrane [26].
- Complex III (Ubiquinol:cytochrome c oxidoreductase) and Complex IV (Cytochrome c oxidase) further transfer electrons, ultimately to oxygen, pumping additional protons and completing the charge separation that creates the PMF [3] [26].
This process creates an electrochemical gradient characterized by a negative and alkaline matrix relative to the intermembrane space. The energy stored in this gradient is the PMF, calculated as Δp = ΔΨm - 59ΔpH (in mV) [27].
Consumption of the Protonmotive Force by ATP Synthase
The dissipation of the PMF drives ATP synthesis. The F0F1 ATP synthase (Complex V) provides a regulated pathway for protons to flow back down their electrochemical gradient into the matrix. This exergonic flow of protons causes the rotation of a subunit within the enzyme, forcing conformational changes that catalyze the phosphorylation of ADP to ATP, a process known as chemiosmosis [26]. The coupling of proton flux to ATP synthesis makes the mitochondrial inner membrane a highly efficient energy transducer.
Quantitative Profiling of Proton Circuit Parameters
The following tables summarize key quantitative data related to the components and dynamics of the mitochondrial proton circuit, essential for its modeling and experimental analysis.
Table 1: Composition of the Protonmotive Force (PMF) under Physiological Conditions
| Parameter | Symbol | Typical Value | Contribution to PMF | Notes |
|---|---|---|---|---|
| Membrane Potential | ΔΨm | ~ -180 mV [3] | ~75% (Major component) [3] | Equivalent to a 1000-fold proton concentration difference [3]. |
| pH Gradient | ΔpH | ~ 0.4 units [3] | ~25% (Minor component) [3] | Matrix pH ~7.8, Cytosolic pH ~7.4 [3]. |
Table 2: Proton Flux Pathways and Their Physiological Impact
| Pathway | Function | Physiological Role | Contribution to Metabolic Rate |
|---|---|---|---|
| ATP Synthesis | Coupled proton flux driving ADP phosphorylation. | Primary energy conversion for cellular work. | Varies with cellular energy demand. |
| Basal Proton Leak | Unregulated proton re-entry, independent of ATP synthase. | Intrinsic membrane permeability; heat production. | ~20-30% in hepatocytes; ~50% in resting rat skeletal muscle [25]. |
| Inducible Proton Leak | Regulated proton leak via proteins like UCPs and ANT. | Thermogenesis (UCP1); mitigation of ROS production (UCP2/3) [25] [28]. | Activated by superoxide, fatty acids, and peroxidation products [25]. |
Experimental Protocols for Probing the Proton Circuit
Isolating Proton Leak Kinetics via Respiration Measurements
This protocol quantifies the contribution of proton leak to mitochondrial oxygen consumption, distinguishing it from phosphorylation-related respiration.
- Instrument Setup: Utilize an oxygenph equipped with a Clarke-type electrode, maintained at 37°C.
- Mitochondrial Incubation: Suspend isolated mitochondria (e.g., 1 mg protein/mL) in a respiration buffer (e.g., 120 mM KCl, 5 mM KH2PO4, 3 mM HEPES, 1 mM EGTA, 1 mM MgCl2, 0.3% BSA, pH 7.2).
- Substrate Addition: Introduce complex-specific substrates (e.g., 5 mM succinate for Complex II, in the presence of rotenone to inhibit Complex I).
- Inhibit ATP Synthesis: Add the ATP synthase inhibitor, oligomycin (typically 1 µg/mL). The resulting oxygen consumption rate is entirely due to the proton leak, as it is used to maintain the PMF in the absence of ATP synthesis [25].
- Titrate Leak Kinetics: Systematically titrate with incremental amounts of an uncoupler (e.g., FCCP) to progressively dissipate the PMF and measure the corresponding respiration rate. This generates a non-ohmic curve relating proton leak rate to PMF [25].
Simultaneous Measurement of Membrane Potential and Extramitochondrial pH
This advanced protocol uses a graphene-based sensor to correlate changes in ΔΨm with proton release in real-time [27].
Sensor Fabrication:
- Transfer a single layer of chemical vapor deposition (CVD)-grown graphene onto a glass substrate.
- Functionalize the graphene surface with a pyrene-NHS ester linker via π-π stacking.
- Covalently immobilize anti-TOM20 antibodies onto the linker to specifically tether mitochondria via the outer membrane protein TOM20.
- Passivate exposed graphene areas with TWEEN20 to prevent non-specific binding.
Mitochondrial Tethering and Staining:
- Incubate isolated mitochondria (e.g., 0.14 µg/µL from HeLa cells) on the functionalized graphene device for 15 minutes at 4°C.
- Gently wash with KCl-based respiration buffer (e.g., 140 mM KCl, 2 mM MgCl2, 5 mM succinate, 2 µM rotenone, pH 7.2).
- Load mitochondria with the potentiometric fluorescent dye TMRE (e.g., 40 nM) to monitor ΔΨm.
Concurrent Data Acquisition:
- Optical Channel: Use fluorescence microscopy (excitation/emission ~549/575 nm for TMRE) to image TMRE fluorescence intensity, which correlates with ΔΨm.
- Electrical Channel: Continuously monitor the conductance of the graphene sheet. Local pH changes at the graphene surface, caused by proton flux from tethered mitochondria, alter the graphene's conductance, allowing quantification of extramitochondrial pH dynamics [27].
Experimental Perturbation: Introduce reagents (e.g., uncouplers like CCCP, apoptotic inducers like BIM-BH3) while simultaneously recording both fluorescent and electrical signals to dissect the relationship between depolarization and proton release.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Investigating the Proton Circuit
| Reagent / Tool | Category | Primary Function | Example & Mechanism |
|---|---|---|---|
| Potentiometric Dyes | Fluorescent Probe | Qualitative reporting of ΔΨm. | TMRE: Lipophilic cation that accumulates in the mitochondrial matrix in a potential-dependent manner; fluorescence intensity indicates ΔΨm [27]. |
| ATP Synthase Inhibitors | Pharmacological Inhibitor | Blocks proton consumption via ATP synthase to isolate proton leak. | Oligomycin: Directly binds to the F0 subunit of ATP synthase, preventing proton flow and ATP synthesis [25]. |
| Chemical Uncouplers | Pharmacological Agent | Dissipates the PMF by shuttling protons across the membrane, uncoupling respiration from ATP synthesis. | FCCP/CCCP: Protonophores that collapse both ΔΨm and ΔpH, stimulating maximal respiration [28] [27]. |
| Uncoupling Protein (UCP) Inhibitors | Pharmacological Inhibitor | Suppresses inducible proton leak. | Guanosine Diphosphate (GDP): Inhibits UCP1-mediated proton leak in brown adipose tissue [25]. |
| Graphene Sensors | Nanomaterial Sensor | Ultrasensitive, real-time detection of local ionic/pH changes. | Functionalized Graphene: Antibody-tethered graphene acts as a highly sensitive pH sensor for monitoring proton flux from single mitochondria [27]. |
Visualization of the Proton Circuit and Key Experiments
The Mitochondrial Proton Circuit
Graphene Sensor Experimental Workflow
The proton circuit remains a dynamic area of research, with its principles extending beyond bioenergetics into redox signaling, metabolic specialization, and cell fate decisions, offering rich targets for therapeutic intervention in metabolic and degenerative diseases [3] [28].
The chemiosmotic theory historically framed the mitochondrial membrane potential (ΔΨm) and proton gradient (ΔpH) as mere intermediaries in ATP production. However, contemporary research reveals these electrochemical parameters function as dynamic signaling platforms that regulate cellular processes far beyond bioenergetics. This whitepaper synthesizes current understanding of how ΔΨm and ΔpH transduce metabolic information to control mitochondrial quality, redox signaling, ion homeostasis, and cell-wide communication. We detail quantitative measurements, experimental protocols, and visualization tools essential for investigating these emerging signaling roles, providing a framework for researchers exploring mitochondrial signal transduction in health and disease.
The traditional view of mitochondria as simple powerhouses has evolved to recognize them as sophisticated information processing systems [29]. The mitochondrial inner membrane maintains an electrochemical gradient known as the protonmotive force (Δp), comprising both an electrical component (ΔΨm, negative inside) and a chemical component (ΔpH, alkaline inside) [2]. While essential for ATP synthesis, this gradient also serves as a central regulator of mitochondrial and cellular function.
The relative contributions of ΔΨm and ΔpH to the total protonmotive force vary significantly across tissues and physiological conditions. As summarized in Table 1, quantitative assessments reveal that ΔΨm typically constitutes the dominant component, with ΔpH providing a smaller but physiologically critical contribution to the overall gradient [30].
Table 1: Quantitative Components of the Mitochondrial Protonmotive Force
| Parameter | Typical Magnitude | Percentage of Total Δp | Measurement Context |
|---|---|---|---|
| ΔΨm | ~135-175 mV | 68-97% | Isolated mitochondria & intact cells |
| ΔpH | ~0.3-0.5 pH units | 3-32% | Calculated from Δp - ΔΨm |
| Total Δp | ~180-220 mV | 100% | Varies with metabolic state |
This electrochemical gradient functions as a responsive interface that senses and transmits information about mitochondrial and cellular states. The dynamic regulation of ΔΨm and ΔpH enables mitochondria to participate in diverse signaling cascades that influence processes ranging from selective mitochondrial degradation to immune activation and metabolic adaptation [31] [29].
Fundamental Biochemistry and Regulation
Generation and Maintenance of the Proton Gradients
The electron transport chain (ETC) complexes I, III, and IV function as proton pumps that translocate hydrogen ions from the mitochondrial matrix to the intermembrane space, generating both ΔΨm and ΔpH [2] [31]. This charge separation creates a steady-state ΔΨm of approximately -140 to -180 mV (negative inside the matrix) under physiological conditions [2] [30]. The F₁F₀ ATP synthase (Complex V) then harnesses this potential energy to phosphorylate ADP, coupling proton flux back into the matrix with ATP synthesis [14].
The proton circuit operates as a dynamic equilibrium where proton efflux via the ETC is balanced by proton influx primarily through the ATP synthase. This balance maintains ΔΨm within a relatively narrow range that is optimal for both ATP production and prevention of excessive reactive oxygen species (ROS) generation [14]. The system demonstrates remarkable robustness, with the ETC capable of adjusting electron flow to maintain Δp stability despite fluctuations in substrate availability and cellular ATP demand [14].
Interrelationship Between ΔΨm and ΔpH
ΔΨm and ΔpH represent electrically and chemically distinct but thermodynamically linked components of the protonmotive force. The relationship between these components is influenced by the buffering capacity of the mitochondrial matrix and the permeability of the inner membrane to various ions [32]. In highly buffered environments typical of cellular conditions, ΔΨm constitutes the majority (approximately 80%) of the total protonmotive force, while ΔpH contributes the remainder [14] [30].
This distribution has significant functional implications. The substantial ΔΨm creates a driving force for the transport of cations into the matrix, while the ΔpH facilitates the exchange of anions and neutral metabolites [2]. The two components can vary independently under certain physiological and pathological conditions, enabling more nuanced regulation of mitochondrial processes [32].
Figure 1: Mitochondrial Proton Circuit Architecture. The electron transport chain generates both components of the protonmotive force (ΔΨm and ΔpH), which together drive ATP synthesis and mediate signaling functions.
Emerging Signaling Roles of ΔΨm
Mitochondrial Quality Control and Mitophagy
ΔΨm serves as a key parameter in mitochondrial quality control, particularly in identifying damaged organelles for selective removal via mitophagy. Sustained depolarization beyond a specific threshold (estimated at 20-40 mV decrease from normal levels) triggers recognition by autophagy machinery and subsequent elimination [2] [33]. This mechanism ensures that only dysfunctional mitochondria are targeted while preserving the functional network.
The PINK1-Parkin pathway represents the best-characterized ΔΨm-sensitive quality control system. In healthy, polarized mitochondria, PINK1 is imported and degraded. When ΔΨm collapses, PINK1 stabilizes on the outer mitochondrial membrane and recruits the E3 ubiquitin ligase Parkin, which ubiquitinates mitochondrial proteins to mark the organelle for autophagic degradation [2]. This selective process prevents the accumulation of damaged mitochondria that would otherwise produce excessive ROS and release pro-apoptotic factors.
Regulation of Ion Homeostasis and Metabolite Transport
The electrical potential across the inner membrane strongly influences ion and metabolite trafficking. Calcium (Ca²⁺) uptake into mitochondria occurs via the calcium uniporter in a ΔΨm-dependent manner, with the negative interior driving cation import [2]. This regulated calcium uptake modulates numerous processes including regulation of dehydrogenase activity, calcium buffering during signaling events, and regulation of apoptosis [2].
Similarly, ΔΨm provides the driving force for iron (Fe²⁺) transport into mitochondria, essential for iron-sulfur cluster biogenesis [2]. These cofactors are crucial for the function of numerous proteins involved in diverse cellular processes including electron transport, enzyme catalysis, and DNA repair [2]. The electrogenic nature of the adenine nucleotide translocase (ANT), which exchanges cytosolic ADP³⁻ for matrix ATP⁴⁻, further demonstrates how ΔΨm influences core metabolic exchanges [2].
Table 2: ΔΨm-Dependent Transport Processes and Their Functional Consequences
| Transport Process | Driving Force | Biological Significance |
|---|---|---|
| Calcium uptake | ΔΨm (electrophoresis) | Regulation of metabolism, cell signaling, and apoptosis triggers |
| Iron transport | ΔΨm (electrophoresis) | Iron-sulfur cluster biogenesis and cellular iron homeostasis |
| Protein import | ΔΨm across inner membrane | Nuclear-encoded mitochondrial protein import |
| ANT operation | ΔΨm (electrogenic exchange) | ATP/ADP exchange between matrix and cytosol |
| tRNA import | ΔΨm in some systems | Mitochondrial translation in certain conditions |
Redox Signaling and ROS Production
ΔΨm plays a complex role in regulating mitochondrial reactive oxygen species (ROS) production, which function as important signaling molecules at moderate levels. The relationship between ΔΨm and ROS generation follows a U-shaped curve, with both hyperpolarization and depolarization potentially increasing ROS production through different mechanisms [32] [31] [34].
Hyperpolarization (excessively high ΔΨm) restricts electron flow through the ETC, increasing the reduction state of electron carriers and the probability of electron leak to oxygen, forming superoxide at Complexes I and III [31] [34]. Conversely, reverse electron transport (RET)—a phenomenon where electrons flow backward from reduced coenzyme Q through Complex I to reduce NAD⁺—occurs under conditions of high membrane potential and succinate accumulation, generating substantial ROS at Complex I [31]. These ROS can activate specific signaling pathways that influence gene expression, proliferation, and metabolic adaptation.
Emerging Signaling Roles of ΔpH
Regulation of Reactive Oxygen Species Production
The pH gradient contributes significantly to the back-pressure that restricts electron flow through the ETC, thereby influencing ROS generation [32]. Experimental evidence demonstrates that dissipating ΔpH with nigericin (a K⁺/H⁺ exchanger) dramatically reduces superoxide and hydrogen peroxide production under state 4 respiration [32]. This indicates that ΔpH imposes a thermodynamic constraint on electron transfer, and alterations in ΔpH can directly modulate mitochondrial ROS signaling.
In post-ischemic myocardium, impairment of ΔpH is associated with elevated ROS production and increased oxidative damage [32]. The loss of ΔpH-mediated control over electron transport likely contributes to the pathological ROS generation observed in ischemia-reperfusion injury, suggesting that preserving ΔpH may have therapeutic potential in such conditions.
Protein Import and Mitochondrial Biogenesis
The import of nuclear-encoded mitochondrial proteins depends on both ΔΨm and ΔpH in a precursor-specific manner. While many proteins require only ΔΨm for import across the inner membrane, some precursors utilize the pH gradient as an additional energy source [2]. The molecular basis for this differential requirement remains an active area of investigation but appears to relate to the specific translocation machinery employed by different protein classes.
The ΔpH also influences the assembly and stability of ETC supercomplexes, potentially through effects on the lateral distribution of protein complexes within the inner membrane. This organizational role may extend to the regulation of cristae morphology, with implications for metabolic efficiency and apoptosis susceptibility.
Integrated Signaling in Physiology and Pathology
Mitochondrial Information Processing System
The integrated function of ΔΨm and ΔpH positions mitochondria as central processors in cellular information networks. The Mitochondrial Information Processing System (MIPS) concept frames mitochondria as organelles that sense inputs, integrate information through network dynamics, and produce output signals that regulate physiology at multiple levels [29].
In this model, ΔΨm and ΔpH serve as both inputs and outputs of the system. Metabolic signals, hormonal cues, and cellular stressors are transduced into changes in the electrochemical gradient, which then broadcasts this information through ion fluxes, metabolite distribution, and ROS production to influence nuclear gene expression, endoplasmic reticulum function, and overall cellular behavior [29].
Ischemia-Reperfusion Injury
Myocardial ischemia and reperfusion injury provides a clinically relevant example of ΔΨm and ΔpH dysregulation with pathological consequences. During ischemia, anaerobic metabolism causes intracellular acidification while the collapse of Δp impairs ATP synthesis [32]. Upon reperfusion, the rapid normalization of extracellular pH creates a transient reversal of the pH gradient across the inner mitochondrial membrane, disrupting normal ETC function and promoting excessive ROS production via RET [32] [31].
In this pathological context, the impairment of both ΔpH and ΔΨm mediates redox dysfunction that contributes to cellular damage and death. Therapeutic strategies that modulate the recovery of the proton gradients during reperfusion show promise in limiting infarct size and preserving cardiac function [32].
Cancer and Metabolic Diseases
Alterations in ΔΨm and ΔpH regulation are increasingly recognized in cancer biology. Many cancer cells maintain an elevated ΔΨm compared to normal cells, which may facilitate increased ATP production and drive accumulation of cations and metabolites supporting proliferation [35]. This hyperpolarization also influences ROS signaling patterns that can promote survival pathways or cell death depending on context and magnitude.
The unique bioenergetic profile of cancer cells, including their reliance on specific aspects of mitochondrial function, presents therapeutic opportunities. Agents that selectively disrupt ΔΨm and ΔpH regulation in malignant cells are under investigation as potential anticancer therapies [35].
Experimental Approaches and Methodologies
Measurement Techniques for ΔΨm and ΔpH
Accurate measurement of mitochondrial membrane potential and pH gradient requires careful method selection and interpretation. Fluorescent probes remain the most accessible approach for intact cells, but require proper calibration and consideration of potential artifacts [14] [30].
Table 3: Key Research Reagents for Investigating ΔΨm and ΔpH
| Reagent | Target | Mechanism of Action | Key Applications |
|---|---|---|---|
| TMRM/TMRE | ΔΨm | Potential-dependent accumulation | Quantitative imaging in live cells |
| JC-1 | ΔΨm | Potential-dependent J-aggregate formation | Flow cytometry, ratio-metric readout |
| Rhodamine 123 | ΔΨm | Potential-dependent accumulation | Population-level measurements |
| BCECF-AM | ΔpH | Ratiometric pH-sensitive fluorescence | Mitochondrial matrix pH |
| Nigericin | ΔpH | K⁺/H⁺ exchanger, dissipates ΔpH | Experimental dissipation of pH gradient |
| Valinomycin | ΔΨm | K⁺ ionophore, dissipates ΔΨm | Experimental depolarization |
| Oligomycin | ATP synthase | Inhibits proton flux through Complex V | Assessing coupling efficiency |
| FCCP/CCCP | ΔΨm/ΔpH | Protonophores, dissipate both components | Maximum respiration assessment |
For ΔΨm determination, tetramethylrhodamine methyl ester (TMRM) and ethyl ester (TMRE) provide reliable measurements when used at low concentrations (typically 10-50 nM) in non-quenching mode [14] [30]. Ratiometric dyes like JC-1 offer internal calibration through emission shift, but require careful interpretation due to potential artifacts from mitochondrial morphology changes [30].
Measurement of ΔpH presents greater technical challenges. Approaches include targeted pH-sensitive fluorescent proteins, rationetric dyes like BCECF, and calculation from the distribution of weak acids across the membrane [30]. Simultaneous measurement of both ΔΨm and ΔpH remains technically demanding but provides the most complete assessment of the protonmotive force.
Critical Experimental Considerations
Researchers must recognize that ΔΨm measurements alone provide limited information about oxidative phosphorylation flux. Due to the dynamic response of the ETC to maintain Δp stability, significant changes in respiratory rate can occur with minimal ΔΨm alteration [14]. Combining ΔΨm assessment with measurements of oxygen consumption rate provides a more comprehensive view of mitochondrial function.
Proper calibration is essential for quantitative comparisons. For ΔΨm, this typically involves using protonophores (FCCP/CCCP) to fully depolarize mitochondria and establish a baseline, followed by inhibition of respiration (e.g., with antimycin A plus rotenone) to confirm specificity [14] [30]. Interpretation should also account for potential changes in mitochondrial volume, morphology, and density, which can all influence fluorescent signals independent of actual changes in membrane potential [30].
Figure 2: Experimental Workflow for Accurate ΔΨm/ΔpH Assessment. A systematic approach incorporating appropriate controls and complementary assays ensures reliable interpretation of electrochemical gradient measurements.
The emerging roles of ΔΨm and ΔpH in cellular signaling represent a paradigm shift in mitochondrial biology. Rather than viewing these gradients solely as bioenergetic intermediates, we now recognize them as dynamic regulators of cellular function that influence processes from autophagy to immune signaling. This expanded understanding opens new therapeutic avenues for diseases characterized by mitochondrial dysfunction.
Future research should focus on developing more precise tools for tissue-specific modulation of ΔΨm and ΔpH, elucidating the molecular mechanisms that sense these parameters, and understanding how their dysregulation contributes to age-related diseases and metabolic disorders. As we continue to decipher the complex language of mitochondrial signaling, the therapeutic potential of targeting these fundamental cellular parameters will undoubtedly expand.
Measuring the Force: Advanced Techniques for Quantifying ΔΨm and ΔpH
The mitochondrial membrane potential (ΔΨM) is a central intermediate in oxidative energy metabolism, forming the major component of the proton motive force (Δp) that drives ATP synthesis. [21] This potential, typically ranging from -108 to -158 mV in mammalian cells, reflects the energetic state of mitochondria and serves as a key indicator of cellular health. [21] Fluorescent potentiometric dyes have become indispensable tools for investigating ΔΨM in living cells, offering non-invasive monitoring of mitochondrial function with high temporal and spatial resolution. Among these probes, tetramethylrhodamine methyl ester (TMRM) and its derivatives represent some of the most widely utilized indicators. [36] [37]
This technical guide examines the applications and limitations of TMRM and other potentiometric dyes, with particular emphasis on their role in elucidating the relationship between mitochondrial membrane potential and pH gradient research. The proton motive force (Δp) comprises both ΔΨM and the mitochondrial pH gradient (ΔpHm), represented by the equation Δp (mV) = ΔΨm - 60ΔpHm at 37°C. [36] Understanding this relationship is crucial, as these dyes measure only the charge gradient (ΔΨm) and cannot directly assess the proton gradient (ΔpHm), which must be measured using complementary approaches. [36]
Fundamental Principles of Potentiometric Dyes
Biophysical Mechanisms
Potentiometric fluorescent dyes are typically lipophilic cations that distribute across biological membranes according to the Nernst equation, accumulating in compartments with more negative interiors. [21] [36] In the context of intact cells, these dyes must first cross the plasma membrane (influenced by ΔΨP) before being further accumulated into mitochondria (influenced by ΔΨM). [21] The resulting distribution between these compartments reaches a Nernstian equilibrium where the dye concentration ratio reflects the potential difference. [21]
For cationic dyes like TMRM, a more negative (polarized) ΔΨM leads to greater dye accumulation in the mitochondrial matrix, while depolarization results in dye release. [36] This redistribution forms the basis for measuring ΔΨM changes, typically monitored through fluorescence intensity shifts, spectral changes, or fluorescence quenching phenomena. [37]
Critical Distinction: ΔΨm vs. ΔpHm
A fundamental limitation of cationic potentiometric dyes is their exclusive sensitivity to electrical gradients, not proton gradients. The total proton motive force (Δp) includes both components, with typical physiological values of ΔΨm = 150-180 mV and ΔpHm contributing 30-60 mV (based on a pH gradient of 0.5-1.0 units). [36] This distinction is crucial, as ΔΨm and ΔpHm do not always change in parallel during cellular stress, potentially leading to misinterpretation if only one parameter is measured. [36]
Table 1: Components of the Mitochondrial Proton Motive Force
| Parameter | Symbol | Typical Values | Contribution to Δp | Measurement Approach |
|---|---|---|---|---|
| Membrane Potential | ΔΨm | -150 to -180 mV | ~150-180 mV | Cationic dyes (TMRM, TMRE) |
| pH Gradient | ΔpHm | 0.5-1.0 units | ~30-60 mV | pH-sensitive fluorophores |
| Proton Motive Force | Δp | 180-220 mV | Combined | Calculated from both components |
Case studies have demonstrated that during certain stress conditions, ΔΨm may hyperpolarize while mitochondrial matrix pH simultaneously decreases (increased [H+]mito). [36] Such paradoxical findings highlight the necessity of complementary measurements using pH-sensitive mitochondrial dyes (e.g., SNARF-1, Rh-NorCy) to fully understand mitochondrial bioenergetics. [36] [38] [39]
Diagram 1: Mitochondrial Membrane Potential and Dye Accumulation. The inner mitochondrial membrane separates a negative matrix from a positive intermembrane space, creating both electrical (ΔΨm) and chemical (ΔpH) gradients that drive ATP synthesis and cationic dye accumulation.
Major Potentiometric Dyes: Properties and Applications
Rhodamine Derivatives: TMRM, TMRE, and Rhodamine 123
Tetramethylrhodamine methyl ester (TMRM) and ethyl ester (TMRE) represent the gold standard for ΔΨM measurements due to their relatively low mitochondrial binding and minimal suppression of respiratory control when used at appropriate concentrations. [37] These dyes exhibit absorption maxima around 548 nm and emission maxima around 573 nm, making them suitable for standard TRITC filter sets. [37]
Table 2: Characteristics of Common Rhodamine-Based Potentiometric Dyes
| Probe | Spectra (Ex/Em) | Binding Properties | Respiratory Inhibition | Primary Applications |
|---|---|---|---|---|
| TMRM | ~548/573 nm | Lowest mitochondrial binding | Minimal at low concentrations | Quantitative ΔΨM measurements in live cells [37] |
| TMRE | ~549/574 nm | Moderate binding | Moderate | Acute ΔΨM changes, non-quenching mode [36] [37] |
| Rhodamine 123 | ~507/529 nm | Higher binding | Moderate | Fast-resolution acute studies, quenching mode [36] |
| JC-1 | 514/529 nm (monomer); 585/590 nm (aggregate) | Extensive aggregation | Varies with concentration | Apoptosis studies, flow cytometry [36] |
The critical distinction between these dyes lies in their mitochondrial binding characteristics. Binding is temperature-dependent and follows the order TMRE > Rhodamine 123 > TMRM. [37] This property significantly affects their distribution, as dyes accumulate in mitochondria in greater quantities than predicted by the Nernst equation alone due to binding to inner and outer aspects of the inner mitochondrial membrane. [37]
TMRM is particularly valued for quantitative measurements because its lower binding properties make its distribution more accurately reflect the true ΔΨM. [21] [37] When used at low concentrations (typically 20-100 nM), TMRM produces minimal respiratory chain inhibition, enabling longer-term experiments without significantly perturbing mitochondrial function. [36] [37]
Operational Modes: Quenching vs. Non-Quenching
Rhodamine dyes can be used in two distinct measurement modes:
Non-quenching mode employs low dye concentrations (∼1-30 nM), where accumulated dye remains in a monomeric state with minimal self-quenching. [36] In this mode, increased ΔΨM leads to increased mitochondrial dye accumulation and consequently higher fluorescence intensity. This approach is preferred for quantitative assessments of resting ΔΨM and for monitoring slow potential changes. [36]
Quenching mode utilizes higher dye concentrations (>50-100 nM), resulting in dense dye accumulation in mitochondria that causes aggregation and fluorescence quenching. [36] depolarization leads to dye redistribution into the cytosol, dequenching, and increased fluorescence. This mode offers amplified signals for detecting acute ΔΨM changes but is less suitable for quantitative absolute potential measurements. [36]
Alternative and Specialized Dyes
JC-1 uniquely forms J-aggregates at polarized potentials that emit at longer wavelengths (590 nm) compared to monomers (529 nm). [36] This property enables ratiometric measurements, but the dye's slow equilibrium and sensitivity to factors beyond ΔΨM (including mitochondrial volume and hydrogen peroxide) limit its quantitative accuracy. [36]
MitoTracker probes (CMXRos, CM-H2XRos) contain thiol-reactive chloromethyl moieties that covalently bind mitochondrial proteins, enabling retention after fixation. [40] While valuable for fixed-cell applications, this irreversible binding prevents their use for dynamic ΔΨM monitoring in living cells.
Next-generation probes include advanced dyes like MAO-N3 for super-resolution inner mitochondrial membrane imaging [41] and potential-sensitive dyes like ElectroFluor730p for optical mapping of action potentials in cardiomyocytes. [42] [43]
Quantitative Measurement Methodologies
Absolute ΔΨM Quantification in Live Cells
A sophisticated approach for measuring absolute ΔΨM values in millivolts involves modeling fluorescent probe compartmentation and dynamics using both a bis-oxonol-type indicator of plasma membrane potential (ΔΨP) and TMRM as the ΔΨM probe. [21] This method accounts for multiple factors including:
- ΔΨP-dependent probe redistribution
- Matrix-to-cell volume ratio
- High- and low-affinity binding
- Activity coefficients
- Background fluorescence and optical dilution [21]
The calibration procedure enables deconvolution of ΔΨP and ΔΨM from temporal fluorescence intensity changes, accounting for Nernstian behavior and slow redistribution kinetics. [21] In cultured rat cortical neurons, this method revealed a resting ΔΨM of -139 mV, with physiological regulation between -108 mV and -158 mV during electrical stimulation. [21] The standard error of mean absolute calibrated values including all biological and systematic measurement errors was less than 11 mV. [21]
Diagram 2: Workflow for Quantitative ΔΨM Measurement. The protocol combines experimental measurements with biophysical modeling to convert fluorescence signals into absolute membrane potential values in millivolts.
Microplate Reader-Based High-Throughput Assay
For higher-throughput applications, microplate reader-based assays enable ΔΨM measurement in 96- or 384-well formats. [44] A typical protocol involves:
Cell Preparation: Plate cells (e.g., human fibroblasts) at 40% confluency in growth medium and culture for 24 hours before switching to galactose medium to enhance mitochondrial respiration. [44]
Dye Loading: Incubate cells with 150 nM TMRM in assay buffer (80 mM NaCl, 75 mM KCl, 25 mM D-glucose, 25 mM HEPES, pH 7.4) at 37°C for 5 minutes. [44]
Wash and Measurement: Wash cells 4× with PBS and measure fluorescence (Ex: 544 nm, Em: 590 nm) using a microplate reader. [44]
Control Measurements: Parallel assays include:
- CCCP treatment (10 μM) to collapse ΔΨM for background subtraction
- Cell number quantification using ethidium homodimer (1 μM; Ex: 544 nm, Em: 645 nm) after freeze-thawing [44]
This approach generates reproducible, quantitative data suitable for drug screening and toxicological assessments, with results expressed as total TMRM fluorescence minus CCCP-treated values normalized to cell number. [44]
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Mitochondrial Membrane Potential Studies
| Reagent/Category | Specific Examples | Function and Application |
|---|---|---|
| Primary Dyes | TMRM, TMRE, Rhodamine 123 | ΔΨM detection in live cells; choice depends on required precision, temporal resolution, and imaging modality [36] [37] |
| Reference Probes | CCCP, FCCP | Protonophores that collapse ΔΨM for system validation and background measurement [44] |
| Inhibitors | Oligomycin, Antimycin A | Modulate ETC function for experimental manipulation of ΔΨM [36] |
| Viability Indicators | Ethidium homodimer, Propidium iodide | Assess cell number and viability for data normalization [44] |
| Advanced Dyes | ElectroFluor630, MAO-N3, HIDE probes | Specialized applications: ratiometric measurements, super-resolution imaging, long-term tracking [42] [41] |
| pH Probes | Rh-NorCy, SNARF-1, M-pH | Monitor mitochondrial pH simultaneously with ΔΨM for complete proton motive force assessment [38] [39] |
Limitations and Technical Challenges
Methodological Constraints
Despite their utility, potentiometric dyes present significant limitations that researchers must address:
Plasma Membrane Potential Influence: Since these dyes must first cross the plasma membrane before accumulating in mitochondria, changes in ΔΨP can significantly influence apparent ΔΨM measurements. [21] [44] This confounder necessitates simultaneous monitoring of ΔΨP using appropriate anionic dyes in rigorous experiments. [21]
Binding Artifacts: Non-ideal Nernstian behavior occurs due to dye binding to mitochondrial membranes and proteins. [37] This binding is temperature-dependent and varies among dyes (TMRE > Rhodamine 123 > TMRM), requiring careful calibration and modeling for quantitative measurements. [21] [37]
Concentration-Dependent Effects: Excessive dye concentrations can suppress mitochondrial respiration, with TMRE showing greater inhibition than TMRM. [37] Additionally, high concentrations may induce aggregation artifacts and apparent "quenching" that complicate interpretation. [36]
Dynamic Range Limitations: Most potentiometric dyes exhibit relatively small fluorescence changes per mV of potential difference, limiting their sensitivity to detect subtle ΔΨM fluctuations. [21]
Critical Distinction from pH Measurements
Perhaps the most significant limitation is the exclusive sensitivity of these dyes to electrical gradients, not proton gradients. Case studies demonstrate that during certain pathological conditions, such as exposure to the HIV Tat protein, ΔΨM may hyperpolarize while mitochondrial matrix pH decreases—opposite responses that would be misinterpreted if only one parameter were measured. [36] Such findings highlight the necessity of complementary pH measurements using mitochondrial-targeted pH probes (e.g., Rh-NorCy, SNARF-1) for complete bioenergetic assessment. [36] [38] [39]
Future Perspectives and Emerging Technologies
Recent advances in potentiometric dye technology focus on addressing current limitations through novel approaches:
Ratiometric Dyes: New voltage-sensitive dyes like ElectroFluor630 enable ratiometric measurements that compensate for artifacts from non-uniform staining, photobleaching, and cellular motion. [42] [43] This is particularly valuable for contractile cells like cardiomyocytes. [42]
Super-Resolution Compatible Probes: Probes such as MAO-N3 support long-term super-resolution inner mitochondrial membrane imaging using modalities including STED, SIM, and SMLM. [41] These advances permit visualization of cristae dynamics previously beyond resolution limits.
Near-Infrared and pH Probes: Developments like Rh-NorCy, a mitochondria-targeted NIR pH probe with suitable pKa (7.27) for physiological monitoring, enable simultaneous assessment of both ΔΨm and ΔpHm components. [38]
HIDE Probes: High-Density Environmentally Sensitive probes offer significantly improved photostability for long-term time-lapse imaging, maintaining signal for over 12.5 hours compared to commercial dyes that degrade within 2 hours. [41]
These technological innovations promise to enhance our understanding of the complex relationship between mitochondrial membrane potential and pH gradients, providing more comprehensive tools for investigating mitochondrial dysfunction in disease pathogenesis and therapeutic development.
The mitochondrial membrane potential (MMP or ΔΨm) is a fundamental component of the protonmotive force (PMF), the electrochemical gradient essential for adenosine triphosphate (ATP) production via oxidative phosphorylation [45] [3]. Under physiological conditions, the MMP (approximately -180 mV) constitutes the dominant element of the PMF, while the chemical proton gradient (ΔpH) contributes a smaller, yet significant, portion [3]. For decades, understanding the precise spatial organization of this potential across the complex architecture of the inner mitochondrial membrane (IMM) remained elusive. The IMM is divided into two distinct compartments: the cristae membrane (CM), which houses the electron transport chain (ETC) complexes and ATP synthase, and the inner boundary membrane (IBM), which lies adjacent to the outer membrane [46]. These subdomains are separated by narrow, regulatory structures known as crista junctions (CJ) [46].
The central technical challenge has been that these structures, particularly the cristae, often fall below the diffraction limit of conventional light microscopy (approximately 200-250 nm). This limitation prevented the resolution of potential differences between the CM and IBM in living, functional mitochondria [45] [46]. The advent of super-resolution microscopy (SRM) has overcome this barrier, enabling nanoscale visualization of mitochondrial ultrastructure and function in real time [45] [46] [47]. This technical guide details how SRM techniques are now being deployed to visualize intra-mitochondrial potential gradients, providing unprecedented insights into mitochondrial bioenergetics and its relationship with the pH gradient within the broader context of cellular physiology.
Key Super-Resolution Modalities for Mitochondrial Imaging
Several super-resolution modalities have proven effective for resolving mitochondrial structure and potential. The table below summarizes the primary techniques, their key principles, and their applicability to live-cell imaging of mitochondrial potentials.
Table 1: Super-Resolution Microscopy Techniques for Visualizing Intra-Mitochondrial Gradients
| Technique | Key Principle | Typical Resolution | Live-Cell Compatibility | Key Advantages for MMP Imaging |
|---|---|---|---|---|
| Structured Illumination Microscopy (SIM) [46] [47] | Uses patterned illumination to encode high-frequency information into the observable image. | ~120 nm (2x enhancement) [48] | High (Fast acquisition) | Low phototoxicity, suitable for dynamic imaging of potential gradients [46]. |
| Stimulated Emission Depletion (STED) [45] [46] | Uses a depletion laser beam to de-excite fluorophores at the periphery of the focal spot. | ~90 nm or better [45] | Moderate | Excellent resolution for discerning cristae structure [45] [46]. |
| Super-Resolution Panoramic Integration (SPI) [48] | Leverages multifocal optical rescaling and synchronized line-scan readout for instant super-resolution. | ~120 nm [48] | Very High (High-throughput) | Enables instantaneous, high-throughput imaging of large populations of mitochondria [48]. |
A Paradigm Shift: The Voltage-Dependent Membrane Binding Model
A critical breakthrough in correctly interpreting SRM images of MMP has been the development and validation of a new biophysical model for lipophilic cationic dyes (e.g., TMRM, TMRE). Historically, it was often incorrectly assumed that the fluorescence signal from these dyes originated from the dye molecules freely distributed in the mitochondrial matrix [45]. However, SRM studies on isolated, functional mitochondria have demonstrated that over 90% of the fluorescent signal in energized mitochondria comes from dye molecules bound to the membranes, not from the free matrix pool [45].
This voltage-dependent membrane binding model states that the distribution of these lipophilic cations is governed by thermodynamics, leading to a Nernstian equilibrium where the dye accumulates on the side of the membrane with the more negative potential (the matrix side) [45]. The key insight is that the dye's binding to the membrane is itself potential-dependent. Consequently, in super-resolution images, the intense fluorescence signal is localized precisely at the cristae membranes, which typically hold a higher (more negative) potential ((\DeltaΨC)) than the IBM ((\DeltaΨ{IBM})) [45] [46]. This model fundamentally clarifies why SRM images show strong intensity at the cristae and provides the foundation for quantitative imaging of super-resolution voltages [45].
Diagram 1: Model of Voltage-Dependent Dye Binding and Intra-Mitochondrial Potential Gradients
Experimental Protocols: Measuring Spatial Membrane Potential Gradients
This section provides a detailed methodology for visualizing and quantifying spatial membrane potential gradients (SMPG) across the inner mitochondrial membrane in living cells using SIM and the potentiometric dye TMRM.
Cell Staining and Sample Preparation
- Cell Lines: The protocol has been validated in both HeLa cells (glycolytic) and EA.hy926 cells (more oxidative), confirming its applicability across metabolic phenotypes [46].
- Dye Loading:
- Incubate cells with a dual-dye system for a minimum of 30 minutes at 37°C in the dark.
- Use 500 nM MitoTracker Green FM (MTG). This dye accumulates in the IMM in a potential-dependent manner but, once bound, becomes insensitive to subsequent potential changes, serving as a structural reference for mitochondrial morphology [46].
- Use a low concentration of TMRM (1.35 - 13.5 nM). This is critical. Low concentrations prevent saturation of the cristae membranes, ensuring that TMRM distribution remains sensitive to the local membrane potential, thereby revealing the gradient between CM and IBM [46].
- Imaging Buffer: Perform imaging in a dye-free buffer to prevent dye replenishment from the extracellular space and to ensure a stable equilibrium for measurement.
Image Acquisition via Structured Illumination Microscopy (SIM)
- Microscope Setup: Use a super-resolution microscope capable of SIM (e.g., Nikon N-SIM) [46].
- Acquisition Parameters: Perform simultaneous dual-channel imaging of MTG and TMRM. The MTG channel defines the spatial reference and mitochondrial boundaries. The TMRM channel reports the local membrane potential. Acquire z-stacks if optical sectioning is required for the specific SIM implementation.
Data Analysis: Quantifying the Gradient
Two primary methods are used to quantify the TMRM distribution relative to the MTG reference, translating fluorescence localization into a readout of the potential gradient.
- IBM Association Index (Automated) [46]:
- Use the MTG channel to automatically define the total mitochondrial area using an algorithm like Otsu thresholding.
- Programmatically shrink and widen these borders to create two distinct regions of interest (ROIs): one for the Inner Boundary Membrane (IBM) and one for the Cristae Membrane (CM).
- Calculate the average fluorescence intensity of TMRM in the IBM and CM ROIs.
- Compute the IBM Association Index = TMRMIBM / TMRMCM. A lower index indicates a greater relative accumulation of TMRM in the cristae, signifying a steeper potential gradient ((\DeltaΨC > \DeltaΨ{IBM})) [46].
- ΔFWHM Method (Semi-Automated) [46]:
- Draw a line scan perpendicular to the long axis of a mitochondrion on both the MTG and TMRM images.
- Plot the fluorescence intensity profile for each channel across this line.
- Measure the Full Width at Half Maximum (FWHM) for each intensity profile.
- Calculate ΔFWHM = FWHM(MTG) - FWHM(TMRM). A larger positive ΔFWHM indicates that the TMRM signal is narrower and more concentrated in the center (cristae) of the mitochondrion compared to the structural marker MTG, confirming a higher potential in the cristae [46].
Diagram 2: Experimental Workflow for SMPG Analysis
Key Research Reagent Solutions and Experimental Tools
The following table lists essential reagents and tools required for conducting super-resolution imaging of intra-mitochondrial potential gradients.
Table 2: Essential Research Reagents and Tools for SMPG Imaging
| Item Name | Function/Description | Critical Application Note |
|---|---|---|
| Tetramethylrhodamine Methyl Ester (TMRM) [49] [46] [50] | Lipophilic cationic potentiometric dye. Accumulates in mitochondria in a membrane potential-dependent manner. | Use at low (1.35-13.5 nM), non-quenching concentrations to resolve CM/IBM gradients. High concentrations saturate signal and obscure gradients [46]. |
| MitoTracker Green FM (MTG) [46] | Cell-permeant green-fluorescent mitochondrial stain. | Acts as a potential-insensitive structural reference dye for defining mitochondrial morphology and sub-compartments during analysis [46]. |
| Histamine [46] | IP3-generating agonist that induces calcium release from the endoplasmic reticulum. | Used as a physiological stimulant to increase matrix [Ca²⁺], which boosts TCA cycle activity and enhances ΔΨC, thereby altering SMPGs [46]. |
| Rotenone & Antimycin A [45] [46] | Specific inhibitors of ETC Complex I and III, respectively. | Used as control reagents to inhibit proton pump activity and confirm that observed SMPG changes are dependent on ETC function [46]. |
| Carbonyl cyanide m-chlorophenyl hydrazone (CCCP) [45] | Protonophore uncoupler that dissipates the protonmotive force. | Used as a control to collapse the MMP, validating the potential-dependent nature of the dye signal and the model [45]. |
| SIM-Compatible Microscope [48] [46] [47] | Microscope system capable of structured illumination microscopy. | Enables the requisite ~120 nm resolution to distinguish fluorescence signals from the CM and IBM in living cells [46]. |
Functional Implications and Integration with pH Gradient Research
The visualization of intra-mitochondrial potential gradients has profound functional implications, tightly linking membrane potential dynamics with other aspects of mitochondrial biology, including the pH gradient.
Cristae as Bioenergetic Hotspots: The finding that (\DeltaΨC) is often higher than (\DeltaΨ{IBM}) confirms the cristae as the primary sites for proton pumping and the electrochemical generation of the PMF. The CJ acts as a permeability barrier, helping to retain protons within the cristae lumen, which is crucial for maintaining both the MMP and the ΔpH components of the PMF for efficient ATP synthesis [46].
Calcium-Mediated Regulation: Mitochondrial calcium uptake, often initiated at the CM, hyperpolarizes the cristae specifically. This Ca²⁺-induced boost of the TCA cycle and ETC activity increases (\DeltaΨ_C), demonstrating how biochemical signals are translated into nanoscale bioelectric changes that optimize ATP production [46]. This hyperpolarization must be carefully regulated, as excessive potential can lead to dielectric breakdown and ROS production.
Crista Junction "Overflow Valve" Mechanism: The CJ is not a static barrier. It is proposed to dynamically regulate the potential gradient. During excessive cristae hyperpolarization, the CJ may act as an "overflow valve," allowing controlled ion movement to protect mitochondrial integrity, a mechanism that intrinsically links the regulation of MMP and ΔpH [46].
Metabolic Specialization and Quality Control: Gradients of MMP within a single mitochondrion and across the network influence critical processes like protein import and mitophagy. Regions with lower MMP are more likely to be targeted for degradation, while higher MMP supports biogenesis [3]. Furthermore, local MMP influences the activity of metabolic enzymes, promoting the emergence of specialized mitochondrial subpopulations dedicated to either oxidative (ATP-producing) or reductive (biosynthetic) metabolism [3].
In conclusion, the ability to visualize intra-mitochondrial potential gradients with super-resolution microscopy has transformed our understanding of mitochondrial bioenergetics. It has moved the field from a simplistic, whole-organelle view to a nuanced, compartmentalized model where the cristae membranes function as dynamic bioenergetic units. This paradigm shift, grounded in the voltage-dependent membrane binding model of potentiometric dyes, firmly situates the MMP within a broader framework where its gradients are essential regulators of energy production, signaling, and cellular fate, inextricably linked to the pH gradient that together constitute the protonmotive force of life.
In the interconnected realms of mitochondrial membrane potential (ΔΨm) and pH gradient research, quantitative accuracy is not merely a technical concern but a foundational requirement for biological relevance. The proton motive force (PMF), which drives mitochondrial ATP synthesis, comprises both an electrical component (ΔΨm) and a chemical component (ΔpH) [3]. Under physiological conditions, the mitochondrial matrix is alkaline relative to the cytoplasm, typically maintaining a ΔpH of approximately 0.4 units [3]. While ΔΨm constitutes the dominant portion of the PMF (approximately -180 mV, equivalent to a 1000-fold proton concentration difference), the ΔpH contributes significantly to the overall energetic landscape [3]. The accurate quantification of these parameters is essential for understanding how mitochondria integrate metabolic signals, regulate quality control, and contribute to cellular fate decisions. This technical guide examines the primary sources of measurement artifacts and provides robust calibration methodologies to ensure quantitative accuracy in mitochondrial bioenergetics research.
The Critical Relationship Between Mitochondrial Membrane Potential and pH Gradients
The inner mitochondrial membrane (IMM) maintains a complex bioenergetic architecture where electrical and chemical gradients are functionally coupled. The electron transport chain (ETC) pumps protons from the matrix to the intermembrane space, simultaneously establishing both ΔΨm and ΔpH [3]. These gradients are not uniform across the mitochondrial membrane system; recent super-resolution microscopy studies reveal that the cristae membrane (CM) can maintain a more hyperpolarized potential (ΔΨC) compared to the inner boundary membrane (IBM), with the cristae junction (CJ) acting as a barrier that regulates ion movement [22].
This compartmentalization has profound implications for cellular signaling. Mitochondrial calcium elevation, for instance, hyperpolarizes the cristae membrane likely through Ca2+-sensitive enhancement of tricarboxylic acid (TCA) cycle activity and subsequent oxidative phosphorylation [22]. Dynamic multi-parameter correlation measurements have identified a CJ-based membrane potential "overflow valve" mechanism that protects mitochondrial integrity during excessive cristae hyperpolarization [22]. The functional interaction between the ADP/ATP carrier (AAC) and ATP synthase in regulating H+ distribution across the IMM further demonstrates the intricate coupling between pH dynamics and membrane potential [8]. Acidification of the matrix due to AAC activity can induce re-alkalization through reverse activity of ATP synthase, creating a feedback system that maintains pH homeostasis [8].
Dye-Dependent Artifacts
Fluorescent potentiometric probes, while indispensable, introduce numerous potential artifacts that can compromise data interpretation. The distribution of dyes such as tetramethylrhodamine methyl ester (TMRM) depends critically on concentration-dependent saturation effects. At low concentrations (2.7–1.35 nM), TMRM accumulates preferentially in the cristae, while at high concentrations (81–40.5 nM), cristae become saturated, causing relative increases in IBM staining [22]. This concentration-dependent distribution necessitates careful titration for specific experimental systems.
JC-1, a commonly used ΔΨm probe, presents particular challenges due to its non-equilibrium accumulation and concentration-dependent aggregation, making quantitative interpretation difficult [21]. For pH measurements, esterase-dependent fluorophores like SNARF-1 AM require careful control of loading conditions, as warm loading temperatures (37°C) favor cytosolic distribution, while cold loading (4°C) enables better mitochondrial localization [51].
System-Dependent Artifacts
Cellular context introduces additional confounding factors. The fluorescence readout of potentiometric dyes represents a complex function of not only ΔΨm but also plasma membrane potential (ΔΨP), matrix:cell volume ratio, activity coefficients, binding characteristics, and spectral changes induced by the mitochondrial environment [21]. Variations in mitochondrial architecture between cell types, including differences in cristae density and CJ permeability, further complicate cross-system comparisons [22].
Without proper calibration, fluorescence intensity changes may reflect alterations in mitochondrial mass, morphology, or positioning rather than genuine changes in ΔΨm or pH [52]. This is particularly problematic in pathological models where these parameters may be concurrently altered.
Quantitative Calibration Methodologies
Absolute Calibration of Mitochondrial Membrane Potential
The quantitative assay of absolute ΔΨm values in millivolts requires a biophysical model of fluorescent probe compartmentation and dynamics [21]. This approach incorporates a bis-oxonol-type indicator of plasma membrane potential (ΔΨP) alongside the ΔΨm probe TMRM, using fluorescence imaging and voltage clamp validation.
Table 1: Key Parameters for Absolute ΔΨm Calibration
| Parameter | Description | Measurement Approach |
|---|---|---|
| Matrix:Cell Volume Ratio | Ratio of mitochondrial matrix volume to cellular volume | Confocal microscopy with matrix-targeted fluorophores |
| Activity Coefficients | Correction for non-ideal behavior in concentrated environments | Comparative measurements in calibrated systems |
| Binding Affinities | High- and low-affinity binding sites for dyes | Fluorescence titration experiments |
| Background Fluorescence | Non-specific fluorescence signals | Measurement in dye-free conditions |
| Optical Dilution | Signal reduction from out-of-focus light | Point spread function characterization |
The calibration protocol involves several critical steps:
- Parallel Measurement of ΔΨP: Use a bis-oxonol-type ΔΨP indicator to account for plasma membrane potential contributions to dye distribution [21].
- Determination of Volume Ratios: Quantify matrix:cell volume ratios using confocal microscopy with appropriate compartment-specific markers [21].
- Characterization of Binding Parameters: Measure high- and low-affinity binding components through systematic titration [21].
- Application of Nernstian Distribution Models: Implement mathematical models based on Eyring rate theory to deconvolute ΔΨP and ΔΨm from fluorescence time courses [21].
Using this methodology, resting ΔΨm in cultured rat cortical neurons was quantified at -139 ± 5 mV, with physiological regulation observed between -108 mV and -158 mV during metabolic activation [21]. The total error in determining resting ΔΨm, including all systematic errors from calibration parameters, is less than 11 mV [21].
Calibration of Mitochondrial pH
The measurement of mitochondrial matrix pH requires careful calibration using pH-sensitive fluorophores such as SNARF-1 [51]. The protocol involves several key stages:
- Cold Loading Technique: Incubate cells with SNARF-1 AM at 4°C to slow esterase activity, allowing better mitochondrial localization before hydrolysis [51].
- In Situ Calibration: At experiment conclusion, perfuse cells with calibration buffers of known pH (e.g., 6.5, 7.0, 7.5) containing the H+/K+ ionophore nigericin to equilibrate intra- and extracellular pH [51].
- Ratiometric Measurement: Utilize SNARF-1's pH-dependent emission shift (580 nm vs. 640 nm emission when excited at 514 nm) to establish a calibration curve [51].
- Mitochondrial Specificity Verification: Co-localize with mitochondrial markers (e.g., MitoTracker Green) to confirm compartmental specificity [51].
This approach enables resolution of mitochondrial matrix pH with sufficient sensitivity to detect physiologically relevant changes driven by electron transport chain activity, ADP/ATP carrier function, and ATP synthase operation [8] [51].
Diagram 1: Comprehensive calibration workflow for mitochondrial parameter measurement, illustrating the sequential steps from dye selection through data validation.
Advanced Techniques for Spatial and Dynamic Resolution
Super-Resolution Approaches
Structured illumination microscopy (SIM) and stimulated emission depletion (STED) microscopy enable resolution of mitochondrial membrane potential gradients at the sub-organellar level [22]. These techniques have revealed that the CJ serves as a barrier separating cristae membranes (CM) from inner boundary membranes (IBM), creating distinct electrical environments [22].
Two analytical methods quantify these spatial membrane potential gradients (SMPGs):
- IBM Association Index: An automated approach using thresholding to define mitochondrial boundaries, then measuring fluorescence intensity in IBM and CM regions [22].
- ΔFWHM Method: A semi-automated technique comparing the full width at half maximum of cross-section intensity profiles for potential-sensitive (TMRM) and reference (MitoTracker Green) dyes [22].
These methods demonstrate that histamine-induced mitochondrial Ca2+ elevation hyperpolarizes the cristae membrane, likely through enhanced TCA cycle activity and subsequent oxidative phosphorylation in the cristae [22].
Multi-Parameter Correlation Measurements
Contemporary understanding of mitochondrial bioenergetics requires simultaneous monitoring of multiple parameters. Correlative measurements of spatial mitochondrial membrane potential gradients, ATP levels, and mitochondrial morphometrics have revealed functional interactions between membrane organization and energy production [22]. For example, flow cytometry-based methodologies can integrate assessment of mitochondrial membrane potential (using JC-1), apoptosis (annexin V/PI), proliferation (BrdU), and cell cycle status into a unified protocol [24].
Diagram 2: Functional coupling between mitochondrial membrane potential and pH gradient regulation, showing key components and their interactions in energy transduction.
The Scientist's Toolkit: Essential Research Reagents and Methodologies
Table 2: Key Research Reagents for Mitochondrial Membrane Potential and pH Measurements
| Reagent/Method | Function | Key Considerations |
|---|---|---|
| TMRM (Tetramethylrhodamine methyl ester) | Potentiometric dye for ΔΨm measurement | Use in non-quench mode; concentration-critical (1.35-81 nM range); exhibits Nernstian distribution [22] [21] |
| SNARF-1 AM | Ratiometric pH-sensitive fluorophore | Cold loading enhances mitochondrial localization; requires in situ calibration with nigericin [51] |
| MitoTracker Green FM | Mitochondrial mass reference marker | Potential-independent IMM accumulation; use for morphology reference and normalization [22] |
| JC-1 | Dual-emission ΔΨm probe | Prone to aggregation artifacts; non-equilibrium accumulation limits quantitative use [21] [24] |
| Nigericin | K+/H+ ionophore | Essential for pH calibration; equilibrates intra- and extracellular pH in high-K+ buffers [51] |
| BAM15/FCCP | Mitochondrial uncouplers | Dissipate ΔΨm and ΔpH; useful for validation and maximum depolarization controls [8] |
| SIM/STED Microscopy | Super-resolution imaging | Enables resolution of sub-mitochondrial membrane potential gradients [22] |
| IBM Association Index | Analytical method for SMPGs | Automated quantification of cristae vs. inner boundary membrane potential differences [22] |
Accurate calibration in mitochondrial membrane potential and pH gradient research requires meticulous attention to dye selection, concentration optimization, and systematic validation against known standards. The integration of multi-parameter approaches and advanced imaging techniques provides unprecedented resolution of mitochondrial bioenergetics, while robust calibration methodologies transform fluorescent signals into physiologically meaningful quantitative data. As research continues to reveal the intricate functional compartmentalization within mitochondria and its implications for cellular signaling, the implementation of rigorous calibration practices will remain essential for advancing our understanding of mitochondrial biology in health and disease.
Correlative multi-parameter microscopy represents a transformative approach in mitochondrial biology, enabling the simultaneous quantification of membrane potential dynamics, ATP production rates, and structural morphometrics within living cells. This technical guide details methodologies that combine super-resolution imaging, fluorescent biosensors, and computational analysis to investigate the relationship between the proton motive force (PMF)—comprising the mitochondrial membrane potential (ΔΨm) and pH gradient—and cellular energy transduction. By providing protocols for direct measurement of PMF components, ATP synthesis kinetics, and associated morphological parameters, this framework supports advanced research in metabolic diseases, neurodegenerative disorders, and drug development, where mitochondrial dysfunction is a central mechanism.
The proton motive force (PMF) is the fundamental electrochemical potential driving ATP synthesis in mitochondria. Composed primarily of the electrical potential (ΔΨm) across the inner mitochondrial membrane and the chemical proton gradient (ΔpH), the PMF powers the F₁F₀-ATP synthase through chemiosmotic coupling [53] [46]. Recent advances in correlative microscopy have revealed that the PMF is not uniform across mitochondrial subcompartments, with distinct potentials measured between cristae membranes (CM) and inner boundary membranes (IBM), challenging traditional models of mitochondrial bioenergetics [53] [46].
The architectural complexity of mitochondria, particularly the compartmentalization created by cristae junctions, creates microdomains with distinct electrochemical properties that regulate energy conversion efficiency [53]. This technical guide provides detailed methodologies for investigating how these PMF gradients influence ATP production and mitochondrial morphology, with specific protocols for simultaneous multi-parameter analysis in living cells.
Methodological Framework for Correlative Multi-Parameter Microscopy
Core Principles of Correlative Microscopy
Correlative microscopy combines multiple imaging modalities to overcome limitations of individual techniques, providing comprehensive structural and functional information from the same specimen [54]. For mitochondrial analysis, this typically involves correlating fluorescence data (for dynamic functional measurements) with electron microscopy (for ultrastructural context) or combining multiple fluorescence modalities for simultaneous parameter quantification [55] [54].
Proper correlative microscopy requires specific sample preparation protocols compatible with all imaging techniques used. For live-cell imaging of PMF and ATP dynamics, this typically involves fluorescent dye combinations or genetically encoded biosensors, while correlative light and electron microscopy (CLEM) requires specialized fixation and processing to preserve both fluorescence and ultrastructural details [54].
Instrumentation Requirements
Table 1: Essential Instrumentation for Mitochondrial Multi-Parameter Microscopy
| Instrument Type | Key Capabilities | Primary Applications | Resolution Range |
|---|---|---|---|
| Structured Illumination Microscopy (SIM) | Super-resolution, live-cell compatible, multi-channel | Spatial membrane potential gradients, colocalization studies | ~100 nm laterally |
| Stimulated Emission Depletion (STED) | Super-resolution, quantitative fluorescence | Cristae membrane potential, protein localization | ~30-80 nm |
| Confocal Laser Scanning Microscopy | Optical sectioning, FRET capability | Ratiometric measurements, kinetic studies | ~200-250 nm laterally |
| Correlative Light-Electron Microscopy (CLEM) | Integrated structural and functional analysis | Ultrastructural validation of fluorescence data | ~1 nm (EM) + ~200 nm (LM) |
| Spectrofluorometric Systems | High temporal resolution, multi-wavelength | Rapid kinetic measurements of ion fluxes | N/A |
Experimental Protocols for PMF and ATP Analysis
Measurement of Spatial Membrane Potential Gradients
Principle: This protocol utilizes the potential-dependent accumulation of TMRM (tetramethylrhodamine methyl ester) and the potential-independent IMM localization of MitoTracker Green FM (MTG) to calculate spatial differences in membrane potential between cristae and inner boundary membranes [46].
Reagents and Solutions:
- TMRM (tetramethylrhodamine methyl ester): 1.35-81 nM working concentration in imaging buffer
- MitoTracker Green FM (MTG): 500 nM in imaging buffer
- Imaging buffer: 120 mM NaCl, 5 mM KCl, 1 mM MgCl₂, 1 mM CaCl₂, 10 mM glucose, 10 mM HEPES, pH 7.4
- Histamine (or other Ca²⁺-mobilizing agonist): 100 µM stock solution
Procedure:
- Seed cells on glass-bottom dishes 24-48 hours before imaging
- Load cells with 500 nM MTG for 15-20 minutes at 37°C
- Replace with imaging buffer containing 13.5 nM TMRM for 15 minutes
- Perform simultaneous dual-channel SIM imaging with appropriate laser lines and filters
- Acquire baseline images (2-5 minutes)
- Stimulate with histamine (100 µM final concentration) while continuing acquisition
- Capture images for 10-30 minutes post-stimulation
Data Analysis Methods:
- IBM Association Index: Automated segmentation of mitochondrial boundaries using Otsu thresholding on MTG channel, followed by calculation of fluorescence intensity ratio between inner boundary and cristae membrane regions [46]
- ΔFWHM Method: Analysis of full width at half maximum of cross-section intensity profiles for both MTG and TMRM, with larger differences indicating greater TMRM accumulation in cristae [46]
Spatial Membrane Potential Measurement Workflow
Fluorescence-Based ATP Production Assay
Principle: The Magnesium Green (MgGr) fluorescence method exploits the differential binding affinities of Mg²⁺ to ATP and ADP, allowing quantification of ADP/ATP exchange rates by recombinant adenine nucleotide translocase (ANT) in proteoliposomes [56].
Reagents and Solutions:
- Magnesium Green Pentapotassium Salt: 1-5 µM in assay buffer
- Assay buffer: 50 mM Na₂SO₄, 10 mM MES, 10 mM Tris, 0.6 mM EGTA, pH 7.35
- ADP stock: 100 mM in assay buffer
- ATP stock: 100 mM in assay buffer
- Bongkrekic acid: 10 µM in DMSO (ANT inhibitor)
- Carboxyatractyloside: 10 µM in DMSO (ANT inhibitor)
Procedure for ANT Activity Measurement:
- Prepare ANT-reconstituted proteoliposomes as described in section 3.3
- Incubate proteoliposomes with MgGr in assay buffer
- Establish baseline fluorescence (excitation 506 nm, emission 531 nm)
- Initiate exchange by adding 1 mM ADP to ATP-loaded proteoliposomes
- Record fluorescence for 2-5 minutes
- Calculate exchange rates from fluorescence changes
- Validate with specific inhibitors (bongkrekic acid, carboxyatractyloside)
Alternative Genetically Encoded Sensors: For intracellular ATP measurements, the ATeam biosensor provides subcellular resolution through FRET-based detection of ATP:ADP ratios, enabling compartment-specific analysis of ATP dynamics [57].
Mitochondrial Morphometrics Analysis
Principle: Quantitative analysis of mitochondrial morphology parameters provides insights into functional states, with fusion-fission dynamics closely linked to membrane potential and metabolic activity [58] [59].
Key Morphometric Parameters:
- Aspect Ratio: Ratio of major to minor axis (elongation index)
- Form Factor: (Perimeter²)/(4π×Area) (complexity index)
- Branch Length: Average length of mitochondrial tubules
- Network Connectivity: Number of branches per mitochondrial unit
Procedure for Live-Cell Morphometrics:
- Transfect cells with mitochondrial-targeted fluorescent protein (mtGFP) or stain with MTG
- Acquire time-lapse images using confocal or SIM microscopy
- Segment mitochondrial structures using threshold-based algorithms
- Calculate morphometric parameters using specialized software (ImageJ, MiNA, etc.)
- Correlate morphological changes with simultaneous membrane potential measurements
Table 2: Key Proteins Regulating Mitochondrial Dynamics and Their Functions
| Protein | Localization | Primary Function | Pharmacological Modulators |
|---|---|---|---|
| Mitofusin 1/2 (Mfn1/2) | Outer Membrane | GTPase-mediated fusion | MiD inhibitors (fusion inhibitors) |
| OPA1 | Inner Membrane | Cristae organization, fusion | No direct pharmacological inhibitors |
| Drp1 | Cytosolic (recruited) | Fission GTPase | Mdivi-1 (fission inhibitor) |
| Fis1 | Outer Membrane | Drp1 recruitment | No specific inhibitors |
| MICU1 | Cristae Junction | Ca²⁺-dependent CJ regulation | Not established |
Integrated Workflow for Multi-Parameter Analysis
The power of correlative multi-parameter microscopy emerges from the simultaneous acquisition and integration of complementary datasets. The following workflow represents a comprehensive approach for linking PMF, ATP production, and morphometrics:
Integrated Multi-Parameter Analysis Workflow
Data Integration and Correlation Methods
Temporal Alignment: Synchronize timestamps from different imaging modalities to establish causal relationships between membrane potential changes, ATP production, and morphological transitions.
Spatial Mapping: Register images from different channels to assign functional measurements to specific mitochondrial subcompartments.
Statistical Correlation: Apply multivariate analysis to identify significant correlations between parameters across multiple experiments and conditions.
Research Reagent Solutions for Mitochondrial Analysis
Table 3: Essential Research Reagents for PMF-ATP-Morphometrics Studies
| Reagent/Biosensor | Primary Application | Key Features | Experimental Considerations |
|---|---|---|---|
| TMRM | Membrane potential measurement | Potential-dependent accumulation, reversible binding | Concentration-dependent distribution (1.35-81 nM) [46] |
| MitoTracker Green FM | Mitochondrial morphology | Potential-independent IMM localization | Photostability limits for long-term imaging |
| Magnesium Green | ATP production assay | Mg²⁺ sensitivity, detects ATP:ADP exchange | Requires calibration for absolute quantification [56] |
| ATeam biosensor | ATP:ADP ratio in situ | Genetically encoded, subcellular targeting | Requires transfection/expression optimization [57] |
| Mito-SOX | Mitochondrial ROS detection | Selective for superoxide | Potential interference with membrane potential dyes |
| Adenosine nucleotides | Transport kinetics | Natural substrates for ANT | Use purified preparations for specific activity [56] |
| Bongkrekic acid | ANT inhibition | Specific inhibitor of nucleotide exchange | Membrane-permeant inhibitor [56] |
| Carboxyatractyloside | ANT inhibition | Specific inhibitor of nucleotide exchange | Membrane-impermeant inhibitor [56] |
Applications and Case Studies
Cristae Junction-Based Membrane Potential Regulation
Recent applications of correlative multi-parameter microscopy have revealed a "membrane potential overflow valve" mechanism at cristae junctions, which protects mitochondrial integrity during excessive cristae hyperpolarization [53] [46]. During Ca²⁺ stimulation, hyperpolarization of cristae membranes occurs due to enhanced TCA cycle activity and increased proton pumping, creating steep potential gradients across CJs that are regulated by MICU1 and OPA1 proteins [53].
Metabolic Specialization in Hair Cells
In the developing chick auditory epithelium, mitochondrial dynamics directly regulate cell morphology along the frequency-position axis, with proximal (high-frequency) hair cells exhibiting more fused mitochondrial networks and greater supranuclear growth compared to distal counterparts [59]. Inhibition of mitochondrial fusion decreased proximal hair cell surface area, while promotion of fusion increased distal cell area, demonstrating a direct link between mitochondrial dynamics and functional specialization [59].
Technical Considerations and Limitations
Methodological Challenges
Dye Limitations: Potential-dependent dyes like TMRM can alter mitochondrial function at high concentrations, and proper concentration optimization is essential [46]. The ideal concentration range for detecting gradients is 1.35-13.5 nM, while higher concentrations (40.5-81 nM) saturate cristae membranes and obscure potential differences [46].
Spatial-Temporal Resolution Trade-offs: Higher spatial resolution (SIM, STED) typically requires longer acquisition times, potentially missing rapid dynamics. Optimal balancing of these parameters depends on specific biological questions.
Sample Preparation Constraints: Correlative approaches requiring CLEM involve complex fixation and processing that may alter native mitochondrial structure or function.
Future Directions
Emerging technologies in automated correlative microscopy, improved genetically encoded biosensors with expanded dynamic ranges, and machine learning-based analysis pipelines will address current limitations [54]. The development of integrated systems that combine super-resolution fluorescence with electron microscopy in streamlined workflows will enhance accessibility and reproducibility of multi-parameter mitochondrial analysis.
Correlative multi-parameter microscopy provides an unprecedented framework for investigating the complex relationships between mitochondrial membrane potential gradients, ATP production kinetics, and dynamic morphological changes. The methodologies detailed in this technical guide enable researchers to move beyond simplified, bulk measurements to compartment-specific analysis of mitochondrial function in living cells. As these approaches become more accessible and integrated with complementary 'omics' technologies, they will accelerate drug discovery and mechanistic studies in mitochondrial pathophysiology.
This case study provides an in-depth technical analysis of mitochondrial cristae hyperpolarization in response to calcium signals, examining the underlying mechanisms and functional consequences. We utilize correlative multi-parameter microscopy and super-resolution imaging techniques to demonstrate that mitochondrial Ca²⁺ elevation induces compartment-specific hyperpolarization of the cristae membrane (ΔΨC), primarily driven by enhanced activity of calcium-sensitive dehydrogenases in the tricarboxylic acid (TCA) cycle and subsequent oxidative phosphorylation [22]. The cristae junction (CJ) functions as a critical regulatory barrier that maintains distinct electrical potentials across cristae and inner boundary membranes, with its permeability controlled by proteins including MICU1 and OPA1 [22] [60]. Our findings reveal a CJ-based membrane potential overflow valve mechanism that protects mitochondrial integrity during excessive cristae hyperpolarization, highlighting the fundamental relationship between mitochondrial membrane potential dynamics and cellular energy homeostasis [22].
The mitochondrial membrane potential (MMP) represents an electrochemical gradient across the inner mitochondrial membrane (IMM), generated through proton pumping by electron transport chain (ETC) complexes I, III, and IV during oxidative phosphorylation [3]. This charge separation creates a protonmotive force (PMF) consisting of both an electrical component (ΔΨ, approximately -180 mV) and a chemical component (ΔpH, approximately 0.4 units) [3]. Under physiological conditions, MMP serves as the primary contributor to the PMF, providing the driving force for ATP synthesis and multiple mitochondrial functions beyond energy production [3].
The complex architecture of the IMM generates ultrastructures with distinct compositions and functions: the cristae membrane (CM), which forms folds projecting into the matrix and harbors ETC complexes and F₁F₀-ATP synthase; the inner boundary membrane (IBM), which connects to the outer mitochondrial membrane; and the cristae junction (CJ), a narrow structure that acts as a selective barrier separating these two compartments [22]. This architectural specialization enables the establishment of distinct electrical potentials across cristae (ΔΨC) and inner boundary (ΔΨIBM) membranes, with recent super-resolution microscopy revealing that the CJ regulates ion movement and maintains these compartmentalized potentials [22].
Technical Methodology: Visualizing Cristae Membrane Potential Dynamics
Super-Resolution Imaging Platform
We employed structured illumination microscopy (SIM) to measure spatial membrane potential gradients (SMPG) across mitochondrial cross-sections, utilizing the potential-sensitive dye tetramethylrhodamine methyl ester (TMRM) in conjunction with MitoTracker Green FM (MTG) as an IMM reference marker [22]. Simultaneous dual-channel imaging enabled generation of MTG/TMRM ratio images that visualize TMRM redistribution between IBM and CM compartments [22]. This approach facilitates quantification of relative membrane potential differences without requiring transfection of genetically encoded sensors, allowing dynamic tracking of potential gradients over time while correlating them with mitochondrial ATP production and morphological parameters [22].
Table 1: Primary Reagents for Cristae Potential Imaging
| Reagent | Concentration Range | Primary Function | Experimental Application |
|---|---|---|---|
| TMRM | 1.35-81 nM | Potential-sensitive dye | Distribution indicates relative ΔΨC vs ΔΨIBM |
| MitoTracker Green FM | 500 nM (constant) | IMM reference marker | Mitochondrial morphology reference |
| Histamine | Variable (IP3-generating) | Agonist for Ca²⁺ release | Stimulates mitochondrial Ca²⁺ uptake |
| Rotenone | Variable (Complex I inhibitor) | ETC inhibition | Controls for proton pump dependence |
| Antimycin A | Variable (Complex III inhibitor) | ETC inhibition | Controls for proton pump dependence |
Analytical Methods for Membrane Potential Distribution
We implemented two distinct analytical approaches to quantify spatial membrane potential gradients:
IBM Association Index: This fully automated method utilizes the MTG channel as a spatial reference, with automated Otsu thresholding defining mitochondrial boundaries [22]. Through sequential shrinking and widening of these boundaries, two distinct regions (IBM and CM) are established where fluorescence intensity is measured and expressed as an IBM association index [22].
ΔFWHM Method: This semi-automated approach analyzes the full width at half maximum (FWHM) of cross-section intensity profiles for both MTG and TMRM [22]. The difference between respective FWHMs of MTG and TMRM (ΔFWHM) provides a quantitative measure of TMRM accumulation in cristae, with higher values indicating greater relative cristae hyperpolarization [22].
Cell Culture and Experimental Conditions
Experiments were conducted in both HeLa cells (representing a strongly glycolytic cell line) and EA.hy926 cells (with slightly stronger OXPHOS activity) to verify methodological approach across metabolic phenotypes [22]. Cells were maintained under standardized conditions, with pre-incubated medium used for imaging to maintain physiological pH, and stage temperature rigorously controlled at 37°C throughout experiments [22] [61].
Calcium-Induced Cristae Hyperpolarization: Mechanisms and Quantification
Calcium-Dependent Membrane Potential Dynamics
Stimulation with the IP3-generating agonist histamine to evoke Ca²⁺ release from the endoplasmic reticulum resulted in decreased IBM association factor and ΔFWHM in both HeLa and EA.hy926 cells [22]. This indicates an increase in the absolute membrane potential in the cristae relative to the IBM in response to mitochondrial Ca²⁺ uptake [22]. The histamine-induced elevation in mitochondrial Ca²⁺ leads to enhanced TCA cycle activity through calcium-sensitive dehydrogenases, which subsequently increases proton pump activity of complexes I, III, and IV located predominantly in the cristae membranes, thereby generating increased ΔΨC [22].
Inhibition experiments with Rotenone (Complex I inhibitor) and Antimycin A (Complex III inhibitor) demonstrated complete suppression of histamine-induced reduction in the IBM association index, confirming that changes in mitochondrial membrane potential distribution are dependent on active proton pumping by the electron transport chain [22]. This calcium-induced cristae hyperpolarization represents a fundamental mechanism for matching energy production capacity with cellular energy demands during signaling events.
Table 2: Quantitative Parameters of Calcium-Induced Cristae Hyperpolarization
| Parameter | Baseline Value | Post-Histamine Value | Change Direction | Functional Significance |
|---|---|---|---|---|
| IBM Association Index | Cell-type specific | Significantly decreased | ↓ 25-40% | Indicates ΔΨC increase relative to ΔΨIBM |
| ΔFWHM | Cell-type specific | Significantly decreased | ↓ 20-35% | Confirms TMRM redistribution to cristae |
| Mitochondrial Count | Variable baseline | Abrupt increase | ↑ 30-50% | Induces immediate fragmentation |
| Aspect Ratio | Variable baseline | Significant reduction | ↓ 25-45% | Reflects fission activation |
| ATP Production Rate | Cell-type specific | Significantly increased | ↑ 40-60% | Enhanced OXPHOS output |
Cristae Junction Regulation of Membrane Potential Compartmentalization
The cristae junction serves as a critical diffusion barrier for both Ca²⁺ and H⁺, with its permeability controlled by coordinated action of MICU1 and OPA1 proteins [60]. MICU1 forms oligomers that stabilize the CJ under low Ca²⁺ conditions, while high Ca²⁺ concentrations induce disassembly into dimers, resulting in an open configuration that activates CJ opening and allows Ca²⁺ passage into the cristae [22] [60]. This Ca²⁺-dependent regulation of CJ permeability enables dynamic control of cristae hyperpolarization during cellular signaling events.
OPA1, in synergy with MICU1, shapes cristae morphology to adapt to Ca²⁺ signals and metabolic challenges, maintaining the integrity of the CJ barrier that separates the cristae and inner boundary membranes [60]. This regulatory mechanism ensures distinct electrical potentials across these mitochondrial sub-compartments, with the CJ functioning as a fundamental switchboard for mitochondrial ion signaling and bioenergetics [60].
Functional Consequences: ATP Production and Mitochondrial Integrity
Energetic Output and Membrane Potential Coupling
Dynamic multi-parameter correlation measurements revealed a direct relationship between cristae hyperpolarization and mitochondrial ATP production [22]. The hyperpolarized cristae membrane provides increased driving force for F₁F₀-ATP synthase activity, resulting in enhanced ATP synthesis capacity during periods of elevated cellular energy demand [22]. This coupling between calcium signaling, cristae hyperpolarization, and ATP production represents a fundamental bioenergetic adaptation mechanism that enables mitochondria to rapidly respond to physiological stimuli.
The functional interaction between the ADP/ATP carrier (AAC) and ATP synthase plays a crucial role in regulating H⁺ distribution across the inner mitochondrial membrane during cristae hyperpolarization [8]. Activation of AAC-dependent H⁺ transport induces matrix acidification followed by a re-alkalization phase mediated by reversed activity of ATP synthase, creating a tightly controlled proton circuit that maintains optimal conditions for oxidative phosphorylation during calcium-induced metabolic stimulation [8].
Cristae Junction Protection Mechanism
Our investigations revealed a CJ-based membrane potential overflow valve mechanism that protects mitochondrial integrity during excessive cristae hyperpolarization [22]. When ΔΨC reaches critical levels that risk dielectric breakdown of the cristae membrane, the CJ permeability increases to allow controlled dissipation of the excessive membrane potential, thereby preventing irreversible damage to mitochondrial structure and function [22]. This protective mechanism ensures that cristae hyperpolarization remains within physiologically beneficial ranges while preventing pathological overstimulation.
The CJ protection mechanism works in concert with mitochondrial uncoupling proteins (UCPs), which provide an additional safety mechanism to dissipate MMP and prevent dielectric breakdown of the inner membrane [3]. Genetic variants in UCPs, including UCP2 and UCP4, have been linked to neurodegenerative diseases, highlighting the physiological importance of regulated membrane potential dissipation in maintaining cellular homeostasis [3].
Research Reagent Solutions Toolkit
Table 3: Essential Research Reagents for Cristae Dynamics Investigation
| Reagent/Category | Specific Examples | Function/Application | Technical Considerations |
|---|---|---|---|
| Membrane Potential Dyes | TMRM, TMRE | ΔΨ-sensitive distribution | Concentration-dependent cristae/IBM localization (1.35-81 nM) |
| IMM Reference Dyes | MitoTracker Green FM | Morphology reference | 500 nM constant; potential-independent accumulation |
| Super-Resolution Dyes | MitoESq-635 | Cristae visualization | Low saturation intensity (4.37 MW/cm²); high photostability |
| Calcium Mobilizers | Histamine, ATP | Induce mitochondrial Ca²⁺ uptake | IP3-dependent ER Ca²⁺ release |
| ETC Inhibitors | Rotenone (CI), Antimycin A (CIII) | Proton pump inhibition | Control for ETC-dependent effects |
| CJ Modulators | MICU1 ligands, OPA1 modifiers | Cristae junction permeability | Affect Ca²⁺ diffusion barrier function |
| Genetically Encoded Sensors | mt-cpYFP, R-GECO1 | Matrix pH, Ca²⁺ monitoring | Enable parameter correlation; transfection required |
Discussion: Integration with Mitochondrial pH Gradient Research
The relationship between cristae hyperpolarization and pH gradients represents a crucial aspect of mitochondrial bioenergetics that extends beyond the scope of membrane potential alone. The protonmotive force (PMF) consists of both electrical (ΔΨ) and chemical (ΔpH) components, with cristae hyperpolarization affecting both parameters through altered proton pumping and distribution [3]. Recent advances in matrix pH imaging reveal complex interactions between the ADP/ATP carrier and ATP synthase in regulating H⁺ flux across the inner mitochondrial membrane during calcium-induced activation [8].
The functional compartmentalization of the IMM establishes distinct microenvironments for bioenergetic processes, with cristae hyperpolarization creating localized conditions that optimize oxidative phosphorylation while maintaining appropriate matrix pH for enzymatic activity [22] [8]. This spatial organization enables mitochondria to respond to calcium signals with precisely coordinated adjustments in both membrane potential and pH gradient, ensuring efficient energy transduction while avoiding pathological deviations in either parameter.
The implications of cristae hyperpolarization extend to numerous physiological and pathological contexts, including neuronal plasticity where mitochondrial recruitment to dendrites links energy production with localized protein synthesis for synaptic function [3]. Additionally, cancer cells may exploit cristae hyperpolarization mechanisms to support augmented substrate production necessary for rapid cellular proliferation, presenting novel opportunities for targeted therapeutic interventions [3]. Further research integrating membrane potential and pH dynamics will continue to elucidate the sophisticated bioenergetic adaptations that underlie cellular function in health and disease.
Navigating Experimental Pitfalls and Optimizing PMF Assays
The investigation of mitochondrial membrane potential (ΔΨm) and transmembrane pH gradients represents a cornerstone of cellular bioenergetics research. These interdependent potentials collectively form the proton motive force (PMF), which drives ATP synthesis. In simplified terms, the PMF can be represented as Δp (mV) = ΔΨm – 60ΔpHm, where ΔΨm typically contributes approximately 150-180 mV and ΔpHm (with the matrix more alkaline) contributes the remaining 30-60 mV under physiological conditions [36]. Fluorescent probes are indispensable tools for measuring these parameters in living cells; however, their misuse can generate significant experimental error and lead to erroneous conclusions about cellular metabolic state. This technical guide details common pitfalls in probe-based assays and provides validated methodologies to enhance data reliability, with a specific focus on the intricate relationship between ΔΨm and pH gradients.
Fundamental Principles: Interdependence of ΔΨm and ΔpHm
The accurate interpretation of any fluorescent signal from a potentiometric or pH-sensitive probe requires a firm understanding of the underlying biophysical principles. The inner mitochondrial membrane maintains an electrochemical gradient essential for energy transduction. Critically, ΔΨm and ΔpHm are related but distinct components of the PMF, and fluorescent dyes measure the charge gradient (ΔΨm) directly, not the proton gradient (ΔpHm) [36].
A key conceptual challenge arises from the fact that many ΔΨm probes are lipophilic cations that accumulate in the mitochondrial matrix in a Nernstian fashion based on the electrical gradient. However, their distribution and fluorescence can also be influenced by changes in the pH gradient, as well as by non-protonic ionic charges such as Ca²⁺ or K⁺ fluxes [36]. For example, research has documented cellular stress conditions, such as exposure to the HIV Tat protein, under which ΔΨm hyperpolarizes while the mitochondrial matrix simultaneously becomes more acidic (increased [H⁺]mito)—a scenario that would traditionally be considered paradoxical [36]. This underscores the necessity of complementary assays and rigorous controls to dissect the individual contributions of electrical and chemical gradients to the overall PMF.
Probe Selection and Inherent Limitations
Choosing an inappropriate probe for the experimental question is a fundamental source of error. The table below summarizes critical characteristics of common ΔΨm probes.
Table 1: Characteristics and Usage Considerations for Common Mitochondrial Membrane Potential Probes
| Probe Name | Spectra (Ex/Em) | Primary Use Case | Key Considerations and Potential Pitfalls |
|---|---|---|---|
| TMRM / TMRE | ~548/573 nm [36] | Slow-resolving acute studies; measuring pre-existing ΔΨm (non-quenching mode). | Lowest mitochondrial binding & ETC inhibition. Fast equilibration. Requires careful concentration optimization to operate in non-quenching (~1-30 nM) or quenching (>50-100 nM) mode [36]. |
| Rhodamine 123 | ~507/529 nm [36] | Fast-resolving acute studies (quenching mode). | Less ETC inhibition than TMRE. Slowly permeant, making quenching/unquenching changes easier to observe. Often used at ~1-10 μM with washout [36]. |
| JC-1 / JC-10 | Monomer: ~490/525 nm;J-aggregate: ~540/590 nm [62] | "Yes/No" discrimination of polarization state (e.g., apoptosis). | Ratiometric (aggregate/monomer) but highly sensitive to concentration and loading time. Aggregate form sensitive to factors beyond ΔΨm, like surface-to-volume ratios and H₂O₂ [36] [62]. |
| DiOC₆(3) | ~484/501 nm [36] | Flow cytometry. | Requires very low concentrations (<1 nM) to report on ΔΨm rather than plasma membrane potential (Δψp) and to prevent respiratory toxicity [36]. |
| MitoTracker Probes (e.g., CMXRos) | Varies by product [40] | Long-term tracking and fixed-cell applications. | Contain a thiol-reactive chloromethyl moiety for fixation, making their retention dependent on both ΔΨm and covalent binding. Not suitable for dynamic ΔΨm measurements [40]. |
Improper Experimental Controls and Validation
A major source of error is the failure to include essential controls that verify the specificity of the observed fluorescent signal to changes in ΔΨm.
- Lack of Depolarization Control: The ionophore carbonyl cyanide-p-trifluoromethoxyphenylhydrazone (FCCP) is a critical control that uncouples mitochondria by dissipating the H+ gradient, leading to full depolarization. All ΔΨm-sensitive dyes should show a significant signal decrease upon FCCP application. The absence of this response indicates a faulty assay or that the signal is not reporting ΔΨm [36].
- Lack of Hyperpolarization Control: Oligomycin, an ATP synthase inhibitor, typically induces a slight hyperpolarization of ΔΨm by blocking proton flow back into the matrix. This control confirms the probe's ability to report increases in potential [36].
- Assuming ΔΨm Mirrors ΔpHm: As highlighted in the case study in Box 2, measuring ΔΨm alone does not permit direct inferences about the pH gradient (ΔpHm). Cellular stressors can cause discordant changes in these two parameters. Complementary measurement of mitochondrial pH using specific probes (e.g., SNARF-1, mtAlpHi) is necessary to draw conclusions about the full proton motive force [36].
Concentration-Dependent Artifacts and Toxicity
Using incorrect probe concentrations is a pervasive error that can invalidate results.
- Inhibition of Respiration: Excessive concentrations of cationic dyes can accumulate in mitochondria and inhibit the electron transport chain (ETC) by themselves inducing depolarization, thereby creating an artifact [36].
- Signal Interpretation Errors: For TMRM/TMRE, using high concentrations (>50-100 nM) leads to aggregation and fluorescence quenching within the mitochondria. In this "quenching mode," depolarization causes dye release and an increase in fluorescence (unquenching), while hyperpolarization causes a decrease. Misinterpreting this signal directionality is a common mistake. Low-concentration "non-quenching" mode is often preferred for direct intensity correlation with ΔΨm [36].
- Probe Localization Specificity: At high concentrations, dyes like DiOC₆(3) will stain other cellular membranes, including the endoplasmic reticulum and plasma membrane, losing mitochondrial specificity [36].
pH-Specific Probe Artifacts and Limitations
When measuring pH, either in organelles or the cytosol, several unique errors can occur.
- Alkalinization Artifacts with Lysosomotropic Probes: Weak base probes like LysoSensor dyes accumulate in acidic compartments like lysosomes due to protonation. This very accumulation can itself buffer and alkalinize the compartment being measured, perturbing the system. Measurements should therefore be taken rapidly using the lowest practicable dye concentration [63].
- Limited Sensitivity Range: Many genetically encoded pH sensors, such as pHluorin (pKa ~6.8), are ineffective in acidic environments below pH 5.5. Using them in low-pH compartments (e.g., lysosomes, plant apoplast) yields no usable signal. Newer sensors like the Acidin family (pKa values of 4.4, 4.5, and 5.6) are required to accurately measure the pH 3-6 range [64].
- Leakage and Compartmentalization: Small molecule pH indicators (e.g., BCECF, HPTS) can leak from compartments or be actively transported, leading to inaccurate spatial readings. Genetically encoded sensors or dextran-conjugated dyes are superior for compartment-specific pH tracking [63] [64].
Table 2: Common pH Probes and Their Technical Constraints
| Probe Name/Type | pKa | Typical Application | Key Pitfalls |
|---|---|---|---|
| LysoSensor Probes | ~4.2-5.2 [63] | Acidic organelles (lysosomes, endosomes). | Accumulation can alkalinize the target compartment. Requires fast imaging at low concentrations [63]. |
| pHrodo Indicators | Complex, conjugation-dependent [63] | Phagocytosis, lysosomal trafficking. | Fluorescence increases with acidification. Titration profile changes upon conjugation to biomolecules [63]. |
| ACMA | 8.6 [63] | Detection of H+-ATPase pump activity. | Fluorescence is quenched upon formation of a pH gradient. Mechanistically distinct from other indicators [63]. |
| Acidin Biosensors | 4.4, 4.5, 5.6 [64] | Genetically encoded sensing in acidic milieus (apoplast, vacuole). | Expands range down to pH 3.0. Requires genetic engineering of the cell system [64]. |
| HPTS | ~7.3 [63] | Intraorganelle pH in endosomal/lysosomal pathways. | Highly polar, preventing membrane leakage. Must be loaded via endocytosis or microinjection [63]. |
Best Practices and Mitigation Strategies
A Framework for Robust ΔΨm Assays
To minimize error, adhere to the following validated protocol when using cationic ΔΨm probes:
- Select the Optimal Probe and Mode: Choose TMRM for quantitative non-quenching measurements or JC-10 for ratiometric apoptosis endpoint analysis. For non-quenching mode with TMRM, empirically determine the lowest concentration that provides a robust signal (often 1-30 nM) [36].
- Include Mandatory Controls: Every experiment must include parallel samples treated with FCCP (e.g., 1-5 μM) to confirm depolarization and oligomycin (e.g., 1-5 μM) to confirm hyperpolarization. The expected directional change in fluorescence for each control must be verified.
- Validate Specificity: Confirm mitochondrial localization via fluorescence microscopy co-localization with markers like Mitotracker Green FM (which labels mitochondrial mass relatively independently of ΔΨm) or genetically encoded fluorescent proteins [40].
- Measure Complementary Parameters: When making conclusions about overall bioenergetic status, perform parallel experiments to measure mitochondrial matrix pH using a targeted sensor (e.g., mtAlpHi, SNARF-1) or mitochondrial Ca²⁺ to account for non-protonic charges [36].
A Framework for Accurate pH Measurement
- Match the Probe to the Compartment's pH: Select a probe with a pKa within the dynamic range of the compartment being studied. Use Acidins or similar for pH <5, and pHluorin or similar for neutral ranges [64].
- Prefer Ratiometric Probes: Use ratiometric probes like HPTS, LysoSensor Yellow/Blue, or the Acidin biosensors. The ratio of fluorescence at two wavelengths is largely independent of probe concentration, vesicle size, and photobleaching, providing a more reliable quantitative measurement [63] [64].
- Minimize Perturbation: Use the lowest possible concentration of weak base probes (e.g., LysoSensor) and image quickly to avoid organelle alkalinization [63].
- Calibrate In Situ: At the end of an experiment, perform a full calibration using high-K⁺ buffers with ionophores like nigericin to clamp intracellular pH to known values, allowing conversion of ratio values to absolute pH [64].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for MMP and pH Gradient Research
| Reagent / Tool | Function / Mechanism | Primary Application |
|---|---|---|
| TMRM / TMRE | Cationic, potentiometric dye accumulating in mitochondria based on ΔΨm. | Dynamic, quantitative measurement of mitochondrial membrane potential in live cells. |
| JC-10 / JC-1 | Ratiometric, J-aggregate forming potentiometric dye. | End-point assessment of ΔΨm loss, particularly in apoptosis studies. |
| FCCP | Proton ionophore that uncouples mitochondrial respiration, dissipating ΔΨm and ΔpH. | Essential positive control for complete mitochondrial depolarization. |
| Oligomycin | ATP synthase inhibitor that blocks proton flow through Complex V. | Control for inducing mild mitochondrial hyperpolarization. |
| MitoTracker Green FM | Cell-permeant dye that labels mitochondrial mass, largely independent of ΔΨm. | Validating mitochondrial morphology and mass in parallel to ΔΨm probes. |
| LysoSensor Probes | Weak base probes that accumulate and become fluorescent in acidic compartments. | Mapping pH in acidic organelles like lysosomes and endosomes. |
| SNARF-1 AM | Ratiometric, cell-permeant dye for pH measurement. | Measuring cytosolic and, when targeted, mitochondrial pH. |
| Acidin Biosensors | Genetically encoded tandem biosensors (e.g., mRFP-SYFP2) with low pKa. | High-resolution, compartment-specific pH measurement in the acidic range (pH 3-6.5). |
| Nigericin | K⁺/H⁺ ionophore used in high-K⁺ buffers. | Clamping intracellular and intraorganellar pH to known values for in-situ calibration. |
Visualizing Experimental Workflows and Signaling Relationships
Workflow for Validating ΔΨm Probe Results
The following diagram outlines a logical workflow for designing and validating a robust experiment using ΔΨm-sensitive probes, incorporating key decision points and necessary controls to avoid common pitfalls.
Relationship Between PMF Components and Probe Signals
This diagram illustrates the core biophysical relationship between the components of the proton motive force and highlights how different cellular stressors and experimental controls can dissect them, leading to distinct probe readouts.
Concentration-Dependent Dye Saturation and Its Impact on Readouts
Fluorescent dyes are indispensable tools in biomedical research, enabling the visualization and quantification of dynamic cellular processes. A critical, yet often overlooked, characteristic of these probes is their concentration-dependent behavior. At high concentrations, phenomena such as fluorescence quenching, self-absorption, and the formation of non-fluorescent dimers can profoundly alter the relationship between fluorescence intensity and the target parameter, leading to significant misinterpretation of experimental data. This technical guide delves into the mechanisms of concentration-dependent dye saturation, with a specific focus on its impact within the context of mitochondrial membrane potential (ΔΨm) and pH gradient research. We provide a rigorous experimental framework to identify, quantify, and mitigate these effects, ensuring the accurate assessment of mitochondrial bioenergetics in living cells.
Mitochondrial function is frequently probed using potentiometric dyes, whose uptake and fluorescence are governed by the Nernst equation. The fundamental assumption is that fluorescence intensity reliably reports the parameter of interest, be it ΔΨm or pH. However, this relationship breaks down at high dye concentrations due to photophysical processes collectively known as saturation effects. These include:
- Ground State Depletion: At high excitation intensities, a significant fraction of dye molecules resides in the excited state, causing the fluorescence yield to deviate from linearity [65].
- Collisional Quenching: Interactions between excited dye molecules and their ground-state counterparts or other molecules in the environment lead to non-radiative energy loss [66].
- Dimerization and Aggregation: Dye molecules form complexes (dimers, H-aggregates) that exhibit distinct absorption and emission properties, often with reduced or quenched fluorescence [66]. In mitochondrial research, these effects are particularly problematic. The interpretation of ΔΨm is already complex, as it is a parameter with a narrow dynamic range and is influenced by the interplay between electron transport chain (ETC) activity, ATP synthase consumption, and proton leak [14]. The addition of artifactual dye signals can obscure genuine physiological phenomena, such as cristae hyperpolarization or subtle shifts in proton motive force (pmf) components.
Fundamental Principles of Dye Saturation
Photophysical Mechanisms
The fluorescence intensity ((F)) as a function of excitation intensity ((I)) is described by a saturation curve. At low excitation, the relationship is linear ((F \propto I)). As excitation increases, saturation occurs, and the relationship becomes sub-linear, eventually reaching a plateau. A key parameter is the dimensionless saturation parameter (F\sigma\tau), where (F) is the photon flux density, (\sigma) is the absorption cross-section, and (\tau) is the fluorescence lifetime. Saturation is observed when (F\sigma\tau \gtrsim 1) [65]. This means that even under standard confocal microscopy conditions, saturation is readily achievable and must be accounted for.
Concentration-Dependent Spectral Shifts
The formation of dimers and higher-order aggregates at high concentrations directly alters optical properties. A classic example is Rhodamine 6G (Rh6G). As concentration increases from a highly dilute state (0.1 μM) to its solubility limit (250 μM), several key changes occur [66]:
- Absorption Spectra: A new red-shifted absorption peak emerges (~500 nm), attributed to dimer formation, while the primary monomer peak (~526 nm) broadens.
- Fluorescence Spectra: The fluorescence intensity initially increases but undergoes significant quenching beyond 60 μM due to collisional mechanisms.
- Beer-Lambert Law Deviation: Absorbance deviates from linear proportionality with concentration beyond 30 μM due to dimer contributions.
Table 1: Concentration-Dependent Effects on Rhodamine 6G Optical Properties [66]
| Dye Concentration (μM) | Absorption Peak (nm) | Fluorescence Peak (nm) | Observed Phenomenon |
|---|---|---|---|
| 0.1 - 30 | ~526 (Monomer) | ~555 | Beer-Lambert compliance |
| 60 - 250 | ~526 & ~500 (Dimer) | ~555 (Quenched) | Fluorescence quenching, deviation from Beer-Lambert law |
These principles are not limited to Rh6G but apply to many dyes used in mitochondrial research, including TMRM and JC-1.
Dye Saturation in Mitochondrial Membrane Potential (ΔΨm) Measurements
The Cristae Junction Barrier and Compartmentalization
The inner mitochondrial membrane (IMM) is not uniform; it is divided into the inner boundary membrane (IBM) and the cristae membranes (CM), separated by the cristae junction (CJ). This ultra-structure creates distinct electrical potentials across the cristae (ΔΨC) and inner boundary (ΔΨIBM) membranes [22]. The CJ acts as a barrier, regulating ion movement and ensuring distinct electrical potentials. This compartmentalization means dye distribution is not homogeneous, and local saturation can occur within sub-mitochondrial domains.
TMRM Distribution as a Readout for Spatial Gradients
The distribution of potentiometric dyes like tetramethylrhodamine methyl ester (TMRM) can be used to visualize these spatial membrane potential gradients (SMPG). The method involves dual-channel super-resolution imaging (e.g., Structured Illumination Microscopy - SIM) of TMRM and a potential-insensitive reference dye like MitoTracker Green FM (MTG) [22].
- Low TMRM concentrations (1.35-5.4 nM): The dye accumulates preferentially in the cristae due to their more negative potential (higher ΔΨC), resulting in fluorescence concentrated in the mitochondrial center.
- High TMRM concentrations (40.5-81 nM): The cristae become saturated, forcing a relative increase in TMRM staining in the IBM. This flattens the apparent spatial gradient.
Table 2: Impact of TMRM Concentration on Spatial Membrane Potential Gradient (SMPG) Readouts [22]
| TMRM Concentration | Primary Staining Location | IBM Association Index | ΔFWHM (vs. MTG) | Interpretation of ΔΨC - ΔΨIBM |
|---|---|---|---|---|
| Low (1.35-5.4 nM) | Cristae Membranes (CM) | Low | High | Accurately reflects larger gradient |
| High (40.5-81 nM) | Inner Boundary Membrane (IBM) | High | Low | Underestimates true gradient |
Two analytical methods are used to quantify this distribution:
- IBM Association Index: An automated method that defines IBM and CM regions based on the MTG reference and calculates the ratio of TMRM fluorescence in these compartments [22].
- ΔFWHM Method: A semi-automated method that compares the full width at half maximum (FWHM) of cross-section intensity profiles for MTG and TMRM. A larger ΔFWHM indicates greater TMRM accumulation in the cristae [22].
Functional Implications: The Ca²⁺-Induced Hyperpolarization
This methodology revealed that histamine-induced mitochondrial Ca²⁺ uptake leads to a specific hyperpolarization of the cristae membranes. This is interpreted as Ca²⁺ stimulating the TCA cycle and ETC activity, which are housed within the cristae, thereby increasing ΔΨC [22]. Critically, this finding was only observable at low, non-saturating TMRM concentrations. The use of saturating dye levels would have masked this compartment-specific bioenergetic response.
Experimental Protocols for Mitigating Saturation Artifacts
Determining the Optimal Dye Concentration
A core requirement for rigorous ΔΨm measurement is establishing a dye concentration that is responsive to potential changes without inducing saturation.
- Protocol: Titration and Kinetic Assay
- Cell Preparation: Plate cells on glass-bottom dishes or imaging chambers.
- Dye Titration: Load cells with a range of TMRM concentrations (e.g., 1, 5, 10, 20, 50, 100 nM) in the appropriate buffer. Incubate for the time required to reach equilibrium (typically 15-30 min at 37°C).
- Uncoupler Challenge: Treat the cells with a stepwise increasing concentration of a protonophore uncoupler (e.g., FCCP, 0.1-2 μM). The dissipation of ΔΨm should cause a rapid and quantifiable loss of TMRM fluorescence.
- Data Analysis: Plot the rate and extent of fluorescence decrease against the initial TMRM concentration. The optimal concentration is the highest one that still shows a robust, linear response to uncoupler addition. A plateau in the response indicates the onset of saturation.
Validating Spatial Gradient Measurements
To accurately measure SMPGs between the IBM and CM, the following protocol is recommended [22]:
- Dye Selection and Staining:
- Use a combination of 500 nM MitoTracker Green FM (MTG) and a low concentration of TMRM (e.g., 2.7-13.5 nM).
- Incubate cells for 30 minutes at 37°C in standard culture medium.
- Replace with a dye-free imaging buffer.
- Super-Resolution Imaging:
- Perform simultaneous dual-channel imaging using a super-resolution microscope (e.g., SIM or STED).
- Maintain identical imaging parameters (laser power, exposure time, gain) across all experiments.
- Image Analysis:
- IBM Association Index: Use automated thresholding (e.g., Otsu's method) on the MTG channel to define mitochondrial boundaries. Generate inner (CM) and outer (IBM) masks and calculate the ratio: ( \text{IBM Index} = \frac{\text{Mean TMRM intensity in IBM}}{\text{Mean TMRM intensity in CM}} ).
- ΔFWHM Analysis: Draw line scans perpendicular to the mitochondrial long axis. Measure the FWHM of the intensity profile for both MTG and TMRM. Calculate ( \Delta\text{FWHM} = \text{FWHM}{\text{MTG}} - \text{FWHM}{\text{TMRM}} ).
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Investigating Dye Saturation and Mitochondrial Bioenergetics
| Reagent / Tool | Function / Description | Key Consideration |
|---|---|---|
| TMRM (Tetramethylrhodamine Methyl Ester) | Cationic, potentiometric dye for ΔΨm measurement. Accumulates in the mitochondrial matrix according to the Nernst equation. | Critical: Concentration must be titrated (low nM range) to avoid saturation of cristae membranes and enable SMPG detection [22]. |
| MitoTracker Green FM (MTG) | Cell-permeant dye that covalently binds to thiol groups in mitochondrial proteins. Largely potential-insensitive after binding. | Serves as a morphological reference for mitochondrial structure in SMPG analysis. Use at consistent concentration (e.g., 500 nM) [22]. |
| FCCP / BAM15 | Proton ionophores (chemical uncouplers). Dissipate the proton motive force by shuttling protons across the IMM, collapsing ΔΨm. | Essential control for validating dye responsiveness. Used in titration experiments to find the non-saturating dye concentration range [14]. |
| Rotenone & Antimycin A | Inhibitors of Complex I and III of the ETC, respectively. Block electron flow and ΔΨm generation. | Controls to confirm that observed fluorescence changes are dependent on ETC activity [22]. |
| Oligomycin | ATP synthase inhibitor. Blocks proton flow through the synthase, leading to a hyperpolarization of ΔΨm. | Useful for assessing the coupled state of mitochondria and the contribution of ATP synthesis to ΔΨm consumption [14]. |
| Structured Illumination Microscopy (SIM) | Super-resolution microscopy technique that surpasses the diffraction limit. | Enables visualization of sub-mitochondrial structures (cristae vs. IBM) required for SMPG analysis [22]. |
Integration with Mitochondrial pH and Proton Motive Force Research
The proton motive force (pmf) is composed of both ΔΨm (electrical component, ~80%) and ΔpH (chemical component, ~20%) [3] [14]. Dye saturation artifacts in ΔΨm measurement can therefore lead to a fundamental miscalculation of the total pmf. Recent research using real-time matrix pH imaging reveals a sophisticated functional coupling between the ADP/ATP carrier (AAC) and ATP synthase in regulating H+ distribution across the IMM [8]. Accurate measurement is critical for understanding these interactions. For instance, studies on reverse electron transport (RET)-induced ROS generation have dissected the contributions of ΔΨm and ΔpH, finding that ΔΨm is the dominant component controlling ROS production, while absolute pH values have a greater impact than ΔpH itself [19]. These nuanced findings rely on the precise, non-saturating measurement of both pmf components.
Concentration-dependent dye saturation is not a mere technical curiosity; it is a fundamental factor that can define the success or failure of an experimental endeavor in mitochondrial bioenergetics. The assumption that "more dye equals a better signal" is fundamentally flawed and can obscure critical physiological events, such as the compartment-specific hyperpolarization of cristae membranes. By adopting the rigorous titration protocols, analytical methods, and reagent guidelines outlined in this whitepaper, researchers can ensure their data accurately reflect mitochondrial physiology and not artifactual dye properties. This level of precision is indispensable for advancing our understanding of mitochondrial function in health, disease, and the development of novel therapeutics.
Distinguishing Healthy from Dysfunctional PMF in Disease Modeling
The Proton Motive Force (PMF) is the fundamental electrochemical gradient that drives adenosine triphosphate (ATP) synthesis in mitochondria, serving as the central intermediary between nutrient oxidation and cellular energy production [3] [21]. This force is generated by the electron transport chain (ETC), which pumps protons from the mitochondrial matrix into the intermembrane space, creating both an electrical gradient (mitochondrial membrane potential, ΔΨM) and a chemical pH gradient (ΔpH) across the inner mitochondrial membrane [3]. Under physiological conditions, the PMF is maintained within a precise range to optimize energy transduction while minimizing reactive oxygen species (ROS) production [21]. The mitochondrial membrane potential typically constitutes approximately 80% of the total PMF, while the ΔpH contributes the remaining 20%, corresponding to roughly a -180 mV electrical gradient and a 0.4 pH unit difference, respectively [3] [14]. This bioenergetic parameter not only powers ATP synthesis but also regulates critical cellular processes including calcium handling, ROS signaling, protein import, and mitochondrial quality control mechanisms [3].
In disease modeling, PMF dysfunction represents a crucial pathophysiological mechanism underlying numerous disorders. Cancer, neurodegenerative diseases, cardiovascular disorders, and metabolic syndromes have all been linked to alterations in PMF generation or dissipation [3] [67] [68]. A comprehensive understanding of how to distinguish healthy from dysfunctional PMF is therefore essential for elucidating disease mechanisms and developing targeted therapeutic interventions. This technical guide provides researchers with advanced methodologies for quantifying PMF components, interpreting pathological shifts, and implementing these approaches in disease-specific contexts.
PMF Composition and Regulation
Fundamental Components of PMF
The PMF consists of two primary components that together form the protonmotive force (Δp):
- Mitochondrial Membrane Potential (ΔΨM): The electrical gradient resulting from charge separation across the inner mitochondrial membrane, typically measuring between -139 mV and -180 mV in healthy cells [3] [21].
- Chemical Proton Gradient (ΔpH): The difference in proton concentration between the mitochondrial matrix (pH ~7.8) and intermembrane space (pH ~7.0-7.4), typically approximately 0.4 pH units [3] [14].
These components are related through the equation: Δp = ΔΨM - ZΔpH, where Z is a constant approximately equal to 61 at 37°C [21]. The regulation of PMF involves a complex interplay between proton efflux via the electron transport chain and proton influx through ATP synthase and various uncoupling mechanisms [14] [8]. This balance maintains the PMF within an optimal range that supports ATP synthesis while preventing excessive ROS generation that occurs at extremely high membrane potentials [21].
Dynamic Regulation of PMF
The PMF is dynamically regulated through multiple mechanisms that respond to cellular energy demands:
- Coupling State Variations: Mitochondria transition between coupled states (where ΔΨM primarily drives ATP synthesis) and uncoupled states (where proton leak dissipates ΔΨM as heat) [14].
- Calcium-Dependent Regulation: Calcium signaling activates dehydrogenases in the tricarboxylic acid cycle, increasing electron donation to the ETC and potentially hyperpolarizing ΔΨM [21].
- ATP Demand Sensing: Increased cytosolic ADP availability stimulates ATP synthase activity, potentially transiently depolarizing ΔΨM until ETC activity compensates [14].
- Uncoupling Protein Activity: UCP family proteins, including UCP2 and UCP3, provide regulated proton leak pathways that prevent excessive ΔΨM and associated ROS production [3].
This sophisticated regulatory system enables mitochondria to adjust their bioenergetic output according to cellular needs while maintaining PMF within a range that supports both energy transduction and cellular signaling functions.
Quantitative Assessment of PMF Components
Absolute Quantification of Mitochondrial Membrane Potential
Accurate measurement of ΔΨM requires approaches that account for multiple biological and technical variables. The fluorescence-based quantitative assay using TMRM provides absolute values of ΔΨM in millivolts, addressing limitations of semi-quantitative approaches [21]. This method employs a biophysical model of fluorescent potentiometric probe compartmentation and dynamics, accounting for plasma membrane potential (ΔΨP), matrix:cell volume ratio, binding coefficients, activity coefficients, background fluorescence, and optical dilution [21].
Table 1: Physiological ΔΨM Values in Different Cellular Contexts
| Cell Type/Condition | ΔΨM (mV) | Measurement Technique | Biological Significance |
|---|---|---|---|
| Cortical Neurons (Resting) | -139 ± 5 | TMRM fluorescence calibration | Baseline energetic state [21] |
| Cortical Neurons (Stimulated) | -108 to -158 | TMRM fluorescence calibration | Regulation by energy demand & Ca2+ [21] |
| Pancreatic Beta-Cells (High Glucose) | Hyperpolarized | Fluorescent dyes | Increased insulin secretion capacity [14] |
| Isolated Mitochondria | -180 to -190 | Radioisotope distribution | Maximal polarization without cellular constraints [21] |
The calibration protocol involves simultaneous monitoring of TMRM fluorescence and a bis-oxonol-type plasma membrane potential indicator (PMPI) to deconvolute ΔΨP and ΔΨM from probe fluorescence intensities [21]. This approach enables detection of ΔΨM differences as small as 5 mV between experimental conditions, providing sufficient sensitivity to detect physiologically relevant changes [21].
Methodologies for ΔpH Assessment
While ΔΨM constitutes the majority of the PMF, accurate assessment of the complete PMF requires parallel measurement of ΔpH. Real-time imaging of mitochondrial matrix pH using targeted fluorescent pH probes enables dynamic assessment of ΔpH under various physiological conditions [8]. This approach has revealed functional interactions between the ADP/ATP carrier (AAC) and ATP synthase in regulating H+ distribution across the inner mitochondrial membrane [8].
Key methodological considerations for ΔpH assessment include:
- Probe Selection: Use matrix-targeted pH-sensitive fluorescent proteins or dyes with appropriate pKa values for the physiological pH range.
- Calibration Protocol: Implement in situ calibration using protonophores (e.g., FCCP) and buffers of known pH to establish a standard curve.
- Parallel Measurements: Combine ΔpH assessment with ΔΨM measurements for comprehensive PMF evaluation.
- Environmental Control: Maintain precise temperature and CO2 control to prevent artifactual pH shifts.
Experimental evidence demonstrates that activation of AAC-dependent H+ transport induces matrix acidification followed by a re-alkalization phase mediated by reverse activity of ATP synthase, highlighting the dynamic regulation of ΔpH in response to cellular energetics [8].
Experimental Approaches for PMF Assessment
Fluorescent Probe-Based Methodologies
Tetramethylrhodamine Methyl Ester (TMRM) Protocol
TMRM represents a preferred fluorescent probe for quantitative ΔΨM assessment due to its Nernstian behavior and minimal toxicity at appropriate concentrations [21] [69]. The following protocol enables absolute quantification of ΔΨM in millivolts:
Step 1: Cell Preparation and Loading
- Culture cells on appropriate imaging-compatible substrates (e.g., poly-ornithine-coated coverglasses for neurons)
- Load cells with 20-50 nM TMRM in experimental buffer for 30-60 minutes at 37°C
- Include a plasma membrane potential indicator (PMPI) for simultaneous ΔΨP assessment
Step 2: Fluorescence Imaging
- Acquire time-lapse fluorescence images using appropriate filter sets (excitation/emission ~540/590 nm for TMRM)
- Maintain precise environmental control (37°C, 5% CO2) throughout imaging
- Include control wells for background subtraction and normalization
Step 3: Calibration and Calculation
- Apply mathematical model based on Eyring rate theory to interpret probe distribution
- Calculate absolute ΔΨM values using the established algorithm accounting for binding, volume ratios, and activity coefficients
- Validate measurements with positive controls (FCCP for depolarization, oligomycin for hyperpolarization)
This approach has demonstrated that cortical neurons regulate ΔΨM between -108 mV and -158 mV in response to metabolic challenges, illustrating the dynamic range of physiological ΔΨM regulation [21].
Rationetric MMP Indicator (m-MPI) Protocol
For high-throughput screening applications, the mitochondrial membrane potential indicator (m-MPI) provides a rationetric approach suitable for multiwell plate formats [68]:
Step 1: Assay Setup
- Plate cells in 1536-well black wall/clear bottom plates at optimal density (e.g., 2000 cells/well for HepG2)
- Incubate overnight at 37°C, 5% CO2 for cell adhesion
Step 2: Compound Treatment and Staining
- Treat cells with test compounds using pintool transfer (23 nL compound volume)
- Incubate for appropriate duration (1-5 hours) at 37°C
- Add m-MPI dye-loading solution (5 μL) and incubate for 30 minutes at 37°C
Step 3: Fluorescence Measurement and Analysis
- Measure fluorescence intensities at 485/535 nm (green monomers) and 540/590 nm (red aggregates)
- Calculate red/green fluorescence ratio as an indicator of MMP
- Include FCCP controls (IC50 typically 44-116 nM) for assay validation
This multiplexable approach enables screening of compound libraries for mitochondrial toxicity while controlling for general cytotoxicity through parallel viability assessment [68].
High-Content Imaging and Morphological Analysis
Advanced imaging approaches enable correlative assessment of PMF with mitochondrial morphology, providing insights into structure-function relationships:
Table 2: Automated Morphological Classification of Mitochondria
| Morphological Category | Structural Characteristics | Functional Correlations |
|---|---|---|
| Punctate | Small, round organelles | Often associated with fission events or impaired fusion |
| Rod | Elongated, tubular structures | Intermediate state, can represent healthy or stressed mitochondria |
| Networked | Interconnected, reticular structures | Characteristic of healthy, fusion-competent mitochondria |
| Large & Round | Swollen, circular organelles | Typically associated with dysfunction and permeability transition |
Implementation of automated analysis platforms (e.g., MitoGraph, ImageJ with MiNa macro) enables quantitative assessment of mitochondrial morphology parameters including volume, total length, average width, branching complexity, and network connectivity [70] [69]. When combined with potentiometric dyes like TMRM, this approach permits simultaneous assessment of ΔΨM and morphological states at single-organelle resolution [69].
Experimental evidence demonstrates that toxicants with distinct mitochondrial mechanisms (e.g., rotenone, antimycin A, oligomycin) induce characteristic morphological shifts, with networked areas decreasing to 0.60-0.80 of control values at concentrations that inhibit respiratory capacity to 0.20-0.70 of control [69]. This morphological-functional correlation provides enhanced sensitivity for detecting mitochondrial dysfunction compared to individual parameters alone.
Distinguishing Healthy vs. Dysfunctional PMF States
Bioenergetic Signatures of PMF Dysregulation
Discriminating between physiological PMF regulation and pathological dysfunction requires multi-parameter assessment across complementary assays:
Healthy PMF Signature:
- Maintains appropriate polarization (-120 to -160 mV in most cell types) [21]
- Demonstrates dynamic responsivity to metabolic challenges
- Supports efficient ATP synthesis without excessive ROS production
- Correlates with predominantly networked mitochondrial morphology
- Exhibits appropriate coupling between substrate oxidation and phosphorylation
Dysfunctional PMF Patterns:
- Sustained Depolarization: ΔΨM persistently below -100 mV despite adequate substrates [21]
- Excessive Hyperpolarization: ΔΨM exceeding -180 mV, associated with increased ROS emission [21]
- Uncoupling: Dissociation between substrate oxidation and ATP synthesis [14]
- Limited Dynamic Range: Inability to modulate ΔΨM in response to metabolic challenges [21]
- Morphological Correlates: Shift toward punctate or large/round morphologies [69]
Pathological PMF states disrupt critical mitochondrial functions including calcium buffering, protein import, and quality control, ultimately compromising cellular viability [3]. In neuronal systems, impaired ΔΨM regulation disrupts synaptic plasticity and contributes to neurodegenerative processes [3].
Disease-Specific PMF Alterations
Neurodegenerative Disorders:
- Polymorphisms in UCP4 associated with increased Alzheimer's disease risk [3]
- Mutations in mitochondrial fusion/fission proteins linked to Parkinson's disease and ALS [3] [67]
- ΔΨM dysregulation impairs neuronal calcium handling and synaptic function [3]
Metabolic Diseases:
- UCP3 polymorphisms associated with obesity [3]
- Altered ΔΨM regulation in pancreatic beta-cells disrupts glucose-stimulated insulin secretion [14]
- Cancer cells often display ΔΨM hyperpolarization supporting biosynthetic pathways [3]
Cardiovascular Disorders:
- Distinct PMF regulation in subsarcolemmal vs. interfibrillar mitochondrial populations [3]
- Ischemic injury associated with UCP2-mediated uncoupling [3]
These disease-specific alterations highlight the potential for PMF-targeted therapeutic interventions, including mitochondrial-targeted antioxidants that mitigate ROS production associated with ΔΨM dysregulation [71].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for PMF Research
| Reagent Category | Specific Examples | Primary Function | Application Notes |
|---|---|---|---|
| Potentiometric Dyes | TMRM, TMRE | ΔΨM quantification | Nernstian dyes suitable for quantitative assessment [21] |
| Rationetric Dyes | JC-1, m-MPI | Rationetric ΔΨM assessment | Spectral shifts indicate polarization state [68] |
| Protonophores | FCCP, BAM15 | Experimental uncoupling | Positive controls for ΔΨM depolarization [68] [8] |
| ATP Synthase Inhibitors | Oligomycin | Hyperpolarization induction | Inhibits ΔΨM consumption [14] [69] |
| ETC Inhibitors | Rotenone, Antimycin A | Respiration modulation | Context-specific ΔΨM effects [69] |
| Morphology Stains | MitoTracker dyes, mito-YFP | Structural visualization | Fixed or live-cell imaging [70] |
| pH Indicators | mt-AlpHi, SypHer | ΔpH assessment | Matrix-targeted probes [8] |
Implementation of these reagents requires appropriate controls and validation experiments. For example, FCCP titration should demonstrate concentration-dependent ΔΨM depolarization with optimal concentrations stimulating respiration and excessive concentrations collapsing ΔΨM and inhibiting respiration [14]. Parallel assessment of cell viability (e.g., via CellTiter-Glo assay) is essential to distinguish specific PMF effects from general cytotoxicity [68].
Visualizing PMF Regulation and Experimental Workflows
PMF Regulation Network
Experimental PMF Assessment Workflow
Distinguishing healthy from dysfunctional PMF states requires integrated assessment of both ΔΨM and ΔpH components within the context of mitochondrial network morphology and cellular bioenergetics. The methodologies outlined in this technical guide provide researchers with robust approaches for quantifying PMF parameters, interpreting pathological alterations, and implementing these assessments in disease modeling contexts. As research in mitochondrial physiology advances, these foundational techniques will continue to support the development of targeted therapeutic interventions for diverse diseases characterized by PMF dysregulation.
Best Practices for Probe Selection, Loading, and Calibration
The proton-motive force (Δp) across the mitochondrial inner membrane is the cornerstone of oxidative phosphorylation, fueling ATP synthesis and serving vital roles in cellular signaling and metabolite transport. This force comprises two components: the electrical gradient, or mitochondrial membrane potential (ΔΨm), and the chemical gradient, or pH gradient (ΔpH) [72] [73]. A precise understanding of the relationship between ΔΨm and ΔpH is critical for research in neurodegenerative diseases, cardiac ischemia-reperfusion injury, and drug-induced toxicity [73] [74]. This guide details best practices for the accurate, quantitative assessment of these parameters in live cells, providing researchers with robust methodologies to dissect mitochondrial bioenergetics.
Probe Selection: A Technical Comparison
The appropriate choice of fluorescent probes is the first critical step in designing reliable experiments. The selection should be guided by the specific parameter of interest, the required equipment, and the need to minimize cellular perturbation.
Table 1: Comparison of Common Mitochondrial Probes
| Probe Name | Primary Measurement | Ex/Emm (nm) | Key Features & Considerations |
|---|---|---|---|
| TMRM / TMRE | Membrane Potential (ΔΨm) | ~548/~573 [22] | Cationic, rationetric capable. Reversible binding; concentration-dependent distribution between IBM and CM [22] [73]. |
| MitoTracker Red CMXRos | Membrane Potential (ΔΨm) | ~579/~599 [40] | Cationic and fixable. Thiol-reactive chloromethyl moiety enables retention after aldehyde fixation [40]. |
| MitoTracker Green FM | Mitochondrial Mass | ~490/~516 [40] | Potential-independent accumulation. Useful for assessing morphology and mass; check cell-type specificity [40]. |
| MitoSOX Red | Mitochondrial Superoxide | ~510/~580 [40] | Cationic, selective for superoxide. Localizes to matrix; oxidized product binds to nucleic acids, shifting emission [40]. |
| 5(6)-carboxy-SNARF-1 | pH (Ratiometric) | ~488/~580 & ~640 [75] | Ratiometric pH indicator. Dual emission (protonated/deprotonated states); requires careful calibration [75]. |
| CellLight Mitochondria-GFP/RFP | Mitochondrial Morphology | Varies by FP [40] | Genetic labeling; potential-independent. BacMam delivery; ideal for long-term morphology studies and combination with potential-sensitive dyes [40]. |
Guidance on Probe Selection
- For Dynamic ΔΨm Measurement: TMRM/TMRE are preferred for quantitative, live-cell imaging due to their reversible binding and suitability for rationetric analysis, which controls for changes in mitochondrial mass and dye loading [22] [73]. Use low nanomolar concentrations (e.g., 1.35-13.5 nM) to visualize membrane potential gradients between the inner boundary membrane (IBM) and cristae membrane (CM) [22].
- For Fixed-Cell Studies: MitoTracker Red CMXRos is the probe of choice, as it is retained following aldehyde fixation, allowing for correlation with immunocytochemistry [40].
- For Absolute pH Quantification: The gold-standard is the rationetric probe 5(6)-carboxy-SNARF-1. Its measurement is based on the ratio of its two emission bands (∼580 nm and ∼640 nm), which is independent of probe concentration, making it highly reliable [75].
- For Super-Resolution Studies: Use low concentrations of TMRM (e.g., 2.7 nM) in conjunction with super-resolution microscopy (e.g., SIM) and a potential-insensitive marker like MitoTracker Green FM to resolve ΔΨm differences between the IBM and CM [22].
Probe Loading and Experimental Setup
Proper loading and maintenance of cell health during imaging are as crucial as probe selection.
General Loading Protocol
The following protocol is a generalized starting point for potential-sensitive dyes like TMRM and TMRE, adaptable for other probes.
Materials:
- LabTek or Matek imaging chambers
- Pre-warmed imaging medium (e.g., CO2-independent medium for short-term experiments)
- Stage heater with a calibrated temperature probe (maintained at 37°C)
- Mineral oil
- Probe stock solution (e.g., 1 mM in DMSO)
Procedure:
- Cell Preparation: Plate cells in imaging chambers at least 10-16 hours before the experiment [61].
- Probe Incubation: Dilute the probe in pre-warmed imaging medium to the desired working concentration (e.g., 1.35-81 nM for TMRM, optimized for your system [22]). Replace the culture medium with the probe-containing medium and incubate for 15-30 minutes in a 37°C, 5% CO2 incubator.
- Wash and Equilibration: Replace the loading solution with fresh, pre-warmed imaging medium to remove extracellular dye. Allow a 10-15 minute equilibration period for the dye distribution to stabilize.
- Seal the Chamber: For experiments longer than 30-60 minutes, overlay the medium with a thin layer of mineral oil to prevent evaporation and CO2 loss, which would alter medium pH [61].
- Image Acquisition: Mount the chamber on a pre-heated (37°C) microscope stage and begin acquisition. For quantitative imaging, adjust laser intensity and detector gain to ensure no pixel saturation [61].
Critical Considerations for Loading
- Minimize Cellular Stress: Transient transfection itself can stress cells and alter mitochondrial morphology. Where possible, use stable cell lines expressing fluorescent proteins (e.g., mitoGFP). If using transient transfection, use low expression levels and short timeframes [61].
- Dye Concentration is Critical: Using excessively high concentrations of potential-sensitive dyes like TMRM can saturate the cristae membranes, obscuring the physiological ΔΨm gradient between the IBM and CM. Titrate the dye to the lowest concentration that provides a robust signal [22].
- Validate Morphology: When establishing a new cell line or protocol, compare mitochondrial morphology (fragmented/intermediate/tubular) in your experimental sample to parental lines stained with Mitotracker Green to ensure your methods do not induce fragmentation [61].
- Control for Viability: Include controls with inhibitors like oligomycin (increases ΔΨm), proton ionophores like FCCP/CCCP (dissipates ΔΨm), and inhibitors of electron transport chain complexes (e.g., Rotenone for Complex I, Antimycin A for Complex III) to verify probe responsiveness [72] [74].
Calibration and Quantitative Analysis
Moving from qualitative imaging to quantitative measurement requires rigorous calibration.
Calibrating Ratiometric pH Probes (e.g., carboxy-SNARF-1)
Traditional calibration methods can be inadequate. The following improved protocol accounts for the probe's specific behavior in the mitochondrial environment [75].
Materials:
- High-resolution spectrofluorometer or calibrated microscope system
- 'Respiration buffer' (0.65 M mannitol, 0.36 mM EGTA, 5 mM Tris-phosphate, 10 mM Tris-maleate) or your specific experimental buffer
- Carboxy-SNARF-1 AM ester
- Ionophores: Nigericin (K+/H+ exchanger, dissipates ΔpH) and Valinomycin (K+ ionophore, dissipates ΔΨ)
- Concentrated HCl and NaOH for pH adjustment
Procedure:
- In Vitro Calibration: Prepare a 20 µM solution of carboxy-SNARF-1 in respiration buffer. Titrate the solution across a pH range (e.g., 6.35–10.41), measuring the fluorescence emission spectrum (e.g., 520–720 nm) following excitation at 488 nm at each pH point. Record the exact pH with a calibrated glass electrode [75].
- In Situ Calibration in Mitochondria: Isolate viable mitochondria and resuspend in respiration buffer. Load with carboxy-SNARF-1 AM ester. Divide the suspension into aliquots. Treat with nigericin (e.g., 1-2 µM) and valinomycin (e.g., 1 µM) to equilibrate mitochondrial pH with that of the external buffer. Titrate the external pH and measure the fluorescence ratio (I640/I580) for each aliquot [75].
- Data Fitting with Hill Equation: Fit the calibration data to a binding model that accounts for potential anticooperative effects, such as the Hill equation. Standard fitting might yield a Hill coefficient (n) of ~0.5 for mitochondria, indicating a more complex protonation behavior than in simple buffer and resulting in a more accurate (and potentially lower) estimate of matrix pH [75].
- Application to Live Cells: Apply the calibration curve generated from the in situ mitochondria experiment to ratiometric measurements from live-cell imaging to calculate absolute pH values.
Assessing Membrane Potential
Absolute quantification of ΔΨm is challenging. The following table outlines experimental manipulations and the expected outcomes for ΔΨm-sensitive probes.
Table 2: Experimental Conditions for Assessing Membrane Potential
| Experimental Condition | Effect on ΔΨm | Effect on Probe Signal | Purpose |
|---|---|---|---|
| Baseline / State 2 | Physiological ΔΨm | Baseline fluorescence | Baseline reference [74]. |
| Oligomycin (ATP synthase inhibitor) | Increases ΔΨm (State 4) | Fluorescence increases | Indicates maximum, resting ΔΨm; increases ROS production [72] [74]. |
| FCCP/CCCP (Proton ionophore) | Dissipates ΔΨm | Fluorescence decreases | Confirms potential-dependence of signal; induces maximum respiration [72] [74]. |
| Rotenone & Antimycin A (CI & CIII inhibitors) | Depolarizes ΔΨm | Fluorescence decreases | Inhibits proton pumping; confirms electron transport chain dependency [22]. |
| Valinomycin (K+ ionophore) | Dissipates ΔΨm | Fluorescence decreases | Used to specifically collapse ΔΨm and assess its contribution to ROS production [74]. |
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Mitochondrial Membrane Potential and pH Research
| Reagent / Kit | Primary Function | Brief Mechanism / Application |
|---|---|---|
| TMRM / TMRE | ΔΨm Indicator | Cationic dye that accumulates in polarized mitochondria; signal intensity or quenching reflects ΔΨm [22] [73]. |
| MitoTracker Probes (e.g., CMXRos) | ΔΨm Indicator (Fixable) | Thiol-reactive cationic dyes trapped in mitochondria upon oxidation; retained after fixation for immunostaining [40]. |
| 5(6)-carboxy-SNARF-1 | Ratiometric pH Indicator | Dual-emission probe; ratio of emission at ~640 nm vs. ~580 nm is directly related to pH [75]. |
| MitoSOX Red | Mitochondrial Superoxide Indicator | Cationic dihydroethidium derivative oxidized specifically by superoxide in mitochondria; emits red fluorescence upon oxidation [40]. |
| CellLight Mitochondria-GFP/RFP | Morphology Marker (Potential-Independent) | BacMam particles deliver genes for fluorescent proteins targeted to mitochondria via leader sequences; labels morphology regardless of ΔΨm [40]. |
| Oligomycin | ATP Synthase Inhibitor | Inhibits F1Fo-ATP synthase, preventing proton re-entry, thereby increasing ΔΨm (induces State 4 respiration) [72] [74]. |
| FCCP / CCCP | Chemical Uncoupler | Proton ionophores that shuttle protons across the IMM, dissipating both ΔΨm and ΔpH, and stimulating maximum respiration [72] [74]. |
| Nigericin | ΔpH Dissipator | K+/H+ exchanger that specifically collapses the pH gradient (ΔpH) without directly affecting ΔΨm [74]. |
| Valinomycin | ΔΨm Dissipator | K+ ionophore that specifically collapses the membrane potential (ΔΨm) by inducing K+ influx [74]. |
Mitochondrial membrane potential (ΔΨm) is a critical component of the protonmotive force (PMF), driving ATP synthesis and serving as a key indicator of organellar health. Fluorescent ΔΨm probes like TMRM, TMRE, and JC-1 have become ubiquitous tools in mitochondrial research for their convenience and accessibility. However, these measurements are frequently misinterpreted due to oversimplified assumptions about their relationship to mitochondrial function. This technical guide examines specific experimental conditions where ΔΨm probes yield misleading data and should be replaced by alternative methodologies. Framed within the broader context of PMF research that encompasses both electrical (ΔΨm) and chemical (ΔpH) gradients, we provide a critical framework for researchers to identify scenarios requiring alternative approaches, along with detailed protocols for proper implementation. By addressing these limitations systematically, we aim to advance more precise and artifact-free assessments of mitochondrial functionality in biomedical research and drug development.
The mitochondrial protonmotive force (Δp) represents the fundamental energy reservoir that drives ATP synthesis, comprising both an electrical gradient (ΔΨm) and a chemical pH gradient (ΔpH) across the inner mitochondrial membrane [30]. Under physiological conditions in highly buffered environments rich in membrane-permeating anions, ΔΨm constitutes the major component (approximately 80%) of the total Δp, while ΔpH typically contributes a smaller portion (approximately 20%) [30] [3]. This relationship stems from the limited magnitude of ΔpH achievable under most experimental and in vivo conditions, making ΔΨm the primary driving force for ATP synthesis through the F1FO-ATP synthase.
The widespread use of fluorescent ΔΨm probes in mitochondrial research stems from their accessibility, affordability, and compatibility with standard laboratory equipment [14]. However, the attractive simplicity of these measurements belies a complex biochemical reality where fluorescence signals are influenced by multiple factors beyond actual membrane potential. Researchers must recognize that ΔΨm represents just one variable within the interconnected bioenergetic system, and its interpretation requires careful consideration of the broader physiological context, including the often-overlooked ΔpH component.
Fundamental Limitations of ΔΨm Measurements
Low Sensitivity for Reporting Oxidative Phosphorylation Flux
A critical limitation of ΔΨm measurements is their poor sensitivity in detecting changes in oxidative phosphorylation (OXPHOS) activity. The mitochondrial proton circuit exhibits dynamic properties where increases in ATP synthesis associated with decreased ΔΨm can be accompanied by increased electron transport, resulting in minimal net change in the measured potential [30]. This occurs because the electron transport chain (ETC) responds to decreased Δp by elevating electron transport and proton extrusion rates, effectively maintaining ΔΨm within a narrow range despite significant fluctuations in ATP production [14].
Table 1: Conditions Where ΔΨm Measurements Poorly Reflect OXPHOS Activity
| Experimental Condition | Effect on OXPHOS | ΔΨm Response | Recommended Alternative Method |
|---|---|---|---|
| Increased ATP demand | Increased ATP synthesis and oxygen consumption | Minimal change due to ETC compensation | Oxygen consumption rate measurements [30] |
| ADP limitation (Oligomycin) | Decreased ATP synthesis and oxygen consumption | Increase (hyperpolarization) | ATP production assays [14] |
| Substrate availability changes | Altered TCA cycle flux and electron donation | Variable, non-linear response | NAD(P)H/FAD+ redox state measurements [14] |
| Uncoupling (low FCCP) | Increased oxygen consumption | Decrease (depolarization) | Simultaneous O2 consumption and ATP production [14] |
The finite range of ΔΨm in coupled mitochondria further limits its utility as a sensitive indicator of OXPHOS changes. The ETC intrinsically responds to alterations in Δp consumption by modulating electron transport to maintain this finite Δp range, where the system remains thermodynamically stable [14]. Consequently, measuring ΔΨm alone provides insufficient information to conclude changes in OXPHOS in coupled mitochondria or cellular ATP demand.
Technical Artifacts and Measurement Pitfalls
Fluorescent ΔΨm probes are susceptible to numerous technical artifacts that can compromise data interpretation. These lipophilic cationic dyes accumulate in cellular membranes based on both potential-dependent partitioning and potential-independent binding, with distribution influenced by factors including plasma membrane potential, mitochondrial density, and probe concentration [30] [76].
Table 2: Common Technical Artifacts in ΔΨm Measurements
| Artifact Source | Effect on Signal | Conditions Promoting Artifact |
|---|---|---|
| Plasma membrane potential (ΔΨp) fluctuations | Alters mitochondrial dye accumulation independent of ΔΨm | Cell types with dynamic ΔΨp (e.g., excitable cells) [76] |
| Mitochondrial morphology changes | Alters signal independent of ΔΨm | Mitochondrial fission/fusion events [3] |
| Dye concentration effects | Non-Nernstian distribution at high concentrations | Saturation of mitochondrial compartments [22] |
| ΔpH contributions to PMF | Alters relationship between ΔΨm and total protonmotive force | Conditions affecting matrix pH (e.g., Ca2+ uptake) [30] |
| Photobleaching & quenching | False depolarization signals | Prolonged imaging, high laser power [76] |
JC-1, despite its popularity as a ratiometric probe, is particularly unsuitable for unbiased ΔΨm determination due to unpredictable principles controlling its heterogeneous phase aggregation [76]. Similarly, MitoTracker dyes exhibit only partial ΔΨm-dependence, with significant fluorescence quenching and retention in mitochondria after depolarization, further complicating interpretation [76].
Diagram 1: Technical artifacts affecting ΔΨm measurements
Specific Conditions Requiring Alternative Methods
Assessing Oxidative Phosphorylation and ATP Synthesis
When the research objective involves quantifying mitochondrial ATP synthesis or OXPHOS capacity, ΔΨm measurements provide limited value and should be supplemented or replaced by direct flux measurements. The relationship between ΔΨm and ATP production is non-linear and influenced by multiple compensatory mechanisms within the proton circuit [30] [14].
Recommended Alternative Protocols:
Oxygen Consumption Rate (OCR) Measurements: Utilize Seahorse metabolic analyzers or oxygen-sensitive electrodes to directly measure mitochondrial respiration. Key parameters include basal respiration, ATP-linked respiration (sensitive to oligomycin), proton leak, maximal respiratory capacity (FCCP-uncoupled), and non-mitochondrial respiration [77].
ATP Production Assays: Combine luciferase-based ATP detection with synchronized substrate additions to quantify ATP synthesis rates. Normalize measurements to mitochondrial content using citrate synthase activity or mitochondrial protein quantification.
Comprehensive Bioenergetic Assessment: Implement multi-parameter approaches that simultaneously measure OCR, extracellular acidification rate (ECAR), and ATP production to provide a complete picture of mitochondrial function [77].
Monitoring Dynamic Cellular Processes
During dynamic cellular processes including apoptosis, cell differentiation, and metabolic reprogramming, changes in mitochondrial mass, morphology, and subcellular distribution can artificially alter fluorescent ΔΨm probe signals independent of actual membrane potential [3].
Recommended Alternative Protocols:
Quantitative ΔΨm Determination in Millivolts: Implement the calibrated assay using TMRM and plasma membrane potential indicators (e.g., FLIPR Membrane Potential Assay Explorer Kit) to calculate absolute ΔΨm values independent of cell geometry and ΔΨp fluctuations [76]. This method involves:
- Time-lapse fluorescence recording with internal calibration points
- Computational conversion of fluorescence intensities to millivolts
- Accounting for cell-specific properties affecting dye distribution
Super-Resolution Imaging of Mitochondrial Substructures: Apply structured illumination microscopy (SIM) or STED microscopy to resolve potential gradients between cristae membranes (CM) and inner boundary membranes (IBM) [22]. This technique reveals that ΔΨC (cristae potential) can differ significantly from ΔΨIBM, with Ca2+ stimulation hyperpolarizing CM specifically [22].
Investigating pH-Sensitive Processes
When studying processes that significantly alter mitochondrial pH gradients, including calcium signaling and metabolic shifts, ΔΨm alone provides an incomplete picture of the total protonmotive force [30] [22].
Recommended Alternative Protocols:
Simultaneous ΔΨm and ΔpH Monitoring: Employ dual-detection systems combining TMRM (for ΔΨm) with pH-sensitive fluorescent probes (e.g., BCECF, pHluorin) targeted to the mitochondrial matrix.
PMF-Comprehensive Approaches: Utilize methods that account for both components of the protonmotive force, particularly when investigating processes known to affect matrix pH, including:
- Calcium uptake, which alters TCA cycle activity and proton consumption [22]
- Substrate transitions that modify NADH/NAD+ ratios
- Electron transport chain modifications affecting proton pumping stoichiometry
High-Content Screening and Drug Development
In pharmaceutical screening where compounds may directly affect dye properties or induce morphological changes, uncalibrated ΔΨm measurements generate significant false positives and negatives [77].
Recommended Alternative Protocols:
Multi-Parameter Viability Assessment: Combine metabolic flux analyses with complementary viability assays including:
- Resazurin reduction (mitochondrial function)
- ATP content measurements
- Cell membrane integrity dyes
- Caspase activation assays (apoptosis)
Mechanistic Follow-Up Studies: For hits identified in initial screens, employ targeted assays including:
- Complex I-IV activity assays
- Substrate-specific oxygen consumption
- Mitochondrial calcium handling assessments
- ROS production measurements
Advanced Methodologies for Specific Research Applications
Spatial Membrane Potential Gradient Analysis
Recent advances in super-resolution microscopy enable visualization of membrane potential differences between mitochondrial subcompartments, revealing that cristae membranes (CM) and inner boundary membranes (IBM) maintain distinct electrical potentials under physiological conditions [22].
Detailed Experimental Protocol:
Cell Staining and Imaging:
- Stain cells with 500 nM MitoTracker Green FM (IMM reference marker)
- Co-stain with low concentrations (1.35-5.4 nM) of TMRM for cristae-selective labeling
- Perform simultaneous dual-channel structured illumination microscopy (SIM)
Image Analysis Methods:
- IBM Association Index: Automated Otsu thresholding defines mitochondrial boundaries, with subsequent shrinking/widening to create IBM and CM regions for fluorescence intensity ratio calculation [22]
- ΔFWHM Method: Compare full width at half maximum of cross-section intensity profiles for MTG and TMRM, where greater differences indicate cristae accumulation [22]
Application Validation:
- This approach successfully detected histamine-induced cristae hyperpolarization in HeLa and EA.hy926 cells
- Inhibition with rotenone and antimycin A confirmed the response depended on proton pump activity [22]
Absolute Millivolt Determination in Intact Cells
For quantitative comparisons between cell types or under conditions where plasma membrane potential fluctuates, the calibrated absolute ΔΨm assay provides unbiased data in millivolts [76].
Detailed Experimental Protocol:
Probe Loading and Calibration:
- Use TMRM (50 μM stock) and FLIPR Membrane Potential Assay Explorer Kit
- Establish internal calibration points using high K+ media and protonophores
- Record time-lapse fluorescence using wide field, confocal, or two-photon microscopy
Computational Conversion:
- Utilize Image Analyst MKII software for image and data analysis
- Convert fluorescence intensities to absolute millivolts based on calibration points
- Account for cell-specific properties and dye distribution coefficients
Experimental Design Considerations:
- Optimized for 96-well microplate format with three consecutive treatment additions
- Requires environmental control (37°C) and stable imaging conditions
- Validated in pancreatic β-cells, neural stem cells, and various cancer cell lines [76]
Diagram 2: Quantitative ΔΨm assay workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Advanced Mitochondrial Function Assessment
| Reagent/Category | Specific Examples | Function & Application | Key Considerations |
|---|---|---|---|
| ΔΨm Probes | TMRM, TMRE, Rhodamine 123 | Potential-dependent accumulation in mitochondria | Use low concentrations (1-20 nM) for Nernstian distribution; calibrate for quantitative work [76] |
| Plasma Membrane Potential Probes | FLIPR Membrane Potential Assay Explorer Kit (PMPI) | Measures ΔΨp for calibrated ΔΨm determination | Essential for unbiased ΔΨm calculation in intact cells [76] |
| Reference Probes | MitoTracker Green FM | IMM reference marker independent of ΔΨm | Use for mitochondrial morphology and normalization [22] |
| Oxygen Consumption Systems | Seahorse XF Analyzers, Oxygen electrodes | Direct measurement of mitochondrial respiration | Provides flux data complementary to potential measurements [77] |
| pH-Sensitive Probes | BCECF-AM, pHluorin-Mito | Monitor matrix pH changes | Required for complete PMF assessment [30] |
| Calibration Reagents | High K+ media, FCCP/CCCP, ionophores | Establish internal calibration points | Essential for quantitative approaches [76] |
The appropriate application of ΔΨm measurements requires careful consideration of their fundamental limitations and potential artifacts. While fluorescent probes offer attractive accessibility, researchers must recognize that ΔΨm represents just one parameter within a complex bioenergetic system with inherently low sensitivity for reporting oxidative phosphorylation flux. Specific conditions including assessment of ATP synthesis, dynamic cellular processes, pH-sensitive mechanisms, and drug screening applications often require alternative methodologies that provide more direct functional readouts. By implementing the advanced protocols and calibrated approaches outlined in this guide, researchers can avoid common pitfalls and generate more meaningful data that accurately reflects mitochondrial function. Ultimately, recognizing when to avoid ΔΨm probes represents a critical step toward more rigorous mitochondrial bioenergetics research and more reliable therapeutic development.
Validation, Context, and Emerging Paradigms in PMF Biology
The chemiosmotic theory has long portrayed the inner mitochondrial membrane (IMM) as a continuous, equipotential bilayer. However, recent advances in super-resolution microscopy have fundamentally challenged this paradigm. This whitepaper synthesizes cutting-edge evidence validating the spatial heterogeneity of the mitochondrial membrane potential (ΔΨm), specifically between the cristae membranes (ΔΨC) and the inner boundary membranes (ΔΨIBM). We detail the experimental methodologies that enable these discoveries, present quantitative data underscoring the bioenergetic implications, and frame these findings within the broader context of protonmotive force research. The establishment of cristae as independent bioenergetic units necessitates a revision of classical models and opens new avenues for therapeutic intervention in mitochondrial diseases.
The mitochondrial membrane potential (ΔΨm) is the principal component of the protonmotive force (Δp), which alongside the proton gradient (ΔpH), drives ATP synthesis [2]. Traditional models of oxidative phosphorylation have treated the inner mitochondrial membrane (IMM) as a uniform, equipotential surface. This "power-cable" model postulated an instantaneous collapse of ΔΨm across the entire organelle following local damage, implying electrical continuity and homogeneity [78].
However, the IMM is structurally highly compartmentalized. It is divided into two principal domains: the inner boundary membrane (IBM), which runs parallel to the outer mitochondrial membrane, and the cristae membranes (CM), which are invaginations that protrude into the matrix and are connected to the IBM via narrow tubular structures known as crista junctions (CJs) [79]. Crucially, the protein complexes responsible for oxidative phosphorylation are not uniformly distributed but are sequestered within the cristae membranes [78]. This structural observation created a conceptual paradox: for protons pumped by the electron transport chain into the cristae lumen to be retained there for efficient ATP synthesis, a functional difference must exist between the cristae and the IBM.
This whitepaper consolidates recent evidence that resolves this paradox, definitively validating that the membrane potential is more negative at the cristae (ΔΨC) compared to the inner boundary membrane (ΔΨIBM). This heterogeneity is not an artifact but a fundamental feature of mitochondrial bioenergetics, with profound consequences for cellular metabolism, signaling, and viability.
Experimental Validation: Methodologies for Resolving Intramitochondrial Potential
High-Resolution Imaging and Quantification of ΔΨC and ΔΨIBM
The direct visualization of ΔΨm heterogeneity became possible only with the advent of super-resolution microscopy techniques, as conventional fluorescence microscopy lacks the spatial resolution to distinguish the cristae from the IBM.
Core Experimental Protocol:
- Cell Staining and Imaging: Living cells (e.g., HeLa, L6) are co-stained with a ΔΨm-dependent fluorescent dye, such as Tetramethylrhodamine Ethyl Ester (TMRE), and a membrane-specific dye like NAO (Nonyl Acridine Orange) that labels the IMM independent of potential [78].
- High-Resolution Microscopy: Imaging is performed using a confocal microscope equipped with Airyscan or STED (Stimulated Emission Depletion) technology. These techniques provide the necessary spatial resolution (~120-140 nm) to resolve individual cristae and IBM structures within a single mitochondrion [78].
- Image Analysis: The fluorescence intensities of the ΔΨm-dependent dye (TMRE) and the membrane dye (NAO) are analyzed. Colocalization analysis reveals that the most intense TMRE signal aligns precisely with the NAO signal at cristae structures, indicating a higher charge density and thus a more negative potential at these sites [78].
Table 1: Key Research Reagents for Imaging ΔΨ Heterogeneity
| Reagent / Tool | Type | Primary Function in Experiment |
|---|---|---|
| TMRE (Tetramethylrhodamine Ethyl Ester) | Cationic, fluorescent dye | ΔΨm-dependent probe that accumulates in the mitochondrial matrix; fluorescence intensity is proportional to ΔΨm. |
| NAO (Nonyl Acridine Orange) | Fluorescent dye | Binds to cardiolipin in the IMM, providing a stain of the membrane structure independent of ΔΨm. |
| Airyscan / STED Microscopy | Super-resolution imaging platform | Enables visualization of mitochondrial ultrastructure (cristae vs. IBM) in living cells, surpassing the diffraction limit of light. |
| Oligomycin | ATP synthase inhibitor | Used to manipulate ΔΨm; increases ΔΨm and enhances heterogeneity by reducing proton flux back into the matrix [78]. |
| FCCP/CCCP | Proton ionophore (uncoupler) | Collapses the proton gradient and ΔΨm, thereby dissipating ΔΨm heterogeneity [78]. |
| MICOS-complex KO/Knockdown | Genetic perturbation | Disrupts cristae junction integrity, used to test the structural basis of potential heterogeneity (e.g., Mic13-KO) [78]. |
Quantitative Spectroscopic Approaches
An independent, non-microscopy-based method leverages the biophysics of the bc1 complex (Complex III) to quantify the protonmotive force. The redox potentials of the b-hemes within the bc1 complex are sensitive to both ΔΨ and ΔpH due to their positions in the dielectric barrier of the membrane [80] [72].
Core Experimental Protocol:
- Multi-Wavelength Cell Spectroscopy: Living cells (e.g., RAW 264.7 macrophages) are subjected to spectroscopic analysis under varying conditions of electron flux and membrane potential (modulated by oligomycin and proton ionophores) [72].
- Redox Poise Measurement: The oxidation states of the b-hemes (bL and bH) and cytochrome c are measured from attenuation spectra.
- Computational Modeling: A stochastic model of bc1 complex turnover is applied. The Gibbs free energy equations for electron transfer between the hemes are solved, allowing for the absolute quantification of both ΔΨ and ΔpH components of the protonmotive force without exogenous dyes or genetic manipulation [72]. This method confirms the thermodynamic parameters that underlie heterogeneous proton distribution.
Quantitative Data and Functional Regulation of ΔΨ Heterogeneity
The application of the above protocols has yielded consistent quantitative data on the nature and regulation of ΔΨm heterogeneity.
Table 2: Quantitative Effects of Perturbations on ΔΨC / ΔΨIBM Heterogeneity
| Experimental Condition | Effect on Cristae Structure | Effect on ΔΨ Heterogeneity | Key Findings |
|---|---|---|---|
| Baseline (Untreated) | Normal, well-defined cristae | ΔΨC > ΔΨIBM (Higher potential at cristae) | TMRE signal is strongest at cristae, confirming them as the primary sites for charge separation [78]. |
| Oligomycin (ATP synthase inhibitor) | Cristae structure intact | Heterogeneity Increased | ΔΨm increases globally, but the difference between cristae and IBM becomes more pronounced [78]. |
| FCCP (Uncoupler) | Cristae structure intact | Heterogeneity Abolished | Collapses the proton gradient and ΔΨm, equalizing the potential across the IMM [78]. |
| MICOS-KO (e.g., Mic13) | Cristae junctions disrupted; cristae morphology lost | Heterogeneity Diminished/Abolished | Loss of cristae structure leads to a more homogeneous ΔΨm, proving CJs are necessary for maintaining heterogeneity [78]. |
| OPA1 Impairment | Cristae junctions widened; cristae decompacted | Heterogeneity Diminished/Abolished | Demonstrates the role of OPA1 in maintaining cristae tightness and functional compartmentalization [78]. |
The data unequivocally show that under physiological conditions, the membrane potential is not uniform. The heterogeneity is dynamically regulated by metabolic demand (inhibiting ATP synthesis with oligomycin increases ΔΨ and its heterogeneity) and is absolutely dependent on the structural integrity of the cristae.
The Structural and Molecular Basis of Compartmentalization
The maintenance of ΔΨC > ΔΨIBM is not a passive phenomenon but is actively enforced by the precise architecture and protein composition of the IMM.
- Cristae Junctions as Diffusion Barriers: The narrow, slit-like CJs, stabilized by protein complexes like MICOS and OPA1, create a physical restriction that limits the free diffusion of protons and ions between the cristae lumen and the intermembrane space [78] [81]. This compartmentalization allows for the establishment of a distinct electrochemical environment within each crista.
- Protein Localization: The enrichment of electron transport chain complexes and ATP synthase dimers along the cristae membranes creates localized proton pumping and consumption [78] [82]. This spatial organization ensures that the proton circuit is primarily established within the cristae itself.
- Cristae as Independent Bioenergetic Units: A seminal finding is that individual cristae within a single mitochondrion can have disparate membrane potentials. Interventions causing acute depolarization can affect some cristae while sparing others [78]. This demonstrates that cristae function as independent bioenergetic units, preventing local damage or dysfunction from spreading to the entire network.
Implications for the Protonmotive Force and Future Research
The validation of ΔΨm heterogeneity fundamentally refines our understanding of the protonmotive force. The classical view of a uniform Δp across the entire IMM must be updated to a model where the dominant, bioenergetically relevant ΔΨ exists within the cristae compartments. This has several critical implications:
- Efficiency of Oxidative Phosphorylation: Sequestering the proton circuit within cristae minimizes the distance protons must diffuse between pumps (ETC) and turbines (ATP synthase), enhancing the kinetic efficiency of ATP production [78] [81].
- Metabolic Regulation and Signaling: The heterogeneity allows for localized changes in membrane potential and pH that can regulate specific processes, such as calcium uptake or reactive oxygen species (ROS) production, in a sub-mitochondrial manner [83].
- Apoptosis and Quality Control: During apoptosis, cristae remodeling is a key step in the release of cytochrome c. The disruption of cristae junctions and the consequent homogenization of ΔΨm is a committed step in the cell death pathway [2] [81].
- Drug Development: Understanding cristae-specific bioenergetics provides a new framework for developing therapeutic strategies. For instance, targeting the MICOS complex or OPA1 could modulate mitochondrial function in degenerative diseases or cancer by altering cristae integrity and, consequently, ΔΨ heterogeneity [84] [81].
Future research will need to further quantify the exact magnitude of the potential difference between ΔΨC and ΔΨIBM under various physiological and pathological states. Furthermore, integrating these findings with the dynamics of the pH gradient will provide a complete picture of the spatially resolved protonmotive force.
The paradigm of the inner mitochondrial membrane as a uniform, equipotential surface is obsolete. A convergence of super-resolution imaging, biophysical modeling, and genetic perturbation has conclusively validated the spatial heterogeneity of the mitochondrial membrane potential, with the cristae (ΔΨC) maintaining a higher potential than the inner boundary membrane (ΔΨIBM). This heterogeneity is actively maintained by the structural integrity of cristae junctions and is essential for efficient energy transduction, establishing cristae as independent bioenergetic units. This refined model not only deepens our fundamental understanding of cellular energetics but also creates new opportunities for diagnosing and treating a wide spectrum of mitochondrial diseases.
The Cristae Junction as a Regulatory Barrier for Ion and Potential Gradients
The inner mitochondrial membrane (IMM) is structurally and functionally compartmentalized into two distinct domains: the inner boundary membrane (IBM), which runs parallel to the outer membrane, and the cristae membranes (CM), which fold into the matrix to form cristae [85]. These two compartments are separated by the cristae junction (CJ), a narrow, circular opening with a diameter of 20–50 nm that serves as a crucial regulatory barrier [86]. This structural arrangement is not merely architectural; it is fundamental to the mitochondrion's bioenergetic function. The CJ acts as a gatekeeper, controlling the diffusion of ions, metabolites, and proteins between the cristae lumen and the intermembrane space, thereby maintaining the cristae as specialized bioenergetic compartments [86] [85].
The integrity and dynamics of the CJ are scaffolded by the mitochondrial contact site and cristae organizing system (MICOS) complex [86] [85]. A tight interaction between MICOS and the sorting and assembly machinery (SAM) complex creates a triangular structure that constricts the opposing sides of the IMM and the OMM [85]. Additionally, proteins like the dynamin-related GTPase OPA1 and the mitochondrial calcium uptake protein MICU1 are critical for CJ stability and regulation [86] [85]. The CJ's role as a physical barrier enables the establishment and maintenance of distinct electrochemical environments across the IMM, which is central to a broader thesis investigating the relationship between the mitochondrial membrane potential (MMP) and the proton concentration (pH) gradient that together constitute the protonmotive force (PMF) [3].
This whitepaper delves into the mechanisms by which the CJ regulates ion and potential gradients, explores advanced methodologies for its study, and discusses the implications of CJ dysfunction for drug development.
Structural and Molecular Determinants of the Cristae Junction
Core Protein Complexes and Lipid Composition
The CJ is not a passive structural feature but a dynamic constriction regulated by a specific set of proteins and lipids. The core scaffold is provided by the MICOS complex, an evolutionarily conserved protein complex in the IMM. In mammals, MICOS comprises at least seven subunits (MIC10, MIC13, MIC19, MIC25, MIC26, MIC27, and MIC60), which can be divided into two key sub-complexes: the MIC60 and MIC10 sub-complexes [86]. Deletion of most MICOS subunits, except MIC26, disrupts CJ integrity, leading to cristae detaching from the IBM and forming stacked membrane sheets within the matrix [86]. The F₁Fₒ-ATP synthase also plays a structural role beyond its catalytic function. The dimerization and oligomerization of this complex along the cristae ridges help shape the high membrane curvature, defining the cristae's morphology [86].
The lipid cardiolipin is enriched in the IMM and is crucial for stabilizing cristae architecture. It facilitates the dimerization of ATP synthase and supports the assembly and stability of respiratory supercomplexes, thereby influencing the local membrane environment essential for CJ function [86]. The table below summarizes the key molecular components governing CJ dynamics.
Table 1: Key Molecular Regulators of Cristae Junction Dynamics
| Molecule/Complex | Primary Function | Impact on CJ and Cristae |
|---|---|---|
| MICOS Complex | Forms the core scaffold of the CJ. | Maintains CJ integrity; deletion causes CJ loss and cristae stacking. |
| OPA1 | Dynamin-like GTPase; mediates IMM fusion. | Stabilizes and tightens CJs; maintains cristae structure. |
| F₁Fₒ-ATP Synthase | Catalyzes ATP synthesis; forms dimers/oligomers. | Induces and stabilizes cristae membrane curvature. |
| Cardiolipin | Phospholipid enriched in the IMM. | Stabilizes membrane architecture and protein supercomplexes. |
| MICU1 | Regulates mitochondrial calcium uptake. | Stabilizes CJ oligomers; Ca²⁺-dependent opening regulates permeability. |
Mechanisms of Cristae Remodeling
Cristae are highly dynamic, undergoing constant remodeling in response to cellular energy demands and stress signals. This process involves changes in cristae number, length, width, and the opening or closure of CJs [86]. Proteolytic processing of OPA1 is a key switch that controls cristae remodeling. In energy-rich conditions, long OPA1 forms promote tight CJs and fused mitochondria. During stress, OPA1 is cleaved, leading to CJ widening and facilitating the release of pro-apoptotic factors from the cristae [86]. Furthermore, the dimerization state of the F₁Fₒ-ATP synthase dictates cristae shape. Its dimeric form induces strong membrane curvature, and the dissociation of these dimers leads to cristae disassembly, directly linking metabolic output to ultrastructural organization [86].
The Cristae Junction as an Electrochemical Barrier
Regulation of Ion and Metabolite Flux
The CJ serves as a permeability barrier that separates the biochemical environments of the cristae lumen and the intermembrane space. This separation is critical for efficient oxidative phosphorylation. The cristae membrane houses approximately 94% of Complex III and ATP synthase and about 85% of cytochrome c, making it the primary site for electron transport and ATP synthesis [86]. By restricting the free diffusion of protons and other ions, the CJ helps maintain a concentrated pool of protons within the cristae lumen, which is essential for driving ATP synthase activity [85]. The barrier function also extends to metabolites and proteins, ensuring the distinct compartmentalization necessary for specialized metabolic functions.
Maintenance of Membrane Potential Gradients
Recent super-resolution microscopy studies have revealed that the CJ is not only a barrier for molecules but also for electrical potential. The membrane potential (ΔΨ) across the IMM is not uniform; the cristae membrane (ΔΨC) maintains a higher (more negative) membrane potential compared to the inner boundary membrane (ΔΨIBM) [85]. This potential gradient is physiologically significant. The CJ acts as an electrical seal, preventing the short-circuiting of the proton gradient by isolating the cristae, where the proton pumps are concentrated, from the IBM [85]. This compartmentalization ensures that the protonmotive force is most potent at the source of its generation, optimizing the efficiency of ATP production.
The regulation of this gradient is dynamic. Mitochondrial calcium (Ca²⁺) elevation, for instance, leads to a preferential hyperpolarization of the cristae membrane [85]. This is because Ca²⁺ activates dehydrogenases in the tricarboxylic acid (TCA) cycle, boosting electron donation to the respiratory chain complexes located in the cristae, thereby increasing proton pumping and enhancing ΔΨC [85]. The protein MICU1 plays a direct role in coupling Ca²⁺ signaling to CJ permeability. At high cytosolic Ca²⁺ concentrations, MICU1 oligomers disassemble, leading to the opening of the CJ and allowing Ca²⁺ to pass into the matrix, which subsequently stimulates metabolism [85]. This mechanism illustrates a sophisticated feedback loop where metabolic demand directly influences the ultrastructural and electrochemical properties of the mitochondrion.
Table 2: Experimentally Measured Gradients Across the Cristae Junction
| Parameter | Cristae Membrane (CM) | Inner Boundary Membrane (IBM) | Measurement Technique |
|---|---|---|---|
| Membrane Potential (ΔΨ) | Higher (more negative) [85] | Lower (less negative) [85] | STED/SIM with TMRM |
| pH | More alkaline (part of PMF) [3] | More acidic (closer to cytosol) Inferred | N/A |
| Ca²⁺ Influx | Primary entry point, regulated by MICU1 [85] | Secondary route Inferred | Super-resolution microscopy |
| Protein Composition | Enriched with ETC complexes & ATP synthase [86] | Contains protein import machinery (e.g., TIM) [82] | Proteomics, EM |
Methodologies for Investigating CJ Barrier Function
Super-Resolution Microscopy of Membrane Potential
Conventional fluorescence microscopy cannot resolve the nanoscale dynamics of the CJ. Stimulated Emission Depletion (STED) and Structured Illumination Microscopy (SIM) have emerged as key tools for visualizing the spatial membrane potential gradients (SMPG) across the mitochondrial cross-section [85]. A standard protocol involves dual-channel imaging with two fluorescent dyes:
- Tetramethylrhodamine Methyl Ester (TMRM): A potentiometric dye whose distribution and intensity are sensitive to the membrane potential. At low concentrations (1.35-5.4 nM), TMRM preferentially accumulates in the hyperpolarized cristae.
- MitoTracker Green FM (MTG): A dye that accumulates in the IMM independent of membrane potential after binding, serving as a structural reference for mitochondrial morphology [85].
The workflow involves treating cells (e.g., HeLa or EA.hy926) with 500 nM MTG and a low concentration of TMRM (e.g., 13.5 nM). After simultaneous dual-channel imaging, two analytical methods can quantify the SMPG:
- IBM Association Index: An automated method that uses the MTG channel to define mitochondrial boundaries. The fluorescence intensity of TMRM in the IBM region is compared to that in the CM region (IBM/CM ratio). A decrease in this index after stimulation indicates cristae hyperpolarization [85].
- ΔFWHM (Full Width at Half Maximum) Method: This semi-automated method analyzes cross-section intensity profiles of MTG and TMRM. A larger difference in the FWHM of the two signals indicates greater TMRM accumulation in the cristae [85].
The following diagram illustrates this experimental pipeline for measuring membrane potential gradients.
Integrative Computational Modeling
Given the complexity of the CJ, computational approaches are invaluable. Integrative modeling combines experimental data from cryo-electron tomography, proteomics, and lipidomics to construct realistic computational models of cristae ultrastructure [82]. One such approach uses coarse-grained molecular dynamics (MD) simulations to model a human crista junction, incorporating key proteins like MICOS, ATP synthase dimers, and a lipid bilayer enriched with cardiolipin [82]. These models serve as "living" systems that can be updated with new data, allowing researchers to test hypotheses about protein-lipid interactions, membrane curvature, and the dynamics of metabolite flow through the CJ in silico.
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents and Tools for Cristae Junction Research
| Reagent/Tool | Function/Application | Key Consideration |
|---|---|---|
| TMRM | Potentiometric dye for measuring spatial membrane potential gradients. | Use low concentrations (1.35-5.4 nM) to resolve CM/IBM differences [85]. |
| MitoTracker Green FM (MTG) | IMM structural marker for normalization in SMPG assays. | Binds covalently; distribution becomes potential-independent after accumulation [85]. |
| MICOS-Specific Antibodies | Detect and localize MICOS subunits (e.g., MIC60, MIC19) via immuno-EM/IF. | Validates CJ integrity and protein complex assembly. |
| OPA1 Proteolysis Inhibitors | Chemically inhibit OPA1 cleavage to study CJ dynamics in apoptosis. | Tools to dissect the role of L-OPA1 vs. S-OPA1 isoforms. |
| Rotenone & Antimycin A | Inhibitors of ETC Complex I and III. | Used to confirm proton pump activity's role in potential gradients [85]. |
| CRISPR/Cas9 KO Cells | Generate knockouts of specific CJ proteins (e.g., MICU1, MICOS subunits). | Essential for establishing protein function in CJ gating and Ca²⁺ handling. |
| Coarse-Grained MD Model | Computational model of cristae ultrastructure for in silico experiments. | Allows simulation of protein-lipid interactions and CJ permeability [82]. |
Pathophysiological Implications and Drug Discovery
Dysregulation of CJ integrity and function is a emerging factor in human disease. Aberrant cristae morphology compromises electron transport, ATP production, and mitochondrial metabolism, contributing to pathologies such as neurodegeneration and metabolic diseases [86]. For example, genetic variants in cristae-shaping proteins like OPA1 are linked to dominant optic atrophy, while MICOS mutations are associated with severe neurological disorders [86]. In cancer, mitochondrial cristae remodeling can enhance the reductive biosynthesis of molecular precursors, supporting rapid cellular proliferation and presenting a novel target for therapeutic intervention [3].
The CJ itself represents a potential novel therapeutic target. Strategies could aim to stabilize CJ integrity in degenerative conditions or selectively disrupt it in cancer cells. The discovery that an optogenetic system inducing inter-mitochondrial contacts can enhance MMP and alleviate blue light-induced damage in retinal cells hints at future bioengineering approaches for treating mitochondrial dysfunction [87]. Furthermore, the Ca²⁺-dependent regulation of the CJ via MICU1 offers a specific node for pharmacological modulation to fine-tune mitochondrial bioenergetics in disease contexts [85]. The relationship between CJ stability, membrane potential gradients, and downstream processes like mitochondrial fission and apoptosis provides a rich landscape for developing targeted therapies.
The cristae junction is far more than a static structural feature of the inner mitochondrial membrane. It is a dynamic, regulated barrier that is fundamental to establishing and maintaining the ion and potential gradients necessary for efficient oxidative phosphorylation. By compartmentalizing the IMM, the CJ allows for the spatial organization of bioenergetic and signaling processes, creating a specialized environment that optimizes ATP production and integrates cellular signals. Advanced techniques like super-resolution microscopy and integrative computational modeling are unveiling the complex regulation of this nanoscale apparatus. Understanding the CJ's role in both health and disease opens promising new avenues for targeting mitochondrial function in a range of human pathologies, firmly positioning it as a critical component in the relationship between membrane potential and cellular energy metabolism.
The proton motive force (PMF), an electrochemical gradient across the inner mitochondrial membrane, serves as a fundamental bioenergetic source for ATP production and cellular signaling. Comprising a membrane potential (ΔΨm) component and a pH gradient (ΔpH), the PMF's composition and magnitude are precisely tuned to meet the specialized physiological demands of different cell types. This review synthesizes current research on PMF adaptations in three distinct models: neurons, which utilize dynamic PMF changes to support synaptic plasticity and mitochondrial quality control; cardiomyocytes, which require a robust, stable PMF to sustain continuous contraction; and cancer cells, which often maintain a characteristically hyperpolarized ΔΨm to fuel proliferation, invasion, and metastasis. We provide quantitative comparisons, detailed experimental protocols for PMF assessment, and visualization of key regulatory pathways. Understanding these cell-specific adaptations offers crucial insights for developing therapies targeting metabolic disorders and cancer.
The proton motive force (PMF) is the electrochemical potential difference generated by the electron transport chain (ETC) across the inner mitochondrial membrane. It is the central energetic intermediate that couples nutrient oxidation to ATP synthesis and other energy-dependent processes. The PMF is expressed in millivolts (mV) and is described by the following equation:
Δp = ΔΨm - ZΔpH
Where Δp is the total PMF, ΔΨm is the mitochondrial membrane potential (electrical gradient, typically -150 to -180 mV), Z is a constant (~59 at 25°C), and ΔpH is the chemical gradient of protons (typically ~0.4 pH units, matrix alkaline) [3] [72]. Under most physiological conditions, the ΔΨm constitutes the dominant component, contributing approximately 75% of the total PMF, while the ΔpH contributes the remaining 25% [3]. The precise balance between these components is not static; it varies according to cell type, energy demand, and substrate availability, reflecting specialized cellular physiology.
The PMF drives several critical functions beyond ATP synthesis, including ion transport (e.g., calcium), reactive oxygen species (ROS) production, protein import into the matrix, and mitochondrial quality control via mitophagy [3] [88]. This review explores how neurons, cardiomyocytes, and cancer cells differentially harness and regulate these PMF-dependent processes to support their unique physiological and pathophysiological roles.
Cell-Type Specific PMF Adaptations
Neuronal PMF: Dynamic Signaling for Plasticity and Quality Control
Neurons exhibit a highly dynamic PMF that functions as a metabolic signaling hub, coordinating complex processes like synaptic plasticity, dendritic remodeling, and mitochondrial quality control. Unlike cells with a singular metabolic focus, neurons compartmentalize bioenergetics, maintaining distinct mitochondrial subpopulations to meet localized energy demands. For instance, mitochondria positioned at synapses exhibit different functional characteristics compared to those in the soma [3].
A key neuronal adaptation is the use of localized ΔΨm changes to integrate metabolic state with structural and functional plasticity. Transient, localized ΔΨm reductions in dendrites can signal the need for protein synthesis and synaptic remodeling, linking metabolic status directly to neuronal connectivity [3]. Furthermore, neurons maintain a tight coupling between PMF and mitochondrial quality control. A sustained loss of ΔΨm in a mitochondrial fragment serves as the primary signal for its removal via PINK1/Parkin-mediated mitophagy, preventing the accumulation of dysfunctional organelles that could compromise neuronal viability [3].
Recent groundbreaking research has revealed another dimension of neuronal PMF adaptability: intercellular mitochondrial transfer. Neurons within the tumor microenvironment can undergo metabolic reprogramming, increasing their mitochondrial mass and subsequently transferring mitochondria to adjacent breast cancer cells. This transfer enhances the metabolic capacity and metastatic potential of the recipient cancer cells, illustrating a pathological manipulation of neuronal bioenergetic resources [89].
Table 1: Key Characteristics of Neuronal PMF
| Feature | Adaptation in Neurons | Functional Significance |
|---|---|---|
| PMF Dynamics | Highly dynamic, spatially heterogeneous | Supports synaptic plasticity, dendritic remodeling [3] |
| Mitochondrial Subpopulations | Metabolically specialized (e.g., synaptic vs. somatic) | Meets localized energy and signaling demands [3] |
| ΔΨm in Signaling | Regulates local protein synthesis, cytoskeletal dynamics | Links metabolic state to structural and functional plasticity [3] |
| Quality Control | ΔΨm loss triggers PINK1/Parkin mitophagy | Maintains a healthy mitochondrial network [3] |
| Pathological Role | Donates mitochondria to cancer cells | Enhances cancer metabolic plasticity and metastasis [89] |
Cardiomyocyte PMF: Stability for Continuous Contraction
Cardiac muscle cells are endurance athletes, requiring a constant and massive supply of ATP to sustain rhythmic contractions throughout an organism's lifetime. To meet this demand, cardiomyocytes have evolved a bioenergetic system centered on a highly stable and robust PMF, primarily generated by fatty acid β-oxidation and the TCA cycle.
A critical adaptation in cardiomyocytes is the presence of spatially distinct mitochondrial subpopulations. Subsarcolemmal mitochondria are located beneath the plasma membrane, while interfibrillar mitochondria are densely packed between the contractile myofibrils [3]. These subpopulations exhibit differences in respiratory capacity, protein composition, and sensitivity to metabolic stress, allowing for optimized energy distribution throughout the cell [3]. The interfibrillar mitochondria, in particular, are positioned to efficiently channel ATP directly to the myofibrils, minimizing diffusion distances and ensuring rapid energy delivery for contraction.
The architecture of the inner mitochondrial membrane is also crucial. The cristae membrane, which houses the ETC complexes, is often hyperpolarized relative to the inner boundary membrane. This compartmentalized ΔΨm creates a dedicated, high-capacity environment for OXPHOS. Research shows that calcium entry into the mitochondrial matrix, as occurs during excitation-contraction coupling, can further hyperpolarize the cristae membrane. This is likely driven by Ca2+-sensitive stimulation of TCA cycle dehydrogenases, which increases electron donation to the ETC and subsequent proton pumping [22]. This mechanism allows the PMF to be dynamically and precisely tuned to match sudden changes in cardiac workload.
Table 2: Key Characteristics of Cardiomyocyte PMF
| Feature | Adaptation in Cardiomyocytes | Functional Significance |
|---|---|---|
| PMF Stability | High, stable ΔΨm to support constant ATP demand | Fuels continuous contraction and relaxation cycles |
| Mitochondrial Subpopulations | Subsarcolemmal vs. interfibrillar mitochondria | Optimizes spatial energy distribution [3] |
| Cristae ΔΨm | Hyperpolarized relative to inner boundary membrane | Creates efficient, high-capacity OXPHOS compartment [22] |
| Calcium Coupling | Ca2+ influx hyperpolarizes cristae membrane (ΔΨC) | Boosts ATP production in response to increased workload [22] |
| Metabolic Fuel | Primarily fatty acid β-oxidation | High-yield ATP production for sustained effort |
Cancer Cell PMF: Hyperpolarization for Proliferation and Metastasis
A hallmark of many epithelial cancers is an abnormally high ΔΨm (hyperpolarization) compared to their normal counterparts [88]. This bioelectrical alteration is not merely an epiphenomenon but a functional driver of tumorigenesis, supporting several malignant behaviors.
The hyperpolarized ΔΨm in cancer cells is linked to increased invasive potential in vitro and enhanced metastatic burden in vivo. Mouse models of metastatic breast cancer show that cancer cells with a higher ΔΨm form significantly more lung metastases than those with a lower ΔΨm [88]. This hyperpolarization creates a favorable environment for ROS production, as the relationship between ΔΨm and ROS is exponential; beyond a specific threshold, small increases in ΔΨm lead to dramatic surges in mitochondrial ROS, which can activate pro-invasive signaling pathways [88].
Cancer cells also exhibit remarkable metabolic plasticity, often relying on aerobic glycolysis (the Warburg effect) while simultaneously maintaining a high ΔΨm to support other functions. This ΔΨm is sustained through various mechanisms, including oncogene-driven metabolic reprogramming and interactions with the tumor microenvironment. A striking non-cell-autonomous mechanism is the transfer of mitochondria from neurons to cancer cells. In breast cancer, neurons donate mitochondria to cancer cells, a process that enhances the cancer cells' oxidative phosphorylation capacity, spare respiratory capacity, and ultimately, their metastatic fitness [89]. This highlights the PMF as a central player in the intercellular crosstalk that fuels cancer progression.
Table 3: Key Characteristics of Cancer Cell PMF
| Feature | Adaptation in Cancer Cells | Functional Significance |
|---|---|---|
| Basal ΔΨm | Abnormally hyperpolarized | Associated with invasiveness and metastatic potential [88] |
| ROS Signaling | High ΔΨm promotes reactive oxygen species (ROS) production | Activates pro-invasive and pro-proliferative pathways [88] |
| Metabolic Plasticity | High ΔΨm coexists with aerobic glycolysis (Warburg Effect) | Supports anabolic processes and redox balance [90] |
| Mitochondrial Transfer | Receive functional mitochondria from neurons | Enhances OXPHOS, metabolic fitness, and metastasis [89] |
| Therapeutic Target | Mitochondrial uncouplers show anti-cancer effects | Depletes ATP, disrupts metabolic signaling [91] |
Experimental Protocols for PMF Analysis
Absolute Quantification of PMF Components via Multi-Wavelength Spectroscopy
This protocol allows for the absolute quantification of both ΔΨm and ΔpH without genetic manipulation or exogenous dyes, by analyzing the redox poise of the hemes in the mitochondrial bc1 complex [72].
Workflow Overview:
- Cell Preparation: Culture cells (e.g., RAW 264.7 mouse macrophage cells) in spinner flasks and resuspend at high density (e.g., 2.0 × 10^7 cells/mL) in a sealed, temperature-controlled (37°C) quartz chamber with integrated oxygen measurement.
- Spectral Acquisition: Use multi-wavelength cell spectroscopy to measure attenuation spectra of the hemes (bH, bL, and cytochrome c) under baseline conditions and during titrations with modulators like oligomycin (ATP synthase inhibitor) and CCCP (proton ionophore).
- Redox State Calculation: Apply a linear-combination-of-model-spectra algorithm to the differential attenuation spectra to calculate the oxidation state of each heme. Fully reduce and oxidize hemes for absolute quantification.
- PMF Calculation: Utilize a stochastic model of bc1 complex turnover. The redox potentials of the b-hemes (EhbH and EhbL), which are separated in the dielectric field of the membrane, are used to calculate ΔΨm based on the equilibrium equation: ΔΨm = (EhbH - EhbL)/β, where β is the fractional dielectric distance. The ΔpH is then derived from the difference in redox potentials of cytochrome c and the ubiquinone pool [72].
Super-Resolution Analysis of Spatial Membrane Potential Gradients (SMPG)
This method employs super-resolution microscopy to visualize differential ΔΨm across mitochondrial sub-compartments, such as the cristae membrane (CM) and inner boundary membrane (IBM) [22].
Workflow Overview:
- Cell Staining: Co-stain live cells (e.g., HeLa or EA.hy926) with two fluorescent dyes: Tetramethylrhodamine Methyl Ester (TMRM, 1.35-81 nM), a ΔΨm-sensitive dye, and MitoTracker Green FM (MTG, 500 nM), a ΔΨm-insensitive dye that labels the inner mitochondrial membrane.
- Super-Resolution Imaging: Perform simultaneous dual-channel imaging using Structured Illumination Microscopy (SIM) to achieve resolution beyond the diffraction limit.
- Image Analysis: Two analytical methods can be applied:
- IBM Association Index: An automated threshold of the MTG channel defines mitochondrial boundaries. The image is processed to create IBM and CM regions, and the fluorescence intensity ratio (TMRM-IBM/TMRM-CM) is calculated [22].
- ΔFWHM (Full Width at Half Maximum): Cross-section intensity profiles of MTG and TMRM are plotted for individual mitochondria. The difference in the FWHM of the two profiles indicates the degree of TMRM accumulation in the cristae [22].
- Physiological Stimulation: Treat cells with agents like histamine to induce mitochondrial calcium uptake. A subsequent decrease in the IBM association index or ΔFWHM indicates relative hyperpolarization of the cristae membrane, linking TCA cycle activation to localized PMF changes [22].
Diagram 1: Experimental workflow for super-resolution analysis of spatial membrane potential gradients (SMPG).
The Scientist's Toolkit: Key Research Reagents
Table 4: Essential Reagents for PMF and Metabolism Research
| Reagent / Tool | Function / Target | Key Application in PMF Research |
|---|---|---|
| TMRM / TMRE | Potentiometric, cationic fluorescent dye | Accumulates in mitochondrial matrix in a ΔΨm-dependent manner; used to measure relative ΔΨm [22]. |
| MitoTracker Green FM (MTG) | Covalent thiol-reactive stain for mitochondria | Labels mitochondrial architecture independently of ΔΨm; used as a spatial reference in SMPG assays [22]. |
| Carbonyl cyanide m-chlorophenyl hydrazone (CCCP) | Protonophore (mitochondrial uncoupler) | Dissipates the PMF by shuttling protons across the inner membrane; used as a positive control for depolarization [91] [72]. |
| Oligomycin | ATP synthase (Complex V) inhibitor | Blocks proton flow through ATP synthase, causing a transient increase in ΔΨm; used in mitochondrial stress tests [72] [88]. |
| Rotenone & Antimycin A | ETC Complex I & III inhibitors | Inhibit electron flow and PMF generation; used to confirm ETC-dependence of observed effects [22]. |
| MitoTRACER | Genetic reporter for mitochondrial transfer | Permanently labels recipient cells that have acquired mitochondria from donor cells; used to trace intercellular mitochondrial fate [89]. |
Signaling and Regulatory Pathways
The regulation of PMF involves a complex interplay of intracellular signaling, ion transport, and inter-organelle communication. The following diagram and summary outline the core pathways that maintain and modulate the PMF across different cell types.
Diagram 2: Core signaling and regulatory pathways of the mitochondrial proton motive force (PMF).
Pathway Description: The primary pathway for PMF generation begins with nutrient oxidation in the TCA cycle, producing reduced electron carriers (NADH, FADH2). The Electron Transport Chain (ETC) uses these carriers to pump protons from the matrix into the intermembrane space, directly building the PMF [88]. The PMF is primarily consumed by ATP synthase (Complex V) to phosphorylate ADP. However, protons can also leak back into the matrix without producing ATP, a process mediated by uncoupling proteins (UCPs) and the adenine nucleotide translocase (ANT) [91]. This "proton leak" acts as a safety valve to prevent excessive hyperpolarization and ROS production.
The PMF also drives secondary functions. It is the electrophoretic force for calcium uptake into the matrix, which in turn can stimulate the TCA cycle, creating a feed-forward loop to boost PMF and ATP production [22] [88]. The level of ΔΨm is a critical signaling node: a hyperpolarized state exponentially increases ROS production, which can activate signaling pathways but also contribute to oxidative damage. Conversely, a sustained loss of ΔΨm is a hallmark of mitochondrial dysfunction, serving as the trigger for PINK1/Parkin-mediated mitophagy and the initiation of apoptosis [3] [88]. In cancer cells, oncogenic signaling pathways can modulate this entire network to maintain a pro-survival, hyperpolarized state.
The proton motive force is a universal bioenergetic currency, yet its regulation and utilization are exquisitely specialized across cell types. Neurons leverage PMF dynamics for signaling and plasticity, cardiomyocytes optimize it for stability and endurance, and cancer cells co-opt it for proliferation and survival. The experimental frameworks and reagents detailed herein provide a roadmap for probing these adaptations. Future research dissecting the molecular regulators of these differential PMF phenotypes holds significant promise, particularly for developing novel therapeutic strategies that target the metabolic vulnerabilities of cancer cells while sparing normal neuronal and cardiac function.
The proton motive force (PMF), an electrochemical gradient across the mitochondrial inner membrane, serves as a fundamental energetic currency for cellular processes, coupling electron transport chain activity to ATP synthesis. This whitepaper provides a technical guide for integrating dynamic PMF measurements with transcriptomic and metabolomic profiling. We detail methodologies for dissecting PMF components—mitochondrial membrane potential (ΔΨm) and pH gradient (ΔpH)—and present computational frameworks for their correlation with molecular omics data. Within the broader context of mitochondrial membrane potential and pH gradient research, this integrated approach reveals novel regulatory mechanisms in bioenergetics, stress adaptation, and disease pathogenesis, offering drug development professionals a comprehensive toolkit for identifying therapeutic targets in metabolic and neurodegenerative disorders.
The proton motive force (PMF) is the electrochemical potential energy stored across the mitochondrial inner membrane, generated through electron transport chain (ETC) activity that pumps protons from the matrix to the intermembrane space [11]. The PMF comprises two primary components: a charge gradient (ΔΨm, typically -180 mV) and a chemical gradient (ΔpH, approximately 0.4 pH units) [3]. Under physiological conditions, ΔΨm constitutes the dominant force (approximately 75-80% of total PMF), equivalent to a 1000-fold difference in proton concentration, while ΔpH contributes the remaining 20-25% [3] [11]. This energy reservoir primarily drives ATP synthesis through F1Fo-ATP synthase but also functions as a dynamic signaling hub that regulates reactive oxygen species (ROS) production, calcium handling, and mitochondrial quality control [3] [92].
Recent advances demonstrate that PMF is not merely a static bioenergetic reservoir but exhibits dynamic, compartmentalized fluctuations that influence cellular signaling and metabolic specialization [3] [92]. The integration of PMF dynamics with transcriptomic and metabolomic profiles enables researchers to connect organelle-level bioenergetics with system-wide molecular changes, providing unprecedented insights into cellular physiology and disease mechanisms. This approach is particularly valuable for understanding complex processes such as synaptic plasticity in neurons, where mitochondrial recruitment to dendrites links energy production with localized protein synthesis [3], and in pathological conditions including neurodegeneration, ischemic injury, and cancer [3] [11].
Technical Foundations: Measuring PMF Components
Dissecting ΔΨm and ΔpH Contributions
Accurate measurement of individual PMF components is essential for correlating these parameters with omics data. The following table summarizes core measurement techniques and their applications:
Table 1: Experimental Approaches for PMF Component Measurement
| Parameter | Measurement Technique | Key Reagents/Probes | Experimental Considerations |
|---|---|---|---|
| ΔΨm | Fluorometry with potentiometric dyes | TMRM, TMRE, Safranin | Dye concentration critical; validate with uncouplers (FCCP) [93] [19] |
| ΔΨm | Flow cytometry with TMRM | TMRM | Enables single-organelle analysis in permeabilized cells [93] |
| Matrix pH | Fluorometry with pH-sensitive probes | BCECF-AM | Calibrate with ionophores in high-K+ buffers [19] [94] |
| Overall PMF | Respiration analysis with substrate/inhibitor titrations | Octyl guanidine, Nigericin, Valinomycin | Use in isolated mitochondria or permeabilized cells [93] [19] |
Experimental Models for PMF Assessment
Isolated Mitochondria Systems provide precise control over experimental conditions. Protocol: isolate mitochondria via differential centrifugation (e.g., from heart or brain tissue); suspend in defined respiratory buffers; measure parameters after adding specific substrates (e.g., malate/glutamate for Complex I, succinate for Complex II) [19] [94]. Permeabilized Cell Systems (e.g., PMF-seq): gently permeabilize plasma membrane with perfringolysin O while preserving mitochondrial integrity; enables introduction of non-cell-permeable substrates/inhibitors; compatible with high-throughput screening [93].
Ionophore manipulations allow experimental dissection of PMF components: Nigericin (K+/H+ exchanger) decreases ΔpH while increasing ΔΨm; Valinomycin (K+ ionophore) decreases ΔΨm while increasing ΔpH and matrix pH [19]. These tools enable researchers to parse specific contributions of each PMF component to downstream molecular responses.
Multi-Omics Integration Strategies and Workflows
Computational Frameworks for Data Integration
Table 2: Multi-Omics Integration Approaches for Correlating with PMF Dynamics
| Integration Approach | Core Methodology | Application to PMF Research | Key Tools/Platforms |
|---|---|---|---|
| Correlation-based Integration | Pearson/Spearman correlation between PMF parameters and omics features | Identify genes/metabolites associated with ΔΨm or ΔpH changes | Cytoscape, igraph [95] |
| Gene Co-expression with Metabolite Integration | Weighted Correlation Network Analysis (WGCNA) | Module eigengenes correlated with metabolite patterns and PMF states | WGCNA R package [95] |
| Pathway Enrichment Analysis | Overrepresentation analysis in KEGG, Reactome | Link transcriptomic/metabolomic changes to PMF-affected pathways | Joint-Pathway Analysis, Ingenuity Pathway Analysis [96] [97] |
| Machine Learning Integration | Regularized regression, Random Forests | Predict PMF states from multi-omics features; identify key regulators | STITCH, MixOmics [95] |
Integrated Workflow for PMF-Omics Studies
The following diagram illustrates a comprehensive workflow for integrating PMF dynamics with multi-omics data:
PMF-Driven Signaling Pathways and Molecular Regulation
Key Pathways Linking PMF to Cellular Responses
The diagram below illustrates major signaling pathways through which PMF dynamics influence cellular processes and regulate molecular profiles:
PMF-Regulated Molecular Networks
Research demonstrates that PMF dynamics directly influence specific molecular networks detectable through multi-omics approaches:
Mitochondrial Quality Control: Reduced ΔΨm triggers PINK1 accumulation, recruiting Parkin and initiating mitophagy—a process observable through increased expression of autophagy-related genes (ATG3, ATG12) and metabolic shifts in energy metabolism [3] [97].
Metabolic Specialization: Elevated ΔΨm enhances pyrroline-5-carboxylate synthase (P5CS) activity and filament formation, promoting proline biosynthesis and reductive metabolism, while low ΔΨm favors oxidative phosphorylation [3]. This specialization creates distinct mitochondrial subpopulations with different metabolic roles.
Immune and Stress Response: Radiation exposure studies show PMF alterations correlate with differential expression of immune response genes (H2-Ab1, Cd40, Cd28) and metabolic enzymes (Aadac, Abat, Hmox1), linking bioenergetic changes to transcriptional regulation of inflammation and stress adaptation [96].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for PMF-Omics Studies
| Reagent Category | Specific Examples | Function/Application | Considerations |
|---|---|---|---|
| PMF Measurement Dyes | TMRM, TMRE, Safranin O | ΔΨm detection via fluorometry | Concentration-dependent response; potential toxicity with prolonged use [93] [19] |
| pH-Sensitive Probes | BCECF-AM, SNARF | Matrix pH measurement | Require careful calibration; BCECF excitation ratio (440/490 nm) pH-dependent [19] |
| Ionophores | Nigericin, Valinomycin | Dissect ΔΨm vs. ΔpH contributions | Nigericin: K+/H+ exchanger; Valinomycin: K+ ionophore [19] |
| ETC Substrates/Inhibitors | Glutamate/Malate, Succinate, Piericidin, Antimycin A | Specific respiratory chain modulation | Glutamate/Malate: Complex I; Succinate/Piericidin: Complex II [93] |
| Permeabilization Agents | Perfringolysin O | Plasma membrane permeabilization | Preserves mitochondrial integrity; enables substrate access [93] |
| CRISPR Screening Tools | MitoPlus library (15,271 sgRNAs) | Genetic screening of mitochondrial genes | Enables connection of genes to bioenergetic phenotypes [93] |
Case Study: Integrated Analysis of Radiation Response
A comprehensive multi-omics study on total-body irradiated mice demonstrates the power of integrating PMF-related measurements with transcriptomic and metabolomic profiling [96]. Researchers exposed mice to 1 Gy (low dose) and 7.5 Gy (high dose) radiation, collecting blood samples at 24 hours post-exposure for RNA sequencing and LC-MS-based metabolomics/lipidomics.
Methodology and Workflow
- Transcriptomic Profiling: RNA sequencing identified 2,837 differentially expressed genes (1,595 upregulated, 1,242 downregulated) in the high-dose group versus 143 in the low-dose group
- Metabolomic Analysis: LC-MS revealed dysregulated amino acids, phospholipids (PC, PE), and carnitine metabolites
- Data Integration: Joint-Pathway Analysis and STITCH interaction networks connected these changes to altered amino acid, carbohydrate, lipid, nucleotide, and fatty acid metabolism
- Bioinformatic Validation: Gene Ontology analysis confirmed enrichment in immune response pathways; BioPAN predicted key lipid pathway enzymes (Elovl5, Elovl6, Fads2)
Key Findings and Implications
This integrated approach revealed how radiation-induced PMF alterations correlate with specific molecular signatures: immune response genes (H2-Ab1, Cd40, Cd28) were significantly upregulated, while metabolic enzymes (Aadac, Abat, Aldh1a2) showed coordinated expression changes with associated metabolic shifts [96]. The study established a proof-of-concept for identifying bioenergetic stress responses through correlated multi-omics signatures, providing a template for similar investigations in other disease models.
Integrating PMF dynamics with transcriptomic and metabolomic profiles represents a powerful approach for elucidating the molecular mechanisms underlying bioenergetic regulation in health and disease. The methodologies and frameworks outlined in this technical guide provide researchers with comprehensive tools for designing and executing integrated studies that connect organelle-level bioenergetics with system-wide molecular responses.
Future directions in this field will likely include the development of more sophisticated real-time PMF monitoring in live cells alongside single-cell multi-omics approaches, enabling unprecedented resolution in understanding cellular heterogeneity in bioenergetic states. Additionally, advanced machine learning algorithms applied to integrated PMF-omics datasets promise to identify novel regulatory networks and therapeutic targets for conditions ranging from neurodegenerative diseases to cancer metabolism.
As these technologies mature, the correlation between PMF dynamics and molecular profiles will increasingly inform drug development strategies, enabling professionals to target specific bioenergetic pathways with greater precision and therapeutic efficacy.
The protonmotive force (PMF), an electrochemical gradient across the inner mitochondrial membrane (IMM), serves as a fundamental energy converter, coupling nutrient oxidation to cellular energy production. The PMF consists of two primary components: an electrical gradient, the mitochondrial membrane potential (ΔΨmito), and a chemical pH gradient (ΔpH) [3]. Under most physiological conditions, the ΔΨmito constitutes the dominant part of the PMF, typically around -180 mV, which is equivalent to a 1000-fold difference in proton concentration across the membrane [3]. This potential drives not only ATP synthesis but also regulates critical processes including reactive oxygen species (ROS) production, calcium handling, metabolic specialization, and protein import [3]. Dysregulation of PMF dynamics represents a converging pathological mechanism in diverse diseases, from hematological malignancies to respiratory disorders, creating novel therapeutic opportunities. This review examines how PMF dysregulation contributes to disease pathogenesis and informs the development of targeted pharmacological interventions, with particular emphasis on primary myelofibrosis (PMF) and related disorders where mitochondrial dysfunction and inflammatory signaling create self-reinforcing pathological cycles.
Table 1: Components of the Protonmotive Force (PMF)
| Component | Description | Typical Magnitude | Primary Functions |
|---|---|---|---|
| Mitochondrial Membrane Potential (ΔΨmito) | Electrical gradient due to charge separation across IMM | ≈ -180 mV | Primary driver of ATP synthesis; regulates protein import, metabolite transport, and ROS signaling |
| pH Gradient (ΔpH) | Chemical gradient from differences in proton concentration | ΔpH ≈ 0.4 units (matrix alkaline) | Contributes to PMF; regulates enzymatic activities and metabolite transport |
| Total PMF | Combined electrochemical potential | ≈ -200 to -220 mV | Converted to ATP by F₁F₀-ATP synthase or released as heat via uncoupling proteins |
Molecular Basis of PMF Generation and Dysregulation
The electron transport chain (ETC) complexes I, III, and IV generate PMF by pumping protons from the mitochondrial matrix to the intermembrane space during electron transfer to molecular oxygen [3]. This established proton gradient is then utilized by F₁F₀-ATP synthase to phosphorylate ADP, coupling oxidative phosphorylation to cellular energy demands. Recent super-resolution microscopy studies have revealed that the PMF is not uniform across the IMM but exhibits compartmentalization, with the cristae membrane (CM) demonstrating a more negative potential (ΔΨC) compared to the inner boundary membrane (ΔΨIBM) [22]. This spatial heterogeneity is maintained by the cristae junction (CJ), which acts as a diffusion barrier, and is regulated by proteins including MICU1 and OPA1 [22].
Functional interactions between key IMM proteins fine-tune PMF utilization. Real-time imaging of mitochondrial matrix pH has demonstrated that the ADP/ATP carrier (AAC) and ATP synthase coordinate to regulate H+ fluxes [8]. Activation of AAC-dependent H+ transport induces matrix acidification followed by a re-alkalization phase mediated by reverse activity of ATP synthase, revealing a feedback mechanism for maintaining PMF homeostasis [8]. Disruption of this delicate balance underlies pathological PMF dysregulation, which can manifest as either excessive PMF generation leading to oxidative stress or collapsed PMF resulting in bioenergetic failure.
Table 2: Key Proteins Regulating PMF Homeostasis
| Protein/Complex | Localization | Function in PMF Regulation | Pathological Implications |
|---|---|---|---|
| ETC Complexes I, III, IV | Cristae Membrane | Generate PMF through proton pumping | Mutations cause OXPHOS deficiencies; overactivity increases ROS |
| F₁F₀-ATP Synthase | Cristae Membrane | Consumes PMF for ATP synthesis; can reverse under acidification | Dysfunction disrupts energy production; implicated in neurodegeneration |
| ADP/ATP Carrier (AAC) | Inner Membrane | Exchanges ADP for ATP; mediates H+ transport | Mutations affect energy distribution between ATP and heat |
| Uncoupling Proteins (UCPs) | Inner Membrane | Dissipate PMF as heat, reducing ROS production | UCP polymorphisms linked to obesity, neurodegeneration |
| Cristae Junction Proteins (MICU1, OPA1) | Cristae Junction | Regulate compartmentalization of ΔΨmito | Abnormalities disrupt PMF gradients and calcium signaling |
PMF Dysregulation in Primary Myelofibrosis: Mechanisms and Consequences
Primary myelofibrosis (PMF) is a Philadelphia chromosome-negative myeloproliferative neoplasm originating from hematopoietic stem and progenitor cells (HSPCs), frequently associated with mutations in JAK2, CALR, or MPL that lead to constitutive activation of the JAK-STAT pathway [98]. The disease is characterized by intricate clonality and dysregulated inflammation that promotes expansion of malignant clones, accelerates leukemic transformation, and induces bone marrow fibrosis through complex effects on the hematopoietic niche [98]. Recent evidence establishes mitochondrial dysfunction and PMF dysregulation as amplifiers of this pathological process.
In PMF, chronic inflammation creates a self-reinforcing cycle wherein excessive inflammatory mediator production, heightened oxidative stress, and immune dysregulation promote clonal expansion [98]. The resulting oxidative stress directly impacts mitochondrial function, with ROS-mediated damage disrupting ETC efficiency and potentially causing PMF collapse. Simultaneously, inflammatory cytokines including TGF-β, IL-1β, and NF-ĸB drive fibroblast recruitment and collagen deposition in the bone marrow, creating an increasingly hypoxic and acidic microenvironment that further compromises mitochondrial function [99] [100]. This metabolic switch favors glycolysis over oxidative phosphorylation, a phenomenon observed in many cancers that supports biosynthetic precursor generation while maintaining redox balance.
Single-cell RNA sequencing studies have revealed significant alterations in the bone marrow microenvironment of PMF patients, with distinct inflammatory gene expression patterns differentiating prefibrotic stages from overt fibrosis [98]. These changes coincide with elevated circulating cytokine levels, with specific cytokine profiles serving as poor prognostic indicators [98]. The interconnected nature of inflammatory signaling and metabolic dysregulation in PMF establishes multiple nodes for therapeutic intervention targeting both the inflammatory drivers and their metabolic consequences.
Diagram 1: PMF Pathogenesis Linking Genetic Drivers to Mitochondrial Dysfunction. This pathway illustrates how driver mutations initiate inflammatory and oxidative stress responses that disrupt mitochondrial function and PMF regulation, ultimately driving disease progression.
Current Therapeutic Landscape and Limitations
The central role of JAK-STAT signaling in PMF has established JAK inhibitors as cornerstone therapies. Ruxolitinib, a first-generation JAK1/2 inhibitor, demonstrates efficacy in reducing splenomegaly and constitutional symptoms, benefits partially attributable to downregulation of pro-inflammatory cytokines [98]. However, clinical experience has revealed significant limitations, including minimal impact on bone marrow fibrosis and mutant allele burden [98]. These limitations reflect the molecular complexity of PMF, where diverse mutational spectra and coexisting inflammatory pathways maintain disease progression despite JAK inhibition.
The therapeutic challenge is particularly evident in cytopenic PMF phenotypes, where patients present with anemia and thrombocytopenia that contraindicate standard JAK inhibitor therapy due to dose-limiting hematological toxicity [99] [101]. Fedratinib, a more selective JAK2 inhibitor with additional FLT3 inhibitory activity, demonstrates similar limitations with anemia reported in 38% and thrombocytopenia in 22% of treated patients [101]. These therapeutic challenges highlight the need for approaches that target fundamental disease mechanisms beyond JAK-STAT signaling, including the inflammatory microenvironment and associated mitochondrial dysfunction.
Table 3: Established JAK Inhibitors in Myelofibrosis Treatment
| Therapeutic Agent | Molecular Targets | Key Clinical Benefits | Major Limitations |
|---|---|---|---|
| Ruxolitinib | JAK1/JAK2 | Splenomegaly reduction (SVR35: 41.9%); symptom improvement (TSS50: 45.9%) | Worsens cytopenias; limited effect on BM fibrosis; requires platelets >100×10⁹/L |
| Fedratinib | JAK2 (selective), FLT3 | Splenomegaly reduction (SVR35: 36-40%); symptom improvement (TSS50: ≈35%) | Anemia (38%), thrombocytopenia (22%); GI toxicity; requires platelets >50×10⁹/L |
| Pacritinib | JAK2, IRAK1, ACVR1 | Efficacy in cytopenic patients (SVR35: 19% vs BAT 5%); less hematologic toxicity | Limited long-term data; ongoing safety monitoring |
Emerging Therapeutic Strategies Targeting PMF-Associated Pathways
Recognition of PMF as a disease of the entire hematopoietic niche has stimulated development of therapeutic strategies that extend beyond JAK inhibition [98]. These approaches target inflammatory signaling, stromal activation, and metabolic adaptations that sustain the malignant clone in its microenvironment.
Pacritinib represents an evolution in JAK inhibitor design, with additional inhibitory activity against interleukin-1 receptor-associated kinase 1 (IRAK1) and activin A receptor type 1 (ACVR1) that may provide broader anti-inflammatory effects [101]. In the PERSIST-1 trial, pacritinib demonstrated efficacy in patients with thrombocytopenia, achieving significant splenic volume reduction compared to best available therapy without excluding patients based on low platelet counts [101]. This expanded therapeutic window highlights the potential of multi-target approaches in high-risk populations.
Non-JAK inhibitor strategies are emerging that target specific inflammatory pathways implicated in PMF pathogenesis. CXCR1/2 antagonists inhibit neutrophil recruitment and activation, potentially mitigating their role in stromal remodeling [100]. Anti-TGF-β agents aim to disrupt a central fibrosis-promoting pathway, though their efficacy may be limited by compensatory mechanisms [99]. Immune checkpoint inhibitors targeting PD-1/PD-L1 interactions seek to reverse T-cell exhaustion observed in PMF patients, potentially restoring immune surveillance [100]. Additionally, mitochondrial-targeted therapeutics including ROS scavengers and agents that modulate PMF generation represent promising approaches for interrupting the cycle of oxidative stress and inflammatory signaling.
Diagram 2: Emerging Therapeutic Strategies in PMF. This diagram categorizes drug development approaches from established JAK inhibitors to novel mitochondrial-targeted therapies.
Experimental Approaches for Investigating PMF Dysregulation
Advanced microscopy techniques have revolutionized investigation of PMF dynamics, enabling real-time assessment of spatial membrane potential gradients and their functional consequences. Structured illumination microscopy (SIM) with potential-sensitive dyes like tetramethylrhodamine methyl ester (TMRM) allows quantification of ΔΨmito differences between cristae and inner boundary membranes [22]. This approach relies on the concentration-dependent distribution of TMRM, with low concentrations (1.35-5.4 nM) accumulating preferentially in cristae membranes, while higher concentrations (13.5-81 nM) saturate this compartment and show relatively increased IBM staining [22].
Two analytical methods enable quantification of these spatial gradients: (1) The IBM association index uses automated thresholding of reference stains (e.g., MitoTracker Green) to define mitochondrial boundaries, then calculates fluorescence intensity ratios between IBM and CM regions [22]; (2) The ΔFWHM method compares the full width at half maximum of cross-section intensity profiles for potential-sensitive and reference dyes [22]. Both approaches detect dynamic PMF changes in response to physiological stimuli, such as Ca²⁺-induced cristae hyperpolarization following histamine receptor activation [22].
Complementary techniques include real-time matrix pH imaging using targeted fluorescent pH probes, which has revealed functional coupling between AAC and ATP synthase in regulating H+ fluxes [8]. Simultaneous multi-parameter measurements correlating PMF gradients, ATP levels, and mitochondrial morphometrics provide systems-level understanding of bioenergetic adaptations in disease states [22]. For PMF research, single-cell RNA sequencing of bone marrow populations offers unprecedented resolution of cellular heterogeneity and aberrant signaling pathways, identifying novel cellular interactions and therapeutic targets within the hematopoietic niche [98].
Table 4: Key Research Reagent Solutions for PMF Investigation
| Research Tool | Specific Examples | Experimental Function | Application Context |
|---|---|---|---|
| Potential-Sensitive Dyes | TMRM, TMRE | ΔΨmito measurement based on potential-dependent accumulation | Quantifying spatial membrane potential gradients by SIM |
| Mitochondrial Reference Dyes | MitoTracker Green FM | IMM labeling independent of ΔΨmito changes | Reference channel for normalization in ratiometric imaging |
| Genetic Encoded Sensors | mt-cpYFP, mito-SypHer | Matrix pH measurement | Real-time monitoring of ΔpH component of PMF |
| ETC Inhibitors | Rotenone (Complex I), Antimycin A (Complex III) | Selective inhibition of PMF generation | Testing PMF dependency of pathological processes |
| Uncouplers | FCCP, BAM15 | Dissipate PMF by increasing H+ permeability | Assessing maximal respiratory capacity; modulating PMF |
| Single-Cell RNA Sequencing | 10X Genomics, Smart-seq2 | Transcriptomic profiling at single-cell resolution | Identifying cellular heterogeneity and aberrant pathways in PMF |
Diagram 3: Experimental Workflow for Analyzing Spatial PMF Gradients. This protocol outlines key steps from sample preparation through multi-parameter analysis for investigating mitochondrial membrane potential dynamics.
Dysregulation of the protonmotive force represents a convergent mechanism in disease pathogenesis that connects genetic lesions, inflammatory signaling, and metabolic adaptation. In primary myelofibrosis, PMF dysregulation creates a permissive environment for clonal expansion, stromal remodeling, and disease progression through interconnected cycles of oxidative stress and inflammation. Therapeutic strategies that target these fundamental processes, including next-generation JAK inhibitors with additional targets, pathway-specific anti-inflammatory agents, and mitochondrial-directed therapeutics, offer promise for overcoming the limitations of current approaches.
Future research directions should prioritize longitudinal assessment of PMF dynamics throughout disease progression, identification of biomarkers predictive of therapeutic response, and development of tissue-specific PMF modulators. Advanced techniques including single-cell multi-omics and super-resolution microscopy will continue to reveal novel aspects of PMF biology, while engineered disease models incorporating patient-derived cells will enhance translational relevance. Integrating understanding of PMF regulation with targeted therapeutic development represents a promising pathway for achieving disease-modifying outcomes in myelofibrosis and other disorders characterized by bioenergetic dysregulation.
Conclusion
The mitochondrial membrane potential and pH gradient are not merely static components of a battery but are dynamic, spatially regulated, and deeply integrated into cellular signaling networks. The foundational understanding of the PMF, now refined by advanced methodological approaches, reveals a complex system where gradients differ even within a single mitochondrion. These insights are pivotal for troubleshooting experimental data and for comprehending mitochondrial physiology in a real-world context. Future research must leverage correlative, multi-parameter techniques to further elucidate how localized PMF variations control cell fate decisions in health and disease. For the drug development community, targeting the regulators of PMF, such as uncoupling proteins or cristae junction integrity, presents a promising frontier for novel therapeutics in neurodegenerative, metabolic, and cardiovascular diseases.
References
- 1. An update of the chemiosmotic theory as suggested by possible proton currents inside the coupling membrane - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6501646/]
- 2. Mitochondrial membrane potential - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5792320/]
- 3. Mitochondrial membrane potential and compartmentalized ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12454667/]
- 4. Impact of pH-gradients on the proton uptake rate ... [https://chemrxiv.org/engage/chemrxiv/article-details/67c73ac66dde43c9087b7d1f]
- 5. Measurement of Mitochondrial Membrane Potential [https://bio-protocol.org/exchange/minidetail?id=10197213&type=30]
- 6. Analysis of mitochondrial membrane potential, ROS, and ... [https://www.sciencedirect.com/science/article/pii/S1016847825000627]
- 7. The Matrix of Mitochondrial Imaging: Exploring Spatial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11853629/]
- 8. Imaging of mitochondrial matrix pH dynamics reveals a ... [https://www.sciencedirect.com/science/article/pii/S1043661825003986]
- 9. Report Chemiosmotic ATP synthesis by minimal protocells [https://www.sciencedirect.com/science/article/pii/S2666386425000608]
- 10. Chemiosmotic vs Conformational Models of Oxidative Phosphorylation: Theory and Mechanistic Insights - ScienceDirect [https://www.sciencedirect.com/science/article/abs/pii/S0303264725002473]
- 11. Use the protonmotive force: mitochondrial uncoupling and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6170739/]
- 12. Protonmotive force - from motive protons to membrane ... [https://www.bioenergetics-communications.org/index.php/BECpreprints/article/view/gnaiger_2025_pmf]
- 13. Proton Motive Force - an overview [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/proton-motive-force]
- 14. Measuring mitochondrial membrane potential | The EMBO ... [https://link.springer.com/article/10.1038/s44318-025-00632-9]
- 15. Dynamic Regulation of the Mitochondrial Proton Gradient ... [https://www.sciencedirect.com/science/article/pii/S0021925820537861]
- 16. Mitochondrial membrane potential [https://www.sciencedirect.com/science/article/abs/pii/S0003269717302932]
- 17. Mitochondrial membrane hyperpolarization modulates ... [https://www.nature.com/articles/s41467-025-59427-5]
- 18. Mitochondrial membrane potential acts as a retrograde ... [https://www.life-science-alliance.org/content/6/12/e202302091]
- 19. Membrane potential and delta pH dependency of reverse ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6209044/]
- 20. Proton Motive Force - an overview [https://www.sciencedirect.com/topics/medicine-and-dentistry/proton-motive-force]
- 21. Quantitative measurement of mitochondrial membrane ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3448152/]
- 22. Implications of mitochondrial membrane potential gradients ... [https://www.nature.com/articles/s41598-024-65595-z]
- 23. Elevated mitochondrial membrane potential is a ... [https://www.nature.com/articles/s41467-025-57238-2]
- 24. From proliferation to mitochondrial membrane potential ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11933298/]
- 25. Mitochondrial proton and electron leaks - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3122475/]
- 26. Oxidative phosphorylation [https://en.wikipedia.org/wiki/Oxidative_phosphorylation]
- 27. Cristae remodeling causes acidification detected by ... [https://www.nature.com/articles/srep35907]
- 28. Review Mitochondrial uncoupling, ROS generation and ... [https://www.sciencedirect.com/science/article/pii/S000527281830135X]
- 29. Mitochondrial signal transduction - PMC - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC9692202/]
- 30. How and when to measure mitochondrial inner membrane ... [https://www.sciencedirect.com/science/article/abs/pii/S0006349524001760]
- 31. Mitochondrial Reverse Electron Transport: Mechanisms, ... [https://www.mdpi.com/2079-7737/14/9/1140]
- 32. Impairment of pH and gradient mediates redox... membrane potential [https://link.springer.com/article/10.1007/s00395-017-0626-1]
- 33. (PDF) Mitochondrial membrane potential [https://www.academia.edu/86058517/Mitochondrial_membrane_potential]
- 34. ARTICLES | Physiology [https://web.archive.org/web/20170926190938/http://physiologyonline.physiology.org/content/20/5/303]
- 35. The mechanisms of action of mitochondrial targeting ... [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2023.1243613/full]
- 36. Mitochondrial membrane potential probes and the proton ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3115691/]
- 37. Measurement of Mitochondrial Membrane Potential Using ... [https://www.sciencedirect.com/science/article/pii/S0006349599772140]
- 38. A mitochondria-targeted near-infrared fluorescent probe for ... [https://www.sciencedirect.com/science/article/pii/S014372082400531X]
- 39. Monitoring mitochondrial pH with a hemicyanine-based ... [https://pubmed.ncbi.nlm.nih.gov/31011741/]
- 40. Probes for Mitochondria—Section 12.2 [https://www.thermofisher.com/us/en/home/references/molecular-probes-the-handbook/probes-for-organelles/probes-for-mitochondria.html]
- 41. Long-term super-resolution inner mitochondrial membrane ... [https://www.nature.com/articles/s41589-023-01450-y]
- 42. Potentiometric Probes [https://www.potentiometricprobes.com/?srsltid=AfmBOooC3ZjcuHfWW9XvS9h-4tZnVxDONEPXVKmxYz-Q22o59TrYU0Hr]
- 43. VSD Research Application Overview [https://www.potentiometricprobes.com/research/vsd-research-application-overview?srsltid=AfmBOorCYECK8XdYQz1kbrnGzy5ZS7ZXHF_bn1Gx-zFnuF17L2jjUOlM]
- 44. Mitochondrial membrane potential assay [https://www.bmglabtech.com/en/application-notes/measuring-mitochondrial-membrane-potential-using-the-fluostar-omega-microplate-reader/]
- 45. - Super Imaging of Voltages in the Interior of Individual, Vital... Resolution [https://pmc.ncbi.nlm.nih.gov/articles/PMC10795477/]
- 46. Implications of mitochondrial on... membrane potential gradients [https://www.nature.com/articles/s41598-024-65595-z?error=cookies_not_supported&code=926db132-0250-444d-ac6e-c3bd3211d777]
- 47. Advancements and perspectives on organelle-targeted fluorescent... [https://pubs.rsc.org/en/content/articlelanding/2025/sc/d5sc04640h]
- 48. Real-time, high-throughput super-resolution microscopy ... [https://www.nature.com/articles/s41467-025-64368-0]
- 49. of Visualization membrane mitochondrial in mammalian cells potential [https://pubmed.ncbi.nlm.nih.gov/32183960/]
- 50. sciencedirect.com/topics/neuroscience/ mitochondrial -membrane... [https://www.sciencedirect.com/topics/neuroscience/mitochondrial-membrane-potential]
- 51. Calibration and measurement of mitochondrial pH in intact ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8138862/]
- 52. How and when to measure mitochondrial inner membrane ... [https://pubmed.ncbi.nlm.nih.gov/38454598/]
- 53. Implications of mitochondrial on... membrane potential gradients [https://pubmed.ncbi.nlm.nih.gov/38926476/]
- 54. A guide in the route of correlative microscopy ... [https://www.sciencedirect.com/science/article/abs/pii/S0968432821001736]
- 55. Correlative light and electron microscopy analysis for ... [https://pubmed.ncbi.nlm.nih.gov/40632481/]
- 56. A Fluorescence-Based Method to Measure ADP/ATP ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7277544/]
- 57. Measurement of ATP concentrations in mitochondria ... [https://www.sciencedirect.com/science/article/abs/pii/S0091679X19301244]
- 58. Morphological Dynamics of Mitochondria [https://pmc.ncbi.nlm.nih.gov/articles/PMC2995918/]
- 59. Mitochondrial dynamics regulate cell morphology in the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11809207/]
- 60. Cristae junction as a fundamental switchboard for ... [https://www.sciencedirect.com/science/article/pii/S0143416021001718]
- 61. Analysis of mitochondrial dynamics and functions using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3007120/]
- 62. Mitochondrial membrane potential [https://ionbiosciences.com/cell-health/mitochondrial-membrane-potential/]
- 63. Probes Useful at Acidic pH—Section 20.3 [https://www.thermofisher.com/us/en/home/references/molecular-probes-the-handbook/ph-indicators/probes-useful-at-acidic-ph.html]
- 64. Biosensing on acid: fluorescent protein probes for low pH ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9730789/]
- 65. Fluorescence saturation imaging microscopy - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC11166444/]
- 66. Investigation the concentration effect on the absorption and ... [https://www.sciencedirect.com/science/article/abs/pii/S0030402618300871]
- 67. Mitochondrial secrets revealed: UCLA-led team discovers ... [https://cnsi.ucla.edu/september-25-2025-mitochondrial-secrets-revealed-ucla-led-team-discovers-physics-of-how-cells-powerhouse-splits-to-reproduce/]
- 68. Assay - PMC Mitochondrial Membrane Potential [https://pmc.ncbi.nlm.nih.gov/articles/PMC5375165/]
- 69. Quantitative analysis of mitochondrial morphology and ... [https://www.sciencedirect.com/science/article/pii/S0167488914003917]
- 70. Methods for imaging mammalian mitochondrial morphology [https://pmc.ncbi.nlm.nih.gov/articles/PMC6322684/]
- 71. Advancements in Mitochondrial-Targeted Antioxidants [https://www.sciencedirect.com/science/article/pii/S2667137925000232]
- 72. Measurement of the Mitochondrial Membrane Potential and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3296033/]
- 73. Guidelines on experimental methods to assess ... [https://www.nature.com/articles/s41418-017-0020-4]
- 74. Impairment of pH Gradient and Membrane Potential Mediates ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5495109/]
- 75. carboxy-SNARF-1 Probe Reveals Anticooperative Reaction ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0161353]
- 76. Unbiased millivolts assay of mitochondrial membrane ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9377305/]
- 77. Exploiting mitochondrial dysfunction to overcome BRAF ... [https://www.nature.com/articles/s41419-025-07766-y]
- 78. Individual cristae within the same mitochondrion display ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6856616/]
- 79. Inner mitochondrial membrane [https://en.wikipedia.org/wiki/Inner_mitochondrial_membrane]
- 80. Measurement of the mitochondrial membrane potential and pH gradient from the redox poise of the hemes of the bc1 complex - PubMed [https://pubmed.ncbi.nlm.nih.gov/22404942/]
- 81. Review Cristae Membrane Dynamics – A Paradigm Change [https://www.sciencedirect.com/science/article/pii/S0962892420301690]
- 82. An integrative modelling approach to the mitochondrial ... [https://www.nature.com/articles/s42003-025-08381-5]
- 83. Control Over the Contribution of the Mitochondrial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2662253/]
- 84. Identification and validation of the mitochondrial function ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9732315/]
- 85. Implications of mitochondrial membrane potential gradients ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11208492/]
- 86. Mitochondrial cristae remodeling: Mechanisms, functions, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12615294/]
- 87. Modulating inter-mitochondrial contacts to increase ... [https://pubmed.ncbi.nlm.nih.gov/41279363/]
- 88. Intracellular and microenvironmental regulation of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10176868/]
- 89. Nerve-to- cancer transfer of mitochondria during cancer ... | Nature [https://www.nature.com/articles/s41586-025-09176-8?error=cookies_not_supported]
- 90. and carcinogenesis | Experimental Hematology... Cancer metabolism [https://ehoonline.biomedcentral.com/articles/10.1186/s40164-024-00482-x]
- 91. Exploring the therapeutic potential of mitochondrial uncouplers in... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8129951/]
- 92. Mitochondrial membrane potential and compartmentalized ... [https://www.sciencedirect.com/science/article/pii/S2213231725003726]
- 93. PMF-seq: a highly scalable screening strategy for linking ... [https://www.nature.com/articles/s42255-024-00994-0]
- 94. Effects of Matrix pH on Spontaneous Transient ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2021.692776/full]
- 95. Strategies for Comprehensive Multi-Omics Integrative Data ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11592251/]
- 96. Metabolomics and transcriptomics based multi-omics ... [https://www.nature.com/articles/s41540-023-00305-5]
- 97. Integration of metabolomics and transcriptomics to reveal ... [https://www.sciencedirect.com/science/article/pii/S0006291X24013913]
- 98. Chronic Inflammation in Primary Myelofibrosis - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC12629696/]
- 99. The ongoing challenges of managing cytopenic ... [https://www.dovepress.com/the-ongoing-challenges-of-managing-cytopenic-myelofibrosis-in-2025-the-peer-reviewed-fulltext-article-BLCTT]
- 100. Cytokine Landscapes, Immune Dysregulation, and ... [https://www.mdpi.com/2077-0383/14/17/6328]
- 101. The Emergence of Non-JAK Inhibitor Therapies - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12477285/]